Aim: The aim of this study is to assess the utilization of CGMs in a GMV setting to improve patient activation and engagement, quality of life, and metabolic outcomes including glycemic control through modified lifestyle behaviors in patients with Type 2 diabetes.
Background: Group medical visits (GMV) are an evidence-based care model shown to improve outcomes in diabetes. Monitoring tools that readily demonstrate cause-and-effect relationships between recommended lifestyle interventions and optimal glycemic control in the short and long term can be utilized to improve diabetes outcomes. Continuous glucose monitoring systems (CGMs) are wearable devices that continuously measure interstitial glucose and provide valuable feedback on glycemic changes in response to diet and exercise. With this data, patients are given nearly immediate feedback, empowering them to make dynamic behavioral changes.
Methods: We implemented GMV for 16 adults from an academic community-based primary care clinic. Two separate cohorts consisting of 8 participants each, met for a total of 14 weeks with sessions every 2 weeks. Baseline HbA1c and lipid panels were collected within 3 months of the first GMV and at the end of the 14 weeks. Validated questionnaires (PAM-13 and SF-12) were collected at the initial and final GMV. Paired t-tests for these differences between the two times were also calculated. Significance was set at p < 0.05.
Findings: At the end of the 14 weeks, participants had a significant reduction in HbA1c (0.4%; p < 0.05), weight (6.4 lbs; p < 0.05), and average glucose (calculated by CGM, 21.1 mg/dl; p < 0.05). 38.5% of participants decreased their HbA1c to the prediabetes range. There was no significant difference for lipids, PAM-13, or SF-12 or blood pressure. Participants reported subjectively that the CGM was helpful to see the relationship of foods and their glycemia.
The American Diabetes Association Standards of Care in Diabetes 2023 highlights the importance of providing patients with the counseling, tools, and resources for individualized self-management to achieve optimal glycemic control [1]. Providers face barriers to translate available tools and resources into improved diabetes outcomes [2]. Traditional care models place significant limits on the development and support of effective behavior change required to reach desired outcomes [2,3]. Patients with diabetes have low levels of confidence in their self-care skills and limited understanding of the relationship between lifestyle choices and key health parameters such as weight and blood glucose [4,5].
The patient activation measure (PAM), a validated tool used to quantify the level of patient engagement, is strongly correlated with improved health outcomes and decreased health costs [6,7]. Group medical visit models (GMV), reconfigure care to allow patients to share visit time with providers and each other. The increased time and sharing supports engagement in decision-making and the development of a valued partnership with the healthcare system [8]. This likely contributes to the demonstrated improvement in metabolic parameters including HbA1c and blood pressure in patients with diabetes [8]. Continuous glucose monitoring systems (CGMs) provide timely feedback on glycemic changes in response to diet and exercise. Blood glucose readings are approximately five to ten minutes ahead of interstitial glucose readings measured by the CGM [9]. Armed with an understanding of these data, patients can make dynamic behavioral changes.
Independently, both the GMV model of diabetes care and utilization of CGM technology have demonstrated lasting effects on patient outcomes [8,10-12]. However, the impact of CGM technology within the GMV paradigm has yet to be studied. The combination of GMVs and CGMs into a comprehensive care model may serve as a platform for the following functions: (1) Patient-provider discussions on glycemic graphs generated from 14-day CGM data between visits; (2) Demonstration of relationships between food and activity choices and glycemic excursions; (3) Discussion of methods to achieve individual goals for glucose time-in-range; and (4) Education on the deleterious effects of hyperglycemia. We hypothesize that incorporating CGM within diabetes GMV will maximize patient activation while simultaneously improving patient engagement, quality of life, and metabolic outcomes including glycemic control through modified lifestyle behaviors. Our primary objective is to compare baseline and 14-week variables, including biometric, metabolic, and quality of life measures.
The study was reviewed and approved by the Wake Forest Institutional Review Board. Participants were provided information on the study and written consent was obtained prior to participation. This was a feasibility study of existing life-style modification GMVs for patients with type 2 diabetes mellitus (T2DM). The inclusion criteria included patients older than 18 who were English speaking, had a hemoglobin A1c (HbA1c) between 6.5%-8%, and willing to wear a CGM. An academic community-based primary care clinic patient panel was queried for the inclusion criteria and an investigator called the possible participants sequentially until the sample size criteria were met.
Outcome variables including HbA1c and lipid panel were collected within three months of the first GMV and again at the end of the study. Weight and blood pressure were also obtained at the initial and final GMV. Participants completed brief questionnaires pre-study (at the first meeting) and post-study (at the last meeting). One questionnaire contained open-ended items eliciting participants' opinions about wearing a CGM and if they thought the device would improve their glucose control or change their meal plan. Two other questionnaires were used including the PAM-13 and the SF-12. The PAM-13 is a patient engagement tool that measures patient knowledge, skill, and confidence for the self-management of diabetes [13,14]. The SF-12 measures health-related quality of life [15].
The CGM used in this study was the FreeStyle Libre 14® day system supplied by Abbott Diabetes Care. This company had no role in the design, analysis or publication of this study. Two GMV series consisting of eight participants were conducted sequentially in time. Each group received seven total GMV sessions spaced two weeks apart for a total of 14 weeks of intervention. The participants were familiarized with the CGMs at the initial visit and were advised to continue their normal diet and lifestyle for the following two weeks. CGM data was uploaded to an online portal or data were downloaded at the beginning of the GMV. For each two-hour GMV, the initial 30 minutes were used to review glucose graphs of participants from the prior two weeks and the remainder of the visit was used in a variety of different formats, including question and answers, group discussions, lectures, and interactive conversations. Specific instructions were given about how participants can leverage their real-time CGM data to facilitate lifestyle changes.
Descriptive statistics were generated to portray the participants' demographic characteristics. CGM Time 1 includes the first two weeks of glucose data and Time 2 contains the last two weeks of glucose data during the study period. These statistics include mean or median values at Time 1 and Time 2; the mean differences of the variables from study period from Time 1 to Time 2; and the standard deviation of that difference. Paired t-tests for these differences between the two times were also calculated, using IBM SPSS Version 26. Significance was set at p < 0.05.
Table 1 presents descriptive characteristics of the participants. The median age of participants was 55 years with a median duration of diabetes of 54 months. 62.5% of participants were female, 37.5% were Black and 62.5% were non-Hispanic White. Two participants were on zero medications for diabetes and 11 out of 13 were taking metformin throughout the study. One participant on metformin was also taking semaglutide injection. At the start of the study, mean HbA1c was 7.1% (SD 0.4). There were two dropouts in the first cohort and one in the second cohort: (Two from GMV classes interfering with work and one from an accident unrelated to the intervention). These three participants did not attend more than half of the visits and therefore were excluded from the analysis.
Table 1: Descriptive characteristics of participants (n = 13). View Table 1
Table 2 depicts the averages of the variables at Time 1 and Time 2. At the end of the 14 weeks for each cohort, participants had a significant reduction in HbA1c (-0.4%; p < 0.05), weight (-6.4 lbs.; p < 0.001), and average glucose as calculated by the CGM, (-21.1 mg/dl; p < 0.005). The percentage of participants that lowered their HbA1c into the prediabetes range was 38.5% (5/13). There were no hypoglycemic events for any participant throughout the study. No significant differences were found in any of the biomarkers in lipid profiles, or blood pressure. There were no significant differences between Time 1 and Time 2 on the PAM-13 and the SF-12. Figure 1 illustrates the change in average glucose between Time 1 and Time 2.
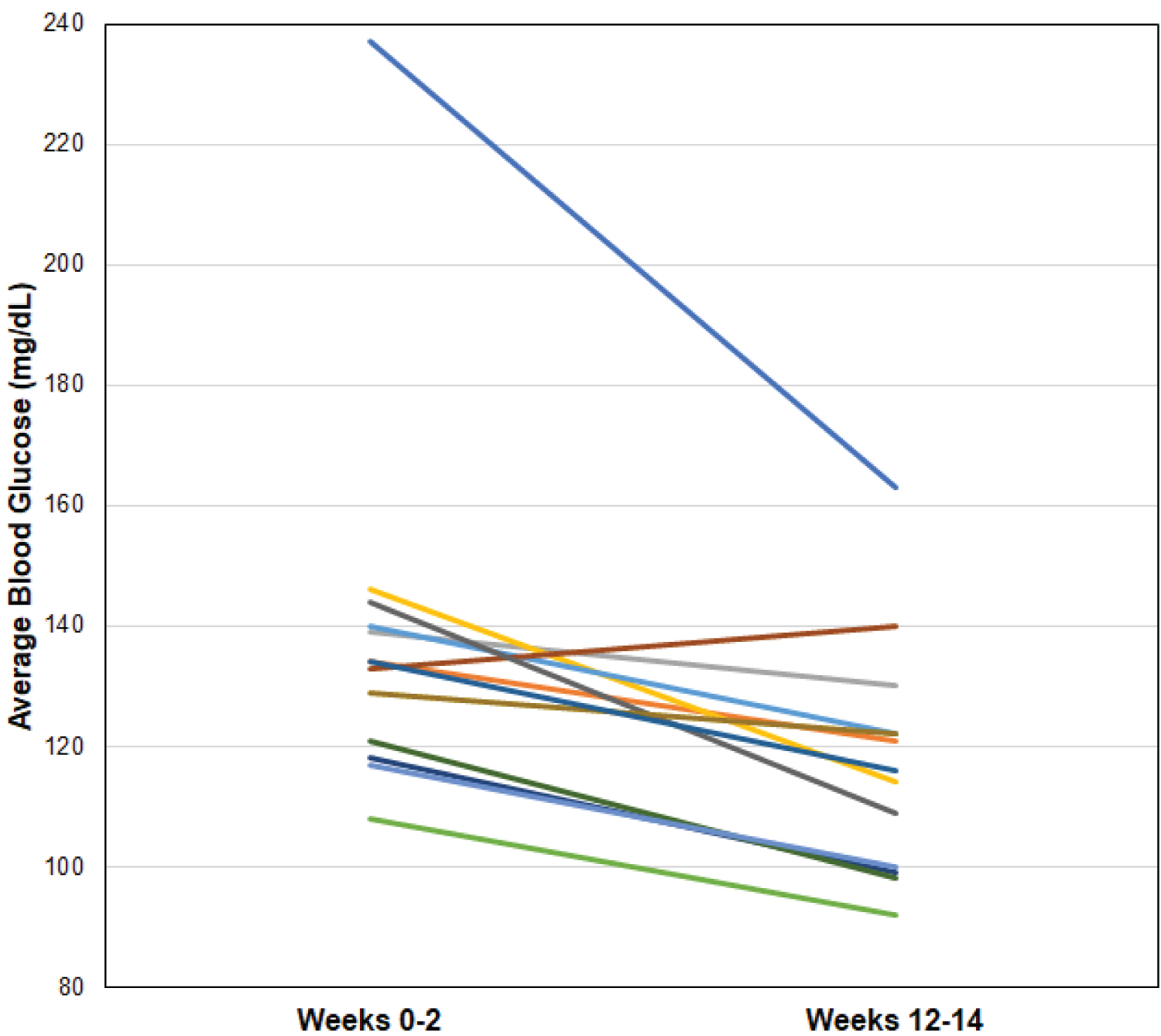 Figure 1: Illustrates the change in average glucose between Time 1 and Time 2.
View Figure 1
Figure 1: Illustrates the change in average glucose between Time 1 and Time 2.
View Figure 1
Table 2: Comparison of patient outcome variables. View Table 2
Participants' responses to the study questionnaire about wearing a CGM indicated that the CGM empowered them to make lifestyle changes to improve their blood sugar and would continue to wear a CGM, if cost was affordable.
In a model combining GMV with CGM, we found significant improvement in metabolic outcomes. Participants statistically significantly lowered their HbA1c by 0.4%, with 38.5% of participants decreasing this value into the prediabetes range. Moreover, there was a significant 21 mg/dl drop in average glucose between Time 1 (the first two weeks of the study) and Time 2 (the last two weeks). The adjusted average change in average glucose still dropped by 16.7 mg/dl between Time 1 and Time 2. An average statistically significant 6.4-pound weight loss occurred among participants over the 14 weeks study period. There were no significant changes in any lipid profile variables, systolic or diastolic blood pressure or in the PAM-13 and SF-12.
Traditional data fail to provide useful guidance on the impact of diet and exercise adjustments. Methods such as CGMs provide a tight cause-and-effect association between lifestyle choices and glucose fluctuations. In turn, this allows patients to dynamically change their eating habits and other behaviors as they are provided near immediate feedback. Glucose variability is an emerging marker of glucose control that is independently correlated to coronary plaque instability and cardiovascular disease [16-19]. Glucose variability from the Freestyle Libre of less than 36% represents low glucose variability [20]. In our cohort, this measure was low at baseline, yet still improved from 22.4% to 20.3%. Participants reduced their weight by an average of 6.4 pounds over the 14-week study period.
While the changes in total, non-HDL and LDL cholesterol were in a favorable direction, average HDL declined and triglycerides increased, non-significantly. It is unclear why the average HDL value decreased, but triglycerides were likely strongly affected by two participants at Time 2 with values over 400 mg/dL. Upon further investigation, the median value for triglycerides declined between the two time periods (147 to 122 mg/dL) which is likely a better picture of group lifestyle changes.
Our hypothesis was that the combination of CGM in a GMV setting would improve patient activation, quality of life, and metabolic outcomes through modified lifestyle behaviors. While there was no significant difference for the SF-12 and PAM-13, there were significant improvements for many subscales of the SF-12 (data not shown). From the brief questionnaire administered at baseline and post-study, every participant reported positive comments regarding using the CGM to make lifestyle changes. Common positive themes among our participants consisted of allowing participants to understand how meals, stress, and physical activity affected their body, and empowering them to make changes to improve their blood sugar. Most reported that their improved understanding changed what they consumed. No participants reported negative effects of using the CGM. Strengths of this study are that it included a diverse population, the intervention was readily applied in a resident-run community based primary care setting, and the mix of patients in the study is representative of our county's and our practice's Type 2 diabetes population.
The limitations of this study included the small sample size, which limited our ability to detect differences which might have truly existed (e.g., a Type II Error) in the SF-12, PAM-13 and other variables. Additionally, there was no control arm to the study. Furthermore, this was a single center study and assignment to wearing CGM was not random. The overall generalizability of the findings would be improved with a larger sample gathered from multiple centers across the country with a randomized clinical trial design. Future research should carry out such a study given the favorable positive results we obtained. Future research should also be dedicated to comparing the use of CGMs in a variety of settings (e.g., long term care facilities, specialty clinics) to improve patient activation, quality of life, and metabolic outcomes.
Using CGMs in a GMV primary care setting impacted patient's dietary decisions, enabling participants to improve their HbA1c and lose weight, without the addition of medications. This was also reflective of patients improved monitoring of glucose by using a CGM versus finger sticks through high adherence with CGM, reduced glucose variability, and other favorable glycemic markers. The approach of incorporating CGM use into GMVs warrants further investigation with larger cohorts to evaluate not only improved biomarkers, but also patient activation, and quality of life. Additionally, studies of longer duration should be conducted to evaluate the sustainability of improved HbA1c, weight loss, and lipid profiles following a GMV using CGM.
We would like to thank Abbott Diabetes Care Inc., for providing continuous glucose monitoring supplies for the study.
Abbott Diabetes Care Inc., was not part of designing the study, conducting the study, data collection, or data analysis. The authors have no conflicts to report.