This study presents the epidemiological data and clinico‐pathological features of oral cancer in patients treated in Prince Sultan Military Medical City‐ Oncology Department over the last 7 years.
We reviewed all available data in our electronic and paper files for patients who had a confirmed pathological diagnosis of oral cavity malignancies between 2009‐2015.
A total of 87 patients with pathologically confirmed oral cancer. The majority of patients were males (M:F = 1.63:1). Median age at diagnosis was 62 years (24-95). History related to risk factors was completed in only 59 patients (68%). Forty-five patients (76%) had a positive history for tobacco usage either smoking and/or chewing (shamma). The use of shamma, (a mixture of powdered tobacco, lime, ash, black pepper, oils and flavorings), was confirmed in 31/59 patients (either alone or associated with smoking). Oral Tongue as a primary site represents about half of the patients 44 (50.6%), followed by alveolar ridge (16.1%) and floor of mouth (11.5%), buccal mucosa (10.3%) and other sites (11.5%). Two thirds of patients (66.6%) presented with advanced stages (III and IV). The 5-year overall survival rates were 77.1% for the early stage disease group and 45.9% for the advanced stage disease group (P = 0.028). The 5-year progression-free survival rates were 65.6% for the early stage disease group compared with 28.1% for the advanced stage disease group (P = 0.02).
Most of our cases had a positive history of tobacco usage. This together with the high percentage of advanced diseases that carried a significantly poor outcome warrant the need for effective anti‐tobacco campaigns, public awareness and primary care physicians education for early detection of disease.
Epidemiology, Oral cancer, Clinicopathological features
Oral cancer is the most common head and neck cancer (HNC). Its global incidence in 2015 was 410,000 cases with 146,000 deaths [1]. In USA, 41,380 patients were diagnosed with HNC in 2013, and oral cavity cancers accounted for almost 30,000 of new cases [2]. In Saudi Arabia, a total number of 211 oral cancer cases were reported in 2014, representing about 1.8% of the total cancer cases [3].
More than 90% of all oral cancers are squamous cell carcinoma (SCC) [4]. While around 10% consists of sarcomas, lymphomas, salivary gland tumors, melanomas, verrucous carcinomas, adenocarcinomas, adenoid cystic carcinomas, and mucoepidermoid carcinoma [2].
Unfortunately, the overall survival of oral cancer patients has not improved significantly during the past decade, with 5-year survival rates between 45-50% [5]. Early diagnosis is the most important factor for improving patient survival, rates as high as 80-90% on these first stages can be achieved. Early diagnosis also minimizes the extent of surgery required [4].
Oral cancers have different incidence rates in different areas of the world and different clinical and epidemiological features, which may be attributed to the difference in prevalence and patterns of exposure to risk factors. Tobacco smoking and chewing, alcohol consumption, and HPV infection are the major risk factors for oropharyngeal and oral cavity cancers, with tobacco and alcohol consumption having synergistic effects [6].
Several epidemiological studies on oral cancer were published in different countries. However, survival outcome of oral cancer in Saudi Arabia was addressed by few studies. The present study aimed to report the clinic-pathological features of our oral cavity cancer patients.
We reviewed all available data in our electronic and paper files for the patients who have a pathologically confirmed diagnosis of oral cavity malignancies between 2009-2015. Tumor stage was determined based on the 2010 American Joint Committee on Cancer (AJCC) TNM classification, 7th edition [7].
All patients were subjected to standard treatment protocols according to primary tumor site, stage and patient's general conditions. All cases were discussed in multidisciplinary ENT/Oncology combined clinic. After receiving primary treatment, patients are followed in the combined oncology and head and neck surgery clinic. Follow up visits are every 2 months for the 1st year, every 3 months in the 2nd year and every 4 months in the 3rd year then every 6 months till completing 5 years, then annually. At each visit, patients are subjected to clinical examination. Radiological evaluations of response are usually performed in the first post-treatment visit by computed tomography (CT), Positron Emission Tomography (PET-CT) or Magnetic Resonance Imaging (MRI). Imaging studies are usually repeated if there is clinical suspicion for distant metastases and for restaging in the case of loco-regional recurrence. Local or regional recurrence must be confirmed pathologically.
Survival times were calculated from the day of diagnosis. Overall survival (OS) and progression free survival (PFS) were calculated at the date of death or relapse or the last date of follow up. Kaplan-Meier method was used for estimation of survival rates. The level of significance was set at p ≤ 0.05.
A total of 87 patients had pathologically confirmed oral cancer. The majority of patients were males (M:F = 1.63:1). Median age at diagnosis was 62 years (24‐95). Tongue cancer was the most common primary tumor site, 44 cases (50.6%). Two thirds of patients presented with advanced stage; III and IV (66.6%). The majority of cases were of squamous histology (91%) (Table 1).
Table 1: Patients Characteristics. View Table 1
History related to risk factors was completed in only 59 patients (68%). Forty-five patients (76%) had a positive history for tobacco usage either smoking and/or chewing (shamma). The use of shamma was confirmed in 31/59 patients (either alone or associated with smoking). History of Shamma usage was relatively more common in females (63%) vs. 49% in males. However, it was not statistically significant (p = 0.4).
Forty-eight patients (55.2%) had surgery either as a primary modality or after neoadjuvant treatment. Neoadjuvant chemotherapy (NACT) used for 25 patients (28.7%) and concurrent chemo-radiotherapy (CCRT) used for 27 patients (31%). Radical and postoperative radiotherapy (PORT) were used in 23 (26.4%) and 32 (36.8%) patients, respectively.
Median follow up time was 15.63 months (1.33-78.8). At the time of analysis, death was confirmed in 22 patients (25.3%) and 41 patients had confirmed disease progression (47.2%). The estimated OS and PFS at 5 years were 57.6% and 40%, respectively (Figure 1 and Figure 2)
 Figure 1: Overall Survival of 87 patients with oral cancer. View Figure 1
Figure 1: Overall Survival of 87 patients with oral cancer. View Figure 1
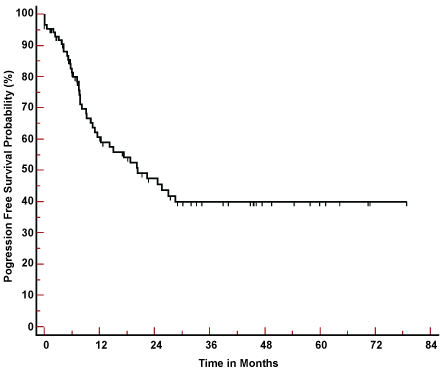 Figure 2: Progression free Survival of 87 patients with oral cancer. View Figure 2
Figure 2: Progression free Survival of 87 patients with oral cancer. View Figure 2
Table 2 shows that early stage at presentation together with the good performance status was associated with better outcomes in term of both OS and PFS at 5 years.
Table 2: Survival outcome according to different prognostic factors. View Table 2
Despite the improvement in cancer management in the last decades, oral cancer still considered as a major health problem, associated with high incidence of morbidity and disfigurement. The advanced stage at presentation, in most cases, and the anatomical consideration further complicate the problem.
In this study, tongue cancer represents more than half of the cases (51%), which is consistent with most of the studies from western world [8-10]. Also, another study from Saudi Arabia showed 45% of oral cancer cases were of primary tongue origin [11]. In a recent review analysis of oral cancer in different Arab countries, tongue was the most common primary tumor site [12]. This is not the case among Asian populations. In a study from Pakistan, buccal cancer was found to be more common than tongue cancer (32% vs. 21.8%, respectively) [13]. Also, in an Indian study tongue represented the primary tumor site in only 17% of the cases while buccal mucosa represented 35.5% [14]. In addition, Researchers in Taiwan found that tongue cancer is the second most common oral cancer after buccal cancer [15]. This may be due to the different habits and exposure related to risk factors. Asian countries have varying patterns of use of tobacco and alcohol with the common use of betel quid/tobacco chewing [16].
A significant number of our patients are residents of the southern region where tobacco chewing (shamma) is a common habit. Shamma is a preparation of smokeless tobacco, being a mixture of powdered tobacco, carbonate of lime, ash, black pepper, oils and flavoring [17]. Shamma usage has been linked to the high frequency of oral cancer in the southwestern region of Saudi Arabia [18].
There is a clear defect in the history of special habits in our cases as it was deficient in about one third of patients. The percentage of shamma users was high (31/59) among patients with completed data; however, the real percentage of shamma users may be underestimated, as shamma is still illegal in Saudi Arabia. Similarly, alcohol is strictly prohibited, and this may explain why only two patients disclosed history of alcohol consumption.
In our cohort, M:F ratio (1.63:1) showed a higher proportion of female patients compared to the global statistics. The global M:F ratio is around 2:1 with some regional differences [19]. Some studies showed a much higher prevalence in male patients [14,20]. Additionally, in our study there is a significant minority of younger male patients. As mentioned above, a significant number of our patients were referred from the southern region of Saudi Arabia where shamma usage is common amongst both male and female individuals. Females, probably, start using shamma later in their lives. Smoking at young age may be the risk factor in the younger male population.
Two-thirds of our cases were presented in advanced stages. This is concordant with the findings in other studies in different geographical areas [12,13,20-22]. However, some studies, especially from the western world, showed a higher percentage of early diseases [10,23,24]. The burden of oral cancer is considered heavier in some developing countries, due to difficulties in primary prevention and application of regulatory policies, to access screening by a dental or medical professional and to have access to treatment when the disease has already progressed [25].
The 5 years survival outcomes in our patients are comparable to the global rates [16]. The stage at presentation was one of the most important factors for outcome at 5 years. Patients with early stage at presentation had better OS (77.1% vs. 45.9%) and PFS (65.6% vs. 28.1%) compared to advanced stage group.
Most of our cases are presented in advanced stages, which reflected on the poor outcome. This warrants the need for increasing public awareness about the disease and for training of the primary health care professionals for early detection of premalignant conditions. Early referral to higher specialized centers ensues a higher opportunity for better management. The positive history of tobacco usage necessitates the need for effective anti-tobacco campaigns to enhance the public knowledge about tobacco induced deleterious effects.