The terms diabetic myonecrosis, diabetic myositis, and diabetic muscle infarction all describe the same microangiopathic rare complication associated with poorly controlled diabetes mellitus [1]. Diabetic myonecrosis is so rare that less than 200 cases have been reported [2]. Diabetic myonecrosis usually presents as a confusing picture of proximal lower extremity edema and extreme pain, with non-specific laboratory values. This presentation of symptoms is often initially treated with prolonged antibiotics for suspected infectious etiologies, and extensively worked up to rule out autoimmune etiologies, before making an accurate diagnosis [3,4]. Diabetic myositis is most common in middle-aged adults, with mean A1c > 9%, and is a complication known for both type 1 diabetes mellitus (T1DM), and type 2 diabetes mellitus (T2DM) [5]. The most common sites of injury are the anterior thigh in 55% of cases, followed by the calf and posterior thigh [1]. Increasing awareness of diabetic myonecrosis in patients with longstanding and poorly managed diabetes mellitus is crucial to continue diagnosing and treating this severe complication.
A 45-year-old male with a past medical history of uncontrolled T2DM, heart failure with reduced ejection fraction (left ventricular ejection fraction < 20%), coronary artery disease (managed with atorvastatin), atrial fibrillation, hypertension, polysubstance use disorder, status post left second toe amputation, initially presented to the emergency department (ED) with 4 weeks of left lower extremity (LLE) pain, erythema, and chills. Prior to presentation, he was treated with antibiotics. On physical exam, the patient was afebrile with a heart rate of 100 beats per min (BPM) and blood pressure of 136/107 mmHg. Physical exam was remarkable for erythema and swelling present from the medial aspect of the left knee to the inguinal crease. Pitting edema was present on the foot, ankle, and leg. An open wound was also present over left second digit amputation site with fibrinopurulent exudate. Dorsal pedal, popliteal and femoral pulses were palpable. The patient did not have a leukocytosis and hemoglobin was low at 11.7 g/dL. Glucose was found to be high at 243 mg/dL and HbA1C > 9%. Creatine Kinase (CK) was normal. C-reactive protein (CRP) was elevated at 42.1 mg/L. Erythrocyte sedimentation rate (ESR) was elevated at > 50 mm/h.
Throughout admission, despite IV antibiotic treatment, the patient eventually developed a leukocytosis with peak white blood cell (WBC) 17.2 K/uL, CRP peaked at 157 mg/L, and ESR peaked at 58. The patient was treated with broad-spectrum antibiotics. Computerized tomography (CT) findings of LLE included extensive subcutaneous swelling in the left leg, and no evidence of abscess, necrotizing fasciitis, or acute osteomyelitis. CT pelvis and lumbar spine were completed for increasing concern of left hip and thigh pain, and the most notable findings included continued subcutaneous edema in the left side with possible myositis of the left proximal thigh musculature. Surgery teams assessed the patient and concluded there was no suspicion for large vessel ischemia of LLE or necrotizing fasciitis. Due to ongoing severe pain in LLE, worsening leukocytosis, and tachycardia, magnetic resonance imaging (MRI) of the left femur was ordered. MRI demonstrated widespread muscle edema with bulging contours to the rectus femoris and loss of internal architecture of the musculature. Findings were also concerning for severe inflammation/infection/ischemic injury, however muscle viability was difficult to assess without contrast administration. Widespread subcutaneous edema was present without discrete fluid collection. The patient was then taken for left thigh tissue debridement, excision and muscle biopsy. Visible surgical diagnoses from the procedure included edematous muscle and fascia without purulence with patchy ischemia of muscle bellies notably involving sartorius and rectus femoris. The initial perioperative diagnosis was ischemic myositis visualized in the operating room without evidence of necrotizing fasciitis. Muscle biopsies were sent for pathology and culture. Orthopedic oncology was consulted to rule out a possible malignant process and the overall opinion was presumed diabetic myonecrosis in a poorly controlled diabetic. Unfortunately, the initial biopsy was inconclusive possibly secondary to necrotic tissue, so the patient was taken for a repeat biopsy.
Differential diagnoses discussed throughout admission were diabetic myonecrosis, pyomyositis, cellulitis, viral myositis, autoimmune myositis, and ischemic myositis secondary to poor perfusion. Autoimmune etiology was negative with normal CK, negative antinuclear antibody (ANA), and a normal C3/C4. The most likely diagnoses while awaiting final pathology results were diabetic myonecrosis vs infectious cellulitis. Leukocytosis, increasing CRP, and elevated ESR throughout admission initially pointed toward the more common etiology of an infectious process. However, when considering the patient remained with negative blood cultures, soft tissue cultures, and the prolonged antibiotic course had not improved the clinical picture, there was no evidence that an infectious process was likely and a decision was made to discontinue antibiotics. Diabetic myonecrosis became the most likely diagnosis with supporting evidence of persistent leukocytosis, persistently elevated ESR, and elevated CRP all related to ongoing inflammation, rather than infection. The patient was discharged with the mainstay of treatment for adequate control of diabetes with home insulin, pain control, and instructions to rest. Final muscle biopsy pathology reports returned the final diagnosis of left rectus femoris skeletal muscle with focal necrosis, diffuse muscle atrophy, acute inflammation, florid reactive myofibroblastic proliferation and connective tissue with vascular congestion, mild acute inflammation and no neoplastic process was identified. This diagnosis was confirmed by pathology consistent with diabetic myonecrosis (Figure 1).
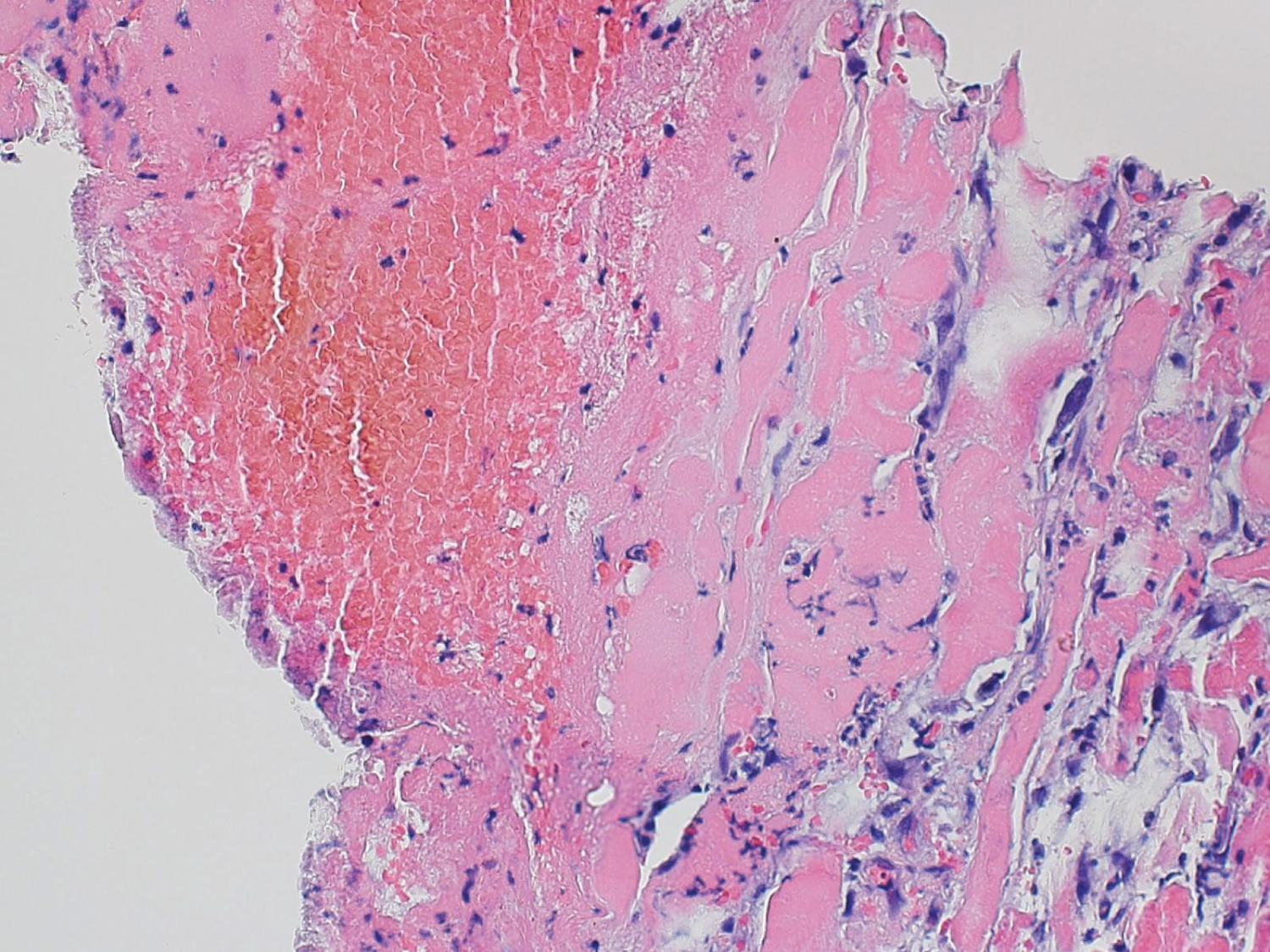 Figure 1: Higher magnification image showing area of pale staining muscle fibers most consistent with acute myonecrosis (hematoxylin and eosin stained formalin fixed paraffin embedded tissue section; 400x original magnification).
View Figure 1
Figure 1: Higher magnification image showing area of pale staining muscle fibers most consistent with acute myonecrosis (hematoxylin and eosin stained formalin fixed paraffin embedded tissue section; 400x original magnification).
View Figure 1
The final diagnosis of diabetic myonecrosis in our patient was made as a diagnosis of exclusion confirmed by muscle biopsy. With final pathology results and ruling out other causes of muscle injury such as autoimmune, infectious, and drug-induced processes, we believe this patient to be a true case of diabetic myositis. Ultimately, methamphetamine-induced muscle injury or statin induced myositis were considered not likely in our patient. The patient had not used methamphetamine for three weeks before presentation and CK was normal, making rhabdomyolysis unlikely [6]. Atorvastatin induced myositis was also less likely as CK was normal [7]. Specific subtypes of statin-induced myositis can be confirmed with anti-HMG-CoA reductase autoantibody and was not assessed in our patient, however, muscle biopsy pathology was not consistent with a diagnosis of statin-induced myositis [8]. Characteristic muscle biopsy of statin-induced myositis is consistent with necrosis with the regeneration of muscle fibers and scarce inflammation, mainly composed of macrophages, which were not seen in our patient’s pathology. Furthermore, endothelial membrane attack complex (MAC) deposition in non-necrotic fibers and MHC class I expression are additional pathologic features of this condition, and with the absence of these immune mediators in our patient’s pathology, statin-induced myositis was further ruled out [9]. In an analysis of 127 cases of diabetic myonecrosis, ESR and CRP were assessed in the majority of cases, and in the cases assessed, ESR was elevated in 83.3% and CRP was elevated in 90% of cases [1]. For our patient throughout admission ESR and CRP were persistently elevated. Furthermore, as seen in larger case analyses, CK was not elevated in a majority of patients diagnosed with diabetic myonecrosis, and CK was similarly not elevated with our patient [1].
It has been suggested, while no specific diagnostic criteria exist, a clinical diagnosis of diabetic myonecrosis can be made through history, examination, and supportive MRI findings. On previous systemic review of cases of myonecrosis, muscle edema with hyperintensity is found in a majority of cases on T2 imaging sequences of MRI [10]. Although the MRI read for our case was unfortunately limited by lack of intravascular contrast which reduced the ability to assess for the absence of muscle enhancement, it was noted that the rectus femoris muscle in our patient appeared to be “relatively swollen with bulging contours to its fascia concerning for severe edema.” Furthermore, loss of intramuscular septae is also a common known finding on T1 imaging of diabetic myonecrosis [11]. T1 imaging in our patient demonstrated a loss of intramuscular architecture, consistent with known characteristics of T1 imaging of diabetic myonecrosis.
While muscle biopsy can provide a definitive diagnosis, it is not currently recommended due to the risk of procedure-associated complications and an associated increase in time to symptomatic improvement, however, when patients do not improve with symptomatic management or the case is atypical biopsy is warranted [12-14]. In our patient, diabetic myonecrosis was not considered until later stages of workup after muscle biopsy was completed and awaiting final pathology results. Further complicating is, because this is such a rare diagnosis, management is not standardized once the diagnosis is made. Historically, conservative management including rest, analgesia, and further glycemic control has proven to have good outcomes [15,16]. However, NSAIDs are often avoided due to concomitant renal disease, and analgesia is controlled with opioids [17]. Physical therapy and surgical resection have been shown to further prolong and complicate the course, and bed rest is preferred once diagnosis is made [18].
The choice to manage long term with opioid analgesia is complicated for patients with a history of substance use and efforts to stay abstinent from substance use, as was concerned with our patient. Recurrence rates for diabetic myonecrosis are high and can be referred to as “migratory diabetic myositis,” as the initial site of myonecrosis is often not the repeat culprit. Often, patients present with contralateral disease after initial diagnosis months later, making this a repeated complication for patients with poorly managed diabetes [11,18].
In 2022, the Centers for Disease Control and Prevention (CDC) reported over 11.8% prevalence of diabetes in the United States [19]. This prevalence is predicted to increase by over 54% by 2030, meaning in turn, complications associated with diabetes will also increase [20]. In relation to these increasing statistics, our case brings attention to a classic presentation of the uncommonly made diagnosis of diabetic myonecrosis. The aims of this case report are to continue discussing this microangiopathic complication known to impact patients with poorly controlled diabetes mellitus, creating awareness for clinicians to add this diagnosis to a differential and consider conservative management before invasive muscle biopsy when MRI imaging and patient history suggest this underlying etiology.
None.