Rapid assessment and treatment of the critically ill in shock is crucial to survival. The RUSH (Rapid Ultrasound in Shock and Hypotension) exam uses Ultrasound (US) to determine the etiology of undifferentiated shock, but data to demonstrate impact of focused US on clinical outcomes is sparse. This study aimed to assess the clinical impact of early incorporation of the RUSH US exam in the care of patients with new onset shock on a population level.
In this single center randomized control trial, sixty patients were enrolled in the RUSH group and received an early protocolized US exam within the first 24 hours of shock onset, and 65 patients were enrolled in the usual care group. The primary outcome investigated was total hospital length of stay. Secondary outcomes included in-hospital mortality, common markers of morbidity, and resource utilization.
There was no difference in the length of stay or resource utilization between the two groups, however the RUSH subjects were more likely to have a lower stage of acute kidney injury by RIFLE staging (p = 0.019). There were non-significant trends towards less total fluid administered, reduced need for new hemodialysis, and fewer ventilator days in the RUSH group.
The use of early US in shock patients leads to improvement in renal outcomes which is known to have significant impact on morbidity and mortality. Future studies are required to further investigate additional clinical outcomes impacted by use of an early protocolized US examination for this population.
Ultrasound, Shock, Kidney injury, RUSH, Focused ultrasound, Bedside ultrasound
Shock is associated with high mortality, therefore rapid assessment, recognition, and treatment of the critically ill in shock is crucial to survival. Physical exam has proven to be unreliable in accurately determining the etiology of shock [1]. Invasive procedures, such as right heart catheterization, provide real time information to aid in diagnosis, but they require specifically trained personnel and are associated with iatrogenic complications without improved outcomes [2]. Ultrasound (US) is an important non-invasive tool that is used to differentiate etiologies of shock in the emergency department (ED) and intensive care unit (ICU). The use of US has increased confidence and certainty in clinical decision-making in these arenas [3]. In a prospective study of 125 patients admitted to a general ICU, a head to toe US (optic nerve, thorax, heart, abdomen, and venous system) performed within 12 hours of admission modified the diagnosis in 31/125 (25.6%), confirmed the diagnosis in 73/125 (58.4%), did not change or confirm the diagnosis in 17/125 (13.6%), and missed the diagnosis in 3/125 patients (2.4%) [4]. US allows for narrower differential diagnoses and more accurate and timely interventions [5]. An observational study in a medical ICU showed the use of US influenced the diagnosis and workup in 33 of 150 patients (22%) with direct influence on the therapeutic plan [6]. Recent advancements in technology have made US devices increasingly portable, user friendly, and of greater image quality. US education is becoming an integral part of most critical care training centers, however the majority of physicians in critical care practice today were trained prior to the advent of bedside ultrasound fellowship education, and therefore its use is not yet pervasive or standardized. The American Society of Echocardiography (ASE) and American College of Emergency Physicians (ACEP) endorse and recommend the use of bedside focused echocardiography by the trained non-cardiologist physician (such as intensivist and emergency physicians) for evaluation of undifferentiated shock [7,8]. There was 100% consensus among experts of the European Society of Critical Care and eleven other critical care societies around the world to support mandatory basic critical care US and echocardiography for ICU physicians [9,10]. Previous studies have shown that appropriately trained non-cardiology physicians can accurately determine left ventricular ejection fraction (LVEF) using bedside echocardiography [11-15]. One can also readily use this technology to obtain basic information about right heart function, valvular anatomy and function, and the pericardium and aorta [16-19]. US can be used to evaluate pleural effusions and lung parenchyma for consolidation, pneumothorax, and edema, as well as deep veins for thrombosis [19,20]. In a prospective study of 52 patients in the Emergency Department, the RUSH protocol allowed for rapid diagnosis of shock etiology, especially in evaluation of obstructive, cardiogenic and hypovolemic shock with excellent specificity [21]. It is widely understood that focused bedside US does not replace the need for formal echocardiographic studies performed by cardiologists, rather that it assists the treating intensivist by providing real time assessment of the patient's condition in a time sensitive manner. Based on several reports, training for basic critical care US is not as time consuming as many have perceived historically [7,22-24]. Training guidelines set forth by ASE recommend a mixture of normal and pathologic cases with ongoing continuing education and practice [7]. At Stanford University, it has been demonstrated that an average of 15 hours of training and 30 scans are required for first year critical care fellows to gain competency in basic critical care US [22]. Certainly, inappropriate use of US can lead to misdiagnosis with potential for incorrect choice for therapy [25], but provider common sense and clinical judgement, along with reviewing images with more experienced operators and continuing education promotes appropriate use. In a different study, 12 hours of training including a combination of didactics, interactive cases, and hands on training were sufficient to gain competency in critical care US for the non-cardiology physician [23].
There have been multiple US protocols described for the evaluation of the unstable patient including FATE (Focused Assessed Transthoracic Examination), eFAST (Extended Focused Assessment with Sonography in Trauma), BLUE-FALLS (Bedside Lung US in Emergency - Fluid Administration Limited by Lung Sonography) and RUSH (Rapid US in Shock and Hypotension) to name a few. There are a number of RUSH protocols described in the literature including RUSH [19], RUSH HI-MAP [26] and RUSH VTI [27]. The RUSH exam described by Perera, et al. [19] uses standard US technology to evaluate the pump (cardiac status), the tank (effective intravascular volume), and the pipes (great vessels) to help determine the etiology of undifferentiated shock. The RUSH protocol is a comprehensive protocol that includes components of several focused protocols which makes it a good choice for the assessment and re-assessment of critically ill patients [28]. To date, there are no prospective randomized studies to investigate the clinical outcomes of implementing such an US protocol in patients with shock on a population level. There are numerous case studies pointing to the fact that use of RUSH has led to earlier recognition of pathology and therefore more timely and successful intervention, but there is a paucity of literature regarding whether use of US on a widespread level will change outcomes for critically ill patients as a whole. This study hypothesized that the addition of early, protocolized point of care bedside US (RUSH) by intensive care physicians would lead to improved ICU health outcomes for the population of patients with new onset shock. The focus of this investigation, unlike prior RUSH studies, is not on whether the US properly diagnosed each subject, but whether use of RUSH US for all-comers presenting to the ICU with shock would make a difference for the group as a whole in terms of their LOS, survival, and morbidity outcomes.
This study was a single center, prospective, randomized, controlled trial with intention to treat analysis aimed at assessing the clinical impact of early incorporation of the RUSH US exam in the care of patients with new onset shock. The study took place in the medical intensive care unit (MICU) at Ronald Reagan Hospital, University of California, Los Angeles (UCLA) from May 2014 to January 2015. Study data was collected and managed using REDCap electronic data capture tools hosted at UCLA Medical Center [29]. REDCap (Research Electronic Data Capture) is a secure, web-based application designed to support data capture for research studies.
Prior to beginning enrollment for the study, designated Pulmonary Critical Care fellows and physicians were trained and certified through a formal curriculum established and studied at the University of California, Irvine [30] and implemented at UCLA. The curriculum included 6 hours of didactic lectures, 14 hours of didactic curriculum with pre and post testing via US simulation technology, 14 hours of hands on simulation, 8 hours of hands on mentored sessions on healthy human volunteers and at least 30 US examinations on ICU patients with various pathologies (i.e. pulmonary edema, pleural effusion, hyperdynamic heart function, pneumothorax, reduced heart function, urinary retention and normal physiology) that were recorded and reviewed by the Director of Bedside Ultrasound. The training was completed with a practical test that evaluated skills in acquisition, interpretation, and implementation of treatment plans based on findings. All operators were trained sufficiently to gain competency in critical care US for the non-cardiology physician.
The point of care US equipment used in this study was that which was routinely used in the MICU for various uses, both diagnostic and therapeutic. There were minimal procedural risks involved in the use of bedside US as it is widely acknowledged to be non-invasive, does not expose the patients to ionizing radiation, and uses hypoallergenic water-based gel coupling solutions. The procedure was performed in a clean manner with careful disinfection between patients. The foreseeable risks associated with US in this study included discomfort from cold gel on the skin and physical pressure from the sonographer during the exam, both of which were transient and resolved at the termination of the US exams. The exam was projected to last 7-10 minutes on average. A waiver for HIPAA authorization was obtained based on minimal risk, the emergent nature of evaluation of patients in shock, and patients' inability to consent adequately when in extremis. Every patient or patient's representative was provided a study information sheet either before or after enrollment and had the ability to withdraw their data from the study at any point.
Trained research assistants screened MICU patients twice daily to identify eligible patients for inclusion in the study. Inclusion criteria included a diagnosis of new onset shock within the previous 24 hours defined as follows: Lactate > 4 mmol/L, fluid resuscitation > 30 ml/kg in a 6-hour time span over the previous 24 hours with a persistent systolic blood pressure (SBP) < 90 mmHg or mean arterial pressure (MAP) < 65 mmHg, or addition/escalation of pressor use. Patients with a hospice or comfort care designation, length of stay (LOS) greater than 5 days at time of shock onset, total hospital LOS less than 1 day or greater than 14 days determined retrospectively after inclusion, pregnancy, age less than 18, and unwillingness to have their data included for analysis once informed about the study were excluded. Qualified patients for the study were enrolled and randomized via drawing of an assignment from a sealed, shuffled, and numbered set of envelopes.
Patients were enrolled in one of two groups: RUSH Intervention or Usual Care (UC). Patients in the RUSH Intervention arm received a protocolized RUSH exam as described by Perera, et al. [14] within the first 24 hours of shock onset. The RUSH exam was performed as soon as was feasible by operators who were trained and certified physicians as previously described. The outcome of the examination was reported to the primary physician caring for the patient and documented in a standardized template in the electronic medical record (EMR). The primary team was blinded to a patient's randomization until RUSH results were conveyed, so treatment decisions were not delayed pending performance of the US exam and management plans occurred solely at the discretion of the primary team. Patients in the UC arm were eligible to receive an US exam at the discretion of the patient's primary physician at any time, but the performance of the examination was not mandated to occur (Figure 1), and its use was largely dependent on the experience and training of the primary treating physician. Those in the UC arm were evaluated using standard ICU modalities based at the discretion of the treating physician, including physical exam, formal radiological and laboratory studies, central venous pressure monitoring, and bedside US, among others. Patient demographics and outcomes data collected for analysis is outlined in Table 1.
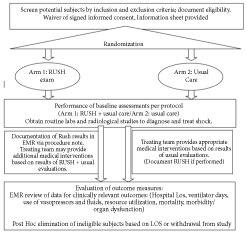 Figure 1: Study design and patient enrollment. View Figure 1
Figure 1: Study design and patient enrollment. View Figure 1
Table 1: Patient outcomes measured. View Table 1
The primary outcome studied was total hospital length of stay. Secondary outcomes included in-hospital mortality and markers of morbidity including acute respiratory failure requiring mechanical ventilation, shock duration, incidence of myocardial infarction, cardiopulmonary arrest, acute kidney injury (AKI), and resource utilization (i.e. laboratory evaluations, number of formal echocardiograms, and radiographic images). These morbidity indicators were chosen because of their potential to be impacted by variable management of shock. All measures were conclusively evaluated after the patients' discharge to allow for complete and accurate reflection of in-hospital outcomes, as well as to enable exclusion of patients whose hospital stays were < 1 or >14 days in total.
In order to achieve 80% power to detect differences in LOS with a two sample t-test with a 0.05 two sided significance level (factoring in a mean LOS of 4 days with a standard deviation of 2.7 based on UCLA MICU data over the two previous years and assumption of approximately 25% intervention effect), the study was designed to require a sample size of 190 per group. Because the use of the RUSH exam was not restricted in this intention to treat study, a predicted 20% crossover rate was factored into the sample size calculation.
The primary analysis was conducted as an intent-to-treat and compared the randomly assigned groups. Bivariate differences between the RUSH intervention and UC group were compared using Welch's t-test (for continuous variables) and Fisher's exact test (for discrete variables). Continuous variables that were not normally distributed, such as total intravenous (IV) fluid intake and peak lactate, were log transformed and described using geometric means and standard deviations. Multivariable linear and logistic regression models were used to assess for differences between the intervention groups after adjusting for age, gender, race, and weight. All analyses were conducted using Stata Version 13.1, Stata Corp LP (College Station, TX).
Although the optimal sample size for a fully powered study was calculated to be 380, the interim statistical analysis after 8 months of data collection revealed low power for the primary aim of detecting differences in LOS. Careful evaluation of the study design and observation of logistical obstacles led to the conclusion that design flaws in the study needed to be addressed before proceeding, and therefore the study was terminated early. For example, the deviation from clinical practice where the bedside ultrasonographer is typically the treating physician and the allowance of a 24 hour evaluation window when in reality the RUSH protocol is usually performed in the first 6-8 hours of new shock were felt to reduce the potential impact of the intervention. Despite the early study closure, the interim data analysis that was performed with the aim of leading to an improved study design revealed intriguing findings, and some compelling significant results and trends were found before the point of study closure that deserve attention, study redesign, further discussion, and exploration.
Demographically, there were no statistical differences between the two groups (see Table 2). The mean age was 60.4 years in the RUSH arm and 64.0 years in the Usual Care arm (p = 0.26). The gender breakdown in the RUSH group was 50% female compared to 38% females in the usual care group (p = 0.19). The two groups were also similar in prognostic scoring systems. The mean APACHE IV score was 79.6 in the RUSH group vs. 80.9 in the control group (p = 0.830). Additionally, the mean Charlson Comorbidity Index was 5.47 in the RUSH arm vs. 5.52 in the usual care arm (p = 0.91). Actual diagnoses were not recorded in this study as it is more difficult to compare patients based on diagnoses than by using objective scoring systems such as APACHE IV and Charlson Comorbidity Index. Based on the primary provider's medical decision making, there was cross-over of the groups such that 6 participants randomized to the RUSH intervention group received only Usual Care and 10 participants randomized to UC group received the RUSH intervention within the first 24 hours which represents a 13% crossover, less than the 25% crossover expected and allowed for in statistical analysis.
Table 2: Patient demographics. View Table 2
There was no statistically significant difference between the two study arms in LOS from shock onset to ICU discharge (p = 0.26) or shock onset to hospital discharge (p = 0.61). There was, however, a statistically significant difference in acute kidney injury incidence by RIFLE staging, with the RUSH group more likely to fall into the less severe stage 1 AKI category as compared with the Usual Care group with a higher incidence of stage 3 AKI (p = 0.019), Figure 2. This difference in AKI staging remained significant when adjusted for age, gender, race, and body weight using a logistic regression model (p < 0.05). Although the total intravenous (IV) fluid intake between the two groups was not statistically different, the RUSH group showed a tendency for less total volume and more tightly controlled delivery of IV fluids (11.5 ± 6.7 L vs. 13.4 ± 15.3 L, p = 0.376). When total IV fluids were adjusted for age, gender, race, and weight, the RUSH group received significantly less IV fluids (32% less). Additionally, there was a non-significant trend towards a lower incidence of new hemodialysis (HD), fewer days on mechanical ventilation (Figure 3) and shorter duration of vasopressor use in the RUSH group, Table 3.
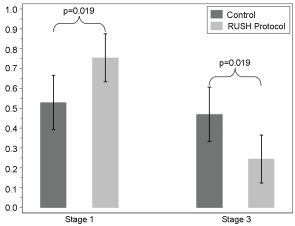 Figure 2: AKI stage 1 (RIFLE staging) was significantly higher in the RUSH group compared to US (control group). AKI stage 3 was significantly higher in the US (control group) vs. RUSH. The probability (proportion) of participants with AKI are presented (y-axis) for the Control and RUSH treatment groups. Values are presented as means with 95% confidence intervals.
View Figure 2
Figure 2: AKI stage 1 (RIFLE staging) was significantly higher in the RUSH group compared to US (control group). AKI stage 3 was significantly higher in the US (control group) vs. RUSH. The probability (proportion) of participants with AKI are presented (y-axis) for the Control and RUSH treatment groups. Values are presented as means with 95% confidence intervals.
View Figure 2
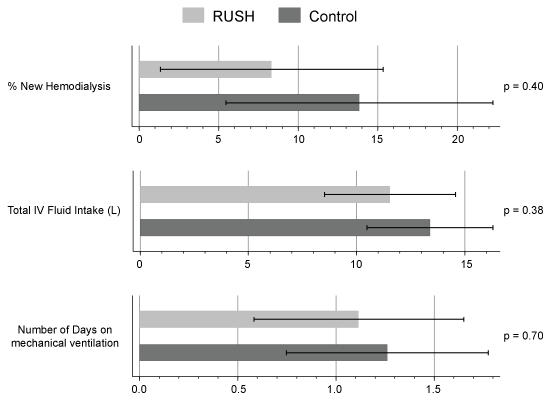 Figure 3: There was lower incidence of new hemodialysis, less total IV fluid intake, and fewer days on mechanical ventilation in the RUSH group vs. US (control), however these differences were not statistically significant. Means/proportions with 95% Confidence Intervals presented in this figure. View Figure 3
Figure 3: There was lower incidence of new hemodialysis, less total IV fluid intake, and fewer days on mechanical ventilation in the RUSH group vs. US (control), however these differences were not statistically significant. Means/proportions with 95% Confidence Intervals presented in this figure. View Figure 3
Table 3: Patient Outcomes. View Table 3
There was no significant difference in mortality during the hospitalization between the two groups, and there was no significant difference in the resource utilization as assessed by the number of echocardiograms, laboratory measures, or radiological tests ordered between the two groups (Table 4).
Table 4: Resource utilization. View Table 4
Focused ultrasound has become an invaluable and accepted diagnostic tool in critical care settings worldwide. There is an US in almost every emergency department and intensive care unit which makes this device immediately available for patient care and an invaluable tool in assessment of undifferentiated shock. Information obtained from focused US provides key physiological and anatomical data necessary to rapidly identify the potential causes of instability and to plan appropriate treatments. Current literature strongly supports the ability of non-radiologists to accurately obtain focused images, interpret the resulting data, and incorporate the resulting knowledge into their diagnostic arsenal.
The ability of US to detect pathology is not in question at this point in the literature. However, the data to demonstrate the impact of focused US on clinical outcomes for populations of patients is sparse to date. This study aimed to determine if systematic incorporation of US has an impact on meaningful outcomes in the critically ill population with shock. We hypothesized that an early protocolized US approach would add precision to diagnostic and treatment efforts and ultimately have a significant impact on LOS, morbidity, and mortality of the shock population as a whole. In our study, patients who received the RUSH protocol US evaluation in the first 24 hours of shock had significantly lower rates of acute kidney injury. This outcome may have been related to the more conservative and precise fluid management strategy for patients in RUSH US group who received 32% less IV fluids after controlling for age, gender, race, and weight, but further study is needed to elucidate cause and effect. Often patients in shock are treated with significant and sometimes excessive amount of fluids, and current literature has described that tissue edema results from positive fluid balance [31], which in turn leads to worsening organ dysfunction. Positive fluid balance is associated with worse outcomes [32] and careful fluid assessment and delivery in shock is crucial. Along the same lines, patients in the RUSH group of this study showed a tendency towards a lower incidence of new HD, fewer days of mechanical ventilation, and shorter duration of vasopressors at the end of resuscitation. These results are congruent with other studies that have suggested improvement in outcomes in patients receiving focused US, though prior studies have been mostly case reports or retrospective and observational in nature. For example, a retrospective non-randomized study looked at 220 patients divided into standard (S) shock management compared to limited echocardiography (LE). This study showed an increased 28-day survival in the LE group who received significantly less IV fluids during the initial 24-hour period from shock recognition and had significantly lower rate of stage 3 kidney injury [33].
From this study, we have learned that the use of US for shock patients early in their assessment and care leads to improvement in renal outcomes which is known to have a significant impact on morbidity and mortality. Prior evidence suggests even small rises in serum creatinine are associated with increased mortality, [34] and this risk is known to increase with higher degrees of kidney injury. There has been reported a four-fold increase in mortality [35], an increased length of stay, and higher costs associated with AKI [36]. The improved renal outcomes with use of US in this study may be due to earlier determination of shock etiology and the subsequent ability to improve perfusion to vital organs with a more tailored fluid approach. Other markers of improvement in morbidity and mortality did not reach significance.
This study did not show a difference in the primary outcome of length of stay, either from shock to hospital discharge or shock to ICU discharge. As was evident from the assessment of resource utilization during the ICU stay, regardless of the randomization, each group received the same intensity of care such as overall density of formal radiological assessment or laboratory evaluation. Therefore, in this investigation, RUSH US did not fast track patients through the ICU by reducing the time needed for routine and traditional assessments. This held true for patients regardless of code status. Additionally, the Charlson Co-morbidity and APACHE scores between the two groups were similar, so by extension, the mortality and length of hospital stay of the groups would reasonably be expected to be similar as was the case in this study. This study only followed the patients for 30 days after discharge, so the long-term mortality impact of early RUSH US was not evaluated but given the known association of AKI with increases in mortality, future investigations would be wise to plan extended follow up on this point.
There are weaknesses of the study that might have impacted the results and influenced possible conclusions, and which contributed to the decision for early cessation of data gathering. In this study, the manner in which the US operators were utilized likely mitigated the full potential effect of inclusion of the RUSH protocol. In order to avoid measurement bias, the study was conducted exclusively by operators who were not directly involved in patient's care. As a result, the operators had minimal knowledge of the clinical background of study patients and did not have the ability to use pre-test probability in their RUSH approach. Also, the protocol of this study only permitted study operators to relay US findings and recommend further management based on the US results, but it did not allow sonographers to be involved with the primary team's ultimate treatment plan or decisions. This arrangement did not simulate the realities of the bedside ultrasonographer's role as an integral part of a treatment team. Therefore, the impact of the information from the RUSH study was dependent on the primary team physician's timely reception and perception of the importance of the information provided from the RUSH protocol. All US findings were documented immediately in the Electronic Medical Record (EMR) after each examination, however the operators did not consistently have the opportunity to directly discuss findings with primary treating physicians. The communication gaps likely decreased the potential impact of the US data on treatment and management decisions. The act of performing a bedside focused US is not where the benefit of the modality arises, rather it is the incorporation of the data into the clinical context in order to tailor treatment decisions that gives US the potential to impact outcomes. It is vitally important to efficiently and effectively convey the US results to the primary treating physician in order to gain the maximal benefit from the bedside US in the management of shock, and in this study, this was an area of the study design that was not adequate.
There were several suggestive patterns in the data that did not reach the level of significance, but they were nonetheless intriguing. Study design improvements may allow these variables to be studied in more detail and show that positive impact of RUSH evaluation reaches much further than the scope of this study. Future studies should focus on further development and exploration of the outcomes related to measures of renal function including incidence of new hemodialysis, fluid balance and composition, duration of mechanical ventilation, and ICU length of stay.
Additionally, in order to better evaluate the true impact of the US intervention, it would be advisable to allow or even require that the trained ultrasound operators be members of the treatment team to provide a more realistic scenario and improve timely communication and incorporation of findings into the treatment plan. If it is not possible to have trained operators available at all times, development of a formal, rapid, and direct method of communicating imaging results between the US operator and the treating team may lead to more timely and impactful responses to the information when compared to communication via procedure note that one cannot ensure has been seen or considered in the care plan.
To best approximate clinical practice and to provide an accurate evaluation of the impact of US, it is recommended that future studies incorporate RUSH US within the first 6-8 hours of shock as compared to the 24-hour outside time window of this study. The shorter time window of 6-8 hours is likely the "golden hour" equivalent in non-traumatic shock evaluation and treatment.
Options for US reassessments, systematic and timely over-reading of stored images, and serial studies during the first 24 hours of shock may provide more accurate and realistic outcomes benefits as well.
Although it is more objective and meaningful to compare patients based on objective scoring systems such as APACHE IV, it would be interesting and possibly important to note actual shock diagnoses in future studies to evaluate whether certain subsets of patients benefit more from the intervention, however this would not change the clinical practice of evaluating undifferentiated shock using the RUSH US protocol in the initial presentation of shock symptoms.
Lastly, though length of stay and resource utilization during the 30-day window of this study were not different between groups, long term mortality and morbidity at 3 and 6 months may differ given the noted improvement in renal outcomes in the RUSH intervention group, so an extended follow up for future studies would be recommended.
The authors declare that there is no conflict of interest regarding the publication of this article.