The aim of our study was to evaluate the efficacy of n-Heptane (C7H16) as a xylene substitute for clearing, deparaffinizing and staining procedures in routine histopathology. 30 biopsied oral tissues were divided in two halves, each half was processed by xylene and n-Heptane respectively.
To substitute toxic aromatic solvents with nonhazardous aliphatic derivatives.
The second part of the study was conducted with the objective to reduce the concentration of comparatively expensive n-Heptane.
A sample size of 20 was considered for immunohistochemistry (IHC), with Cytokeratin and Vimentin as epithelial and connective tissue markers respectively.
Kappa statistical analysis established agreement between observers in all criteria's with significant p value < 0.05. The p value (> 0.05) was not statistically significant with Mann-Whitney U Test, for all criteria's indicating comparable results between n-Heptane and xylene. The clarity, uniformity, crispness, section quality as well as cellular parameters of nuclear & cytoplasmic staining, nuclear cytoplasmic contrast, cell morphology were found comparable between n-Heptane and xylene.
n-Heptane is not as hazardous and toxic as aromatic or chlorinated solvents. Thus it can be considered as a convenient substitute to xylene.
n-Heptane, Xylene, Deparaffinization, Clearing, Immunohistochemistry
Xylene is an aromatic hydrocarbon with chemical formula of C6H4(CH3)2 . It is widely used in tissue processing, staining and coverslipping in the laboratory [1,2]. Occupational Safety and Health Administration (OSHA) regulation identifies xylene as a hazardous substance [1]. Xylene has also been documented under International Agency for Research on Cancer (IARC) [3]. The National institute for occupational safety and health (NIOSH) has recommended exposure limits for xylene at 100 ppm (parts per million) as a time weighted average (TWA) for up to a 10 hours work shift and a 40 hour work week and 200 ppm for 10 minutes as a short term limit [2]. In case of Heptane, it should not exceed 85 ppm as a full-shift TWA or 440 ppm in a 15-minute ceiling limit [4]. Agency for Toxic Substances and Disease Registry (ATSDR/EPA) has published a list of 275 chemicals with known or suspected toxicity, amongst which xylene has been ranked 48 [5]. In an effort to reduce health hazards and to create a safer histopathology working environment, it is required to introduce safer and convenient substitutes to xylene. n-Heptane is an aliphatic hydrocarbon, It is inert and non-fluorescent solvent. It does not have an unpleasant smell nor does it irritate the mucous membranes. It is considered a less harmful, relatively inexpensive solvent and may be a convenient substitute to xylene [5]. This study therefore is designed to evaluate the efficacy of n-Heptane to substitute xylene as an alternative in clearing, deparaffinizing and staining procedure in routine histopathology.
The first part of the study consisted of fixation of 30 biopsy human oral tissues from various sites, by immersion in 10% formaldehyde at room temperature for 24 hours. Each tissue was cut into two halves, with one section processed using xylene (Fisher Scientific, Thermo fisher Scientific) and the other using n-Heptane (Rankem, Avantor Performance Materials India). Specimens were washed in tap water for 24 hours and dehydrated in 30, 50, 70, 96 and 100% ethanol for 1 hour at each concentration. Clearing for the first 30 samples was accomplished using xylene and embedded as usual in paraffin wax (Rankem).The remaining 30 tissue samples were cleared with two changes of n-Heptane (99% purity) for 10 minutes. The tissues cleared in n-Heptane were placed in a mixture of 1:1 v/v (volume/volume) n-Heptane: paraffin oil at 56 ℃ for 1 hour, then embedded in paraffin wax at 56 ℃ for 24 hours [5]. Sections (Rotory microtome , Thermo Fisher Scientific, HM340E) obtained were deparaffinized in 1:1 v/v n-Heptane: paraffin oil for 15 minutes and hydrated in descending grades of alcohols (100, 96 and 70% ethanol, 10 min at each concentration) to water. Xylene was replaced with 1:1 v/v n-Heptane: paraffin oil in routine hematoxylin and eosin staining procedure for sections cleared in n-Heptane. Microscopic observations and photography of sections mounted in DPX (Dysterene Plasticizer Xylene, Fisher Scientific) was carried out.
The second part of the study consisted of reducing the concentration of n-Heptane during tissue processing. 80 tissues were cleared by 2 changes of n-Heptane (99% purity) for 10 minutes, 20 tissues each were immersed in 4 different concentrations of Heptane: paraffin oil, 1:4 v/v, 2:3 v/v, 3:2 v/v and 4:1 v/v for an hour and embedded in paraffin wax at 56 ℃ for 24 hours. Thereafter, the sections were subsequently stained with hematoxylin and eosin.
Another sample size of 20 was considered for immunohistochemistry. 10 each were stained for Cytokeratin and vimentin for tissues processed in xylene and n-Heptane (99% purity). Immunohistochemical analysis was performed on 3 µ paraffin embedded tissue sections on gelatine coated glass slides. After heat drying, sections were deparaffinized in xylene and subsequently rehydrated in gradients of ethanol. Antigen retrieval to unmask antigenic sites was done using 10 mM citric acid solution (pH 6) followed by a washing step with Tris buffered saline (TBS pH 7.6). Further incubations with pre-diluted ready to use primary mouse monoclonal anti-cytokeratin antibody (AE1/AE3, Biogenex, San Ramon), monoclonal mouse anti-vimentin antibody (V9) (Biogenex, San Ramon), was performed at 37 ℃ for 30 minutes. Sections were washed again and incubated with biotinylated secondary antibody (Biogenex, San Ramon) for 30 minutes followed by the streptavidin-biotin-peroxidase (Biogenex, San Ramon) for 30 minutes at room temperature. Colored reactions were developed by incubating with 3'3'-diaminobenzidine (SIGMA, 0.05 diaminobenzidine in 0.05 M Tris buffer, pH 7.6, and 0.01 H2O2) and subsequently counterstained with Harris hematoxylin and mounted with DPX. Positive and negative controls were included in all reactions. Presence of brown colored reaction localized to the nucleus or cytoplasm was considered as positive reaction.
The microscopic slides obtained after processing with n-heptane and Xylene were compared by pathologists, for differences in various staining parameters including clarity (clearness of appearance), uniformity (consistency), crispness (marked by conciseness & briskness),nuclear staining, cytoplasmic staining, nuclear cytoplasmic contrast, cell morphology, section quality (thickness, retraction) and IHC staining.
3 blinded observers scored H& E stained 30 slides each for n-heptane and xylene for above mentioned 9 parameters as Good:3, Average:2 and Poor:1.
IHC slides were scored as No staining, < 10% area stained: 0, Faintly stained, > 10% area stained: 1+, Weak to moderately stained, > 10% area stained: 2+, Strongly stained, > 10% area stained: 3+.
Kappa statistical analysis established agreement between three blinded observers in all above mentioned 9 parameters with significant p value < 0.05.
Mann-Whitney U Test displayed p value (> 0.05) , that was statistically not significant, for all criteria's indicating comparable clarity, uniformity, cell morphology, section quality, crispness, nuclear cytoplasmic contrast, nuclear staining, cytoplasmic staining and IHC staining between n-Heptane and xylene (Figure 1, Figure 2, Figure 3, and Figure 4).
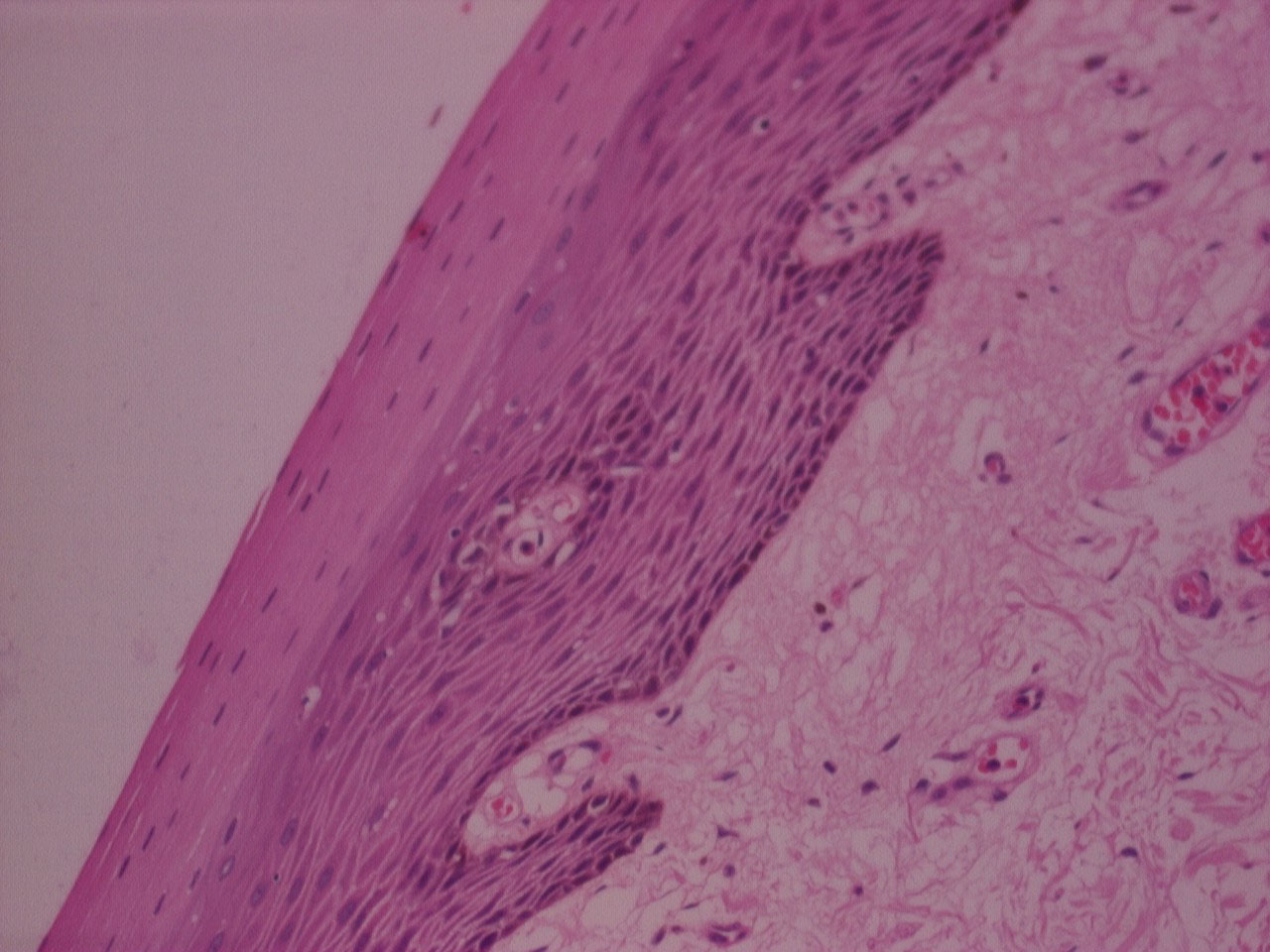 Figure 1: H&E stained photomicrograph of section processed with n-Heptane, 10x. View Figure 1
Figure 1: H&E stained photomicrograph of section processed with n-Heptane, 10x. View Figure 1
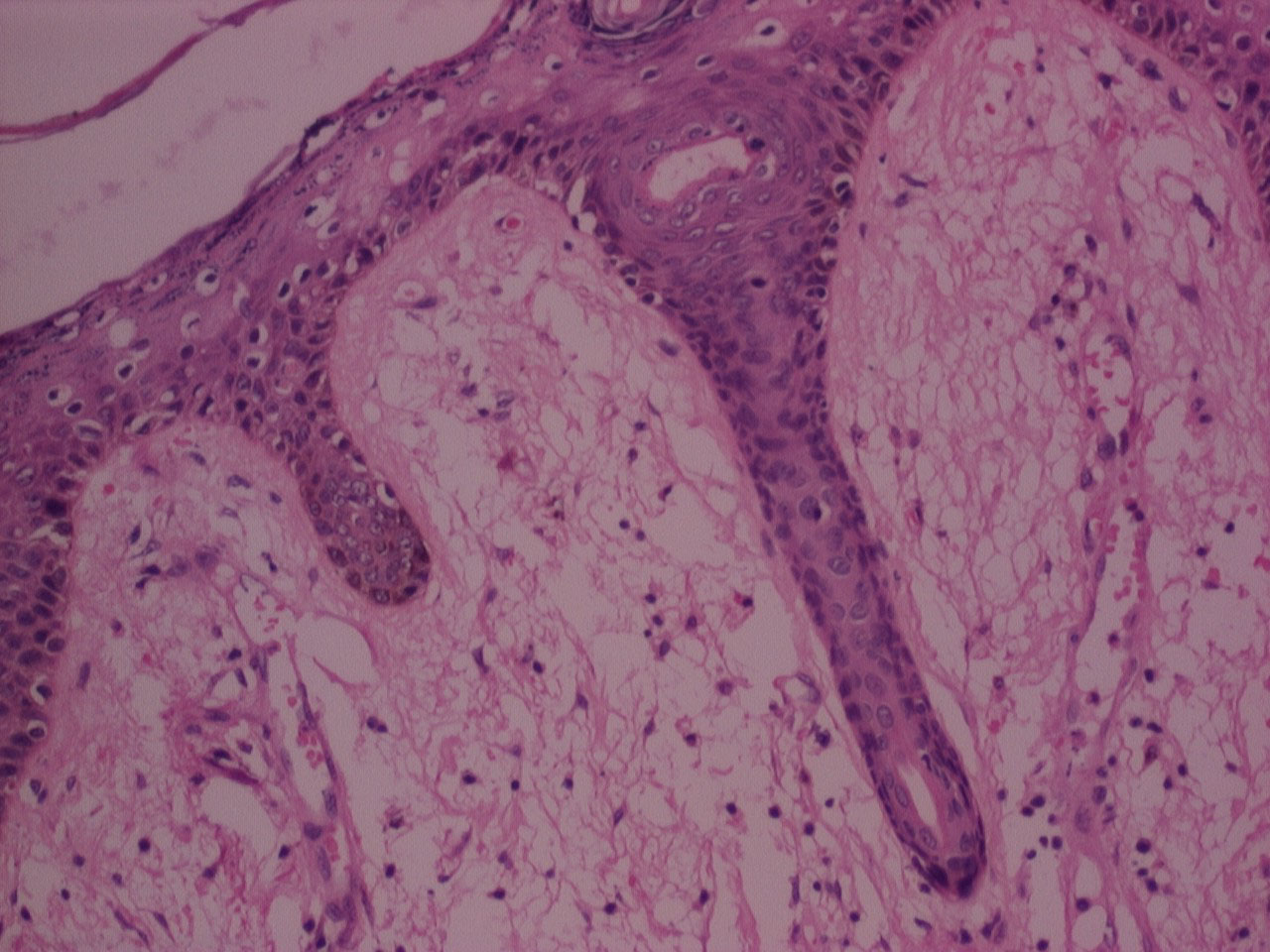 Figure 2: H&E stained photomicrograph of section processed with Xylene, 10x. View Figure 2
Figure 2: H&E stained photomicrograph of section processed with Xylene, 10x. View Figure 2
 Figure 3: Clarity, uniformity, cell morphology and section quality of tissues processed with n-Heptane and xylene. View Figure 3
Figure 3: Clarity, uniformity, cell morphology and section quality of tissues processed with n-Heptane and xylene. View Figure 3
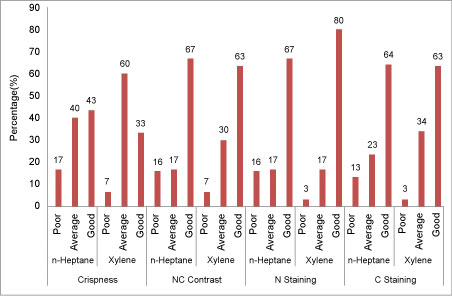 Figure 4: Crispness, nuclear cytoplasmic (NC) contrast, nuclear (N) staining and cytoplasmic (C) staining for tissues processed with n-Heptane and xylene. View Figure 4
Figure 4: Crispness, nuclear cytoplasmic (NC) contrast, nuclear (N) staining and cytoplasmic (C) staining for tissues processed with n-Heptane and xylene. View Figure 4
Better results were obtained with n-Heptane as regards crispness (43%) and nuclear cytoplasmic contrast (67%) as compared to xylene where it was 33% and 63% respectively (Figure 5).
 Figure 5: IHC stained photomicrograph of section showing cytokeratin positivity, processed with n-Heptane, 10x. View Figure 5
Figure 5: IHC stained photomicrograph of section showing cytokeratin positivity, processed with n-Heptane, 10x. View Figure 5
A preparation of 2:3 v/v of n-Heptane: paraffin oil is adequate for sectioning and staining of tissue. Increasing the concentration of n-Heptane yielded marginally better results.
The majority of the samples processed with n-Heptane showed weak to moderate IHC staining for cytokeratin and vimentin (Figure 5, Figure 6, Figure 7, Figure 8 and Figure 9) with none of the sections devoid of staining.
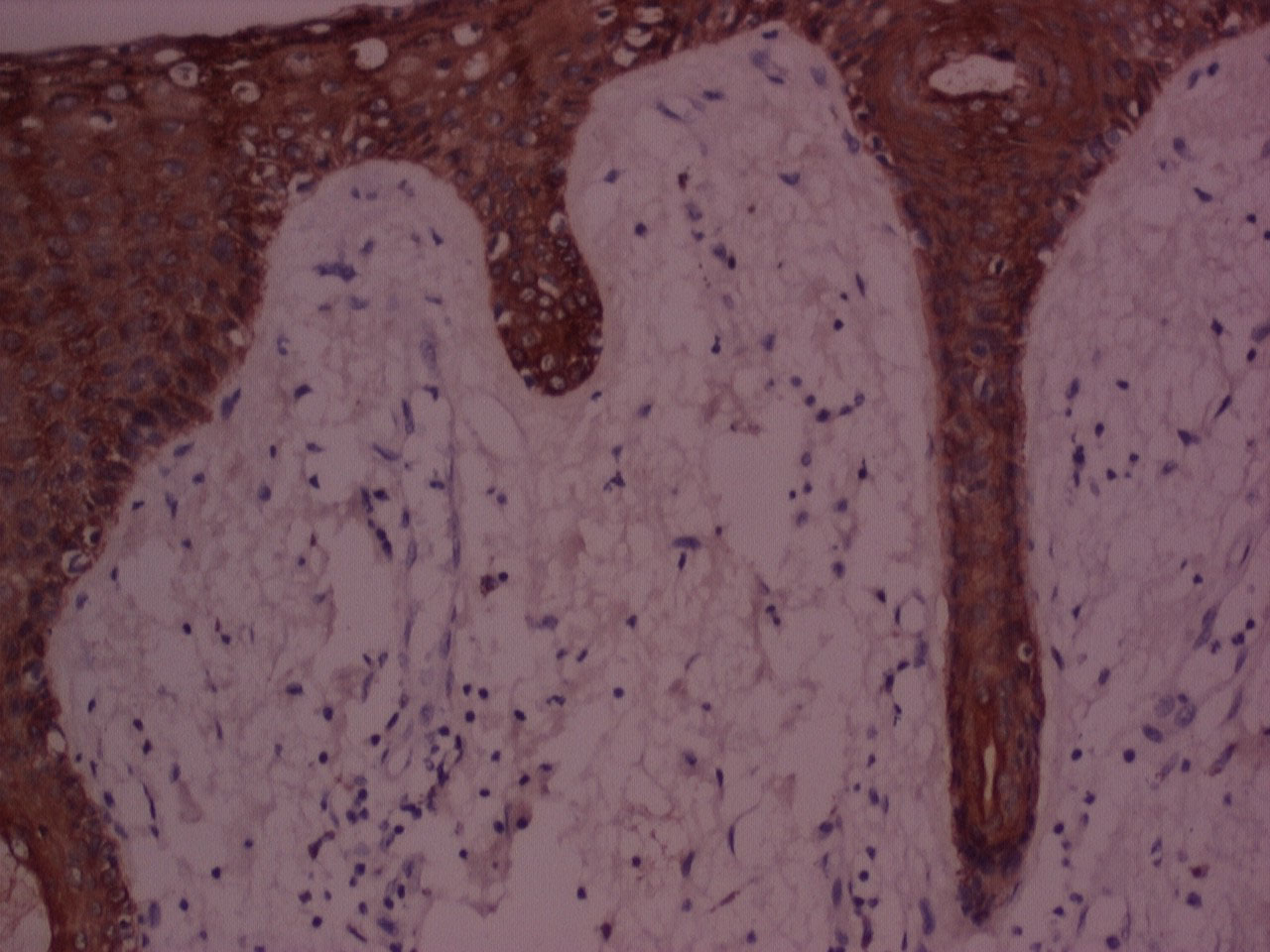 Figure 6: IHC stained photomicrograph of section showing cytokeratin positivity, processed with Xylene, 10x. View Figure 6
Figure 6: IHC stained photomicrograph of section showing cytokeratin positivity, processed with Xylene, 10x. View Figure 6
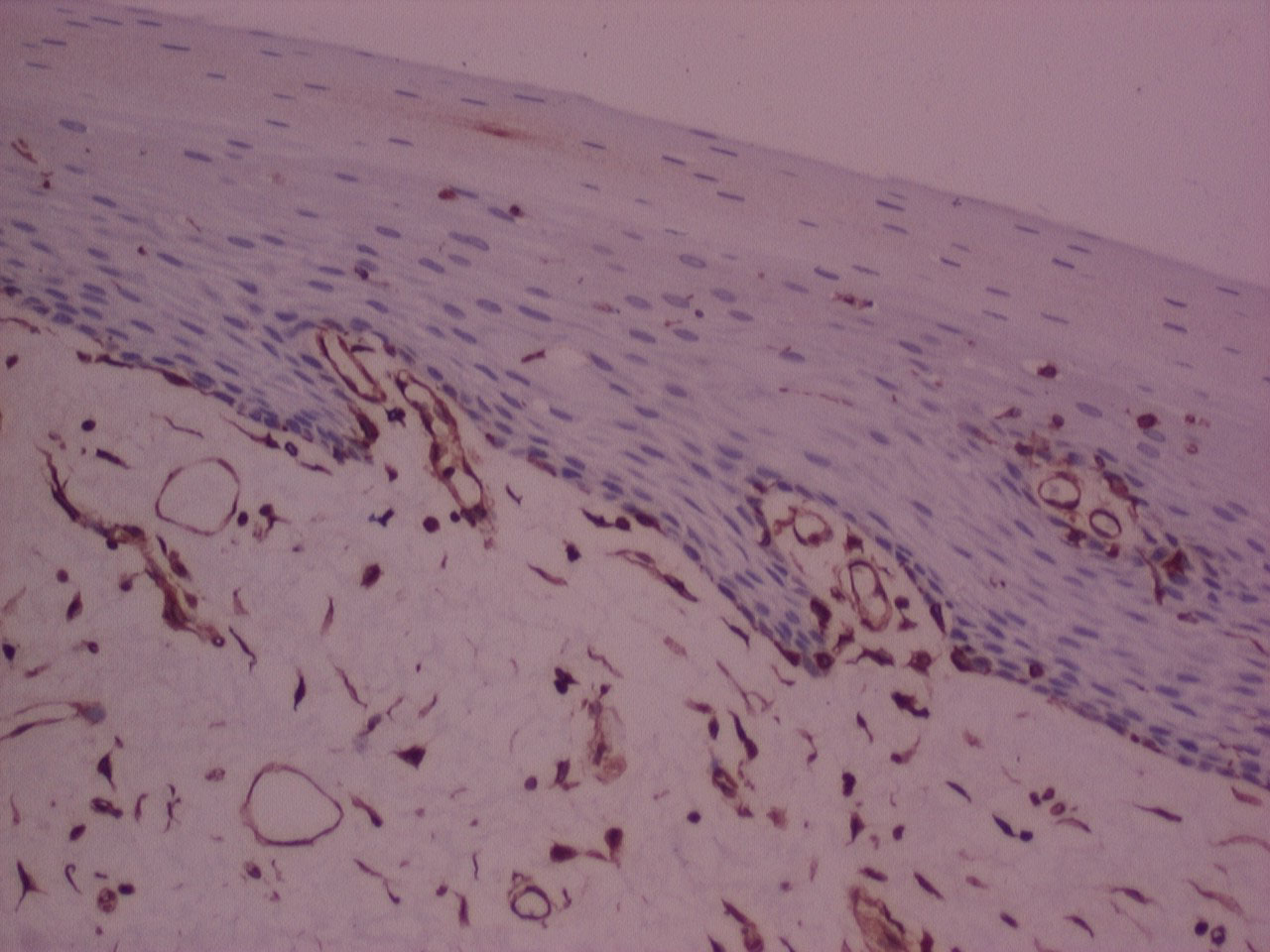 Figure 7: IHC stained photomicrograph of section showing vimentin positivity, processed with n-Heptane, 10x. View Figure 7
Figure 7: IHC stained photomicrograph of section showing vimentin positivity, processed with n-Heptane, 10x. View Figure 7
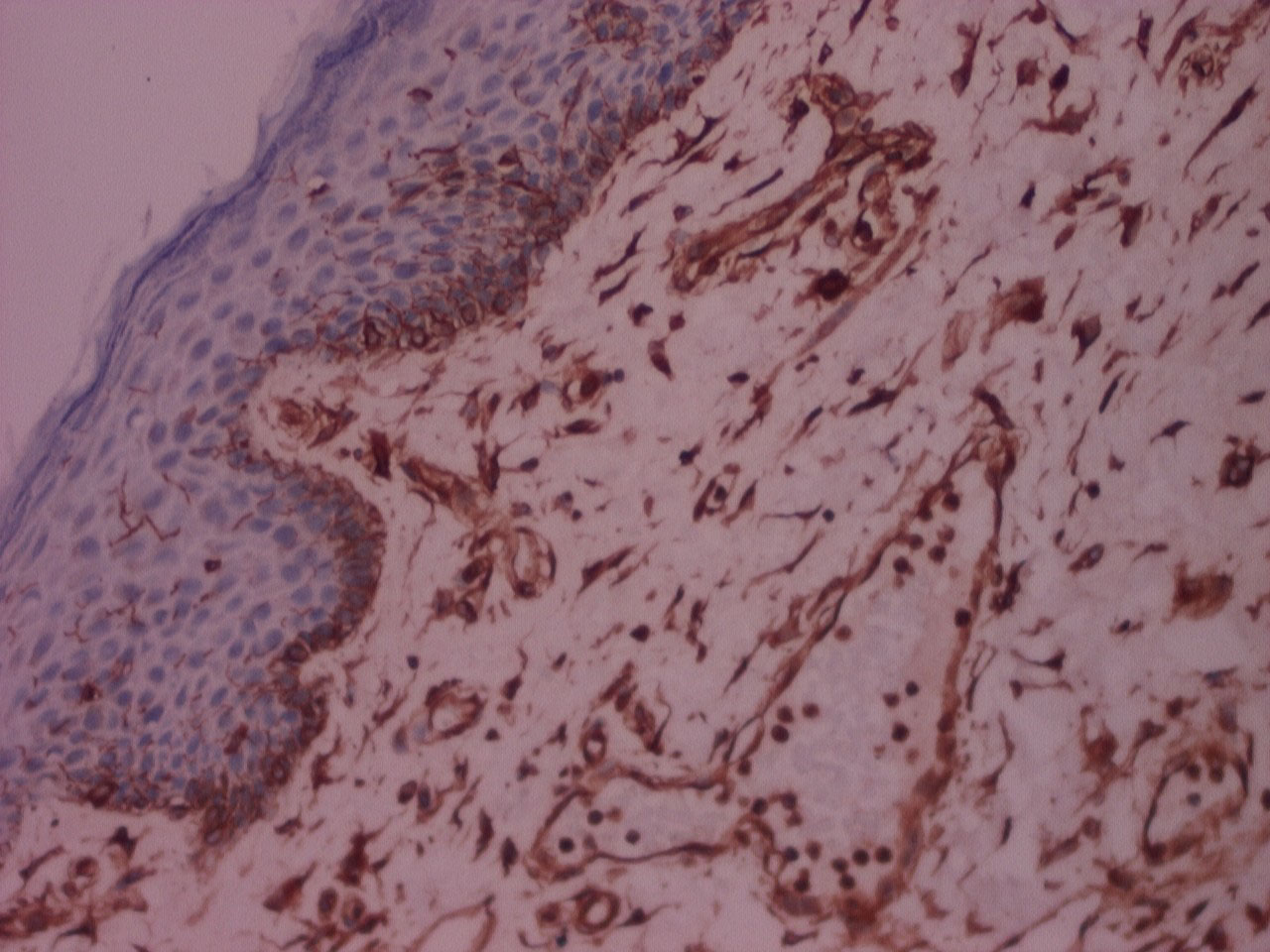 Figure 8: IHC stained photomicrograph of section showing vimentin positivity, processed with Xylene, 10x. View Figure 8
Figure 8: IHC stained photomicrograph of section showing vimentin positivity, processed with Xylene, 10x. View Figure 8
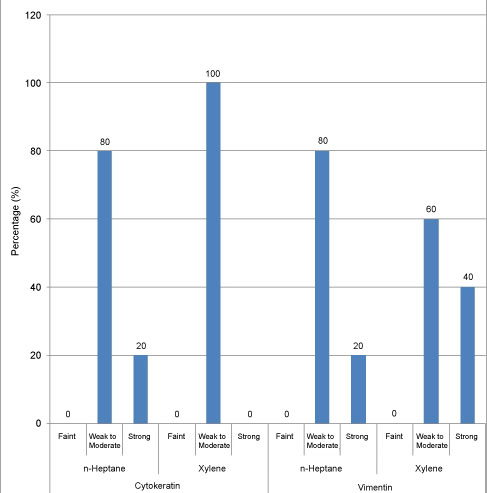 Figure 9: IHC staining with cytokeratin and vimentin staining for tissues processed with n-Heptane and xylene. View Figure 9
Figure 9: IHC staining with cytokeratin and vimentin staining for tissues processed with n-Heptane and xylene. View Figure 9
Xylene traditionally has been employed as a clearing, dewaxing and mounting agent for histology. It is well documented that xylene is an environmental hazard and highly toxic to humans. Accumulating evidences indicate that being repeatedly or excessively exposed to xylene can do harm to the nervous system, skin, liver, kidney, and lung tissues [6-9]. n-Heptane does not have an unpleasant smell nor does it irritate the mucosa. n-Heptane scores over other aromatic and chlorinated compounds in being far less toxic [5].
In a study conducted by Stockert J Cin 2012, paraffin sections of rat tissues processed with n-heptane and stained with hematoxylineosin or Masson's trichrome showed proper embedment, well preserved morphology and excellent staining [5].
Kunhua W, et al. in 2012 assessed, nontoxic xylene substitute (SBO), a mixture of 86% of white oil No.2 and 14% of n-heptane, treated sections stained with hematoxylin-eosin. The sections revealed a good maintenance of cell morphology and structure, and a clear definition of the cytoplasm and the nucleus [9]. The staining results obtained using SBO in IHC staining were comparable or superior to those achieved by using xylene [5].
In other studies in paraffin blocks, on xylene alternatives, Ankle MR, et al. in 2011 and Ramulu, et al. in 2012 also indicated adequate nuclear and cytoplasmic staining, and no statistical difference in clarity using diluted dish washing solution and liquid dish washing soap for xylene and alcohol, in H&E staining respectively [10,11]. The uniformity and crispness in XMF sections in Ankle's study showed down gradation and up gradation respectively [11]. Ramulu found no statistical difference in crispness when substituting liquid dish washing soap for xylene and alcohol in H&E staining [10].
We found no statistical differences in the clarity, uniformity, crispness, section quality between n-Heptane and xylene. Cellular parameters of nuclear & cytoplasmic staining, nuclear cytoplasmic contrast and cell morphology too were comparable between n-Heptane and xylene. The paraffin blocks prepared by using n-Heptane as a clearing agent were easily cut into 4 μm serial sections and ribbon formation was good. No sign of cell shrinkage and deformation in n-Heptane treated sections was observed. In our study 20% of samples processed with n-Heptane showed strong IHC staining for cytokeratin as compared to xylene where 80% of the samples showed weak to moderate IHC staining.
In the second part of our study which consisted of reducing the concentration of n-Heptane during tissue processing. The preparation of 1:4 v/v n-Heptane:paraffin oil was found to be inadequate as the tissues were too soft after processing and sections could not be obtained. Therefore, concentration of n-Heptane:paraffin oil was altered to 2:3 v/v, with minimum 2 parts of n-Heptane.
Xylene, the most common aromatic solvent and chlorinated compound used for paraffin processing in histology and histopathology laboratories is highly toxic. n-Heptane is a safer and less toxic substitute as compared to other aromatic or chlorinated solvents. The working environment for the operator in the laboratory too is amenable with n-Heptane having neither an unpleasant odour nor being an irritant to the mucous membranes.
This study was an attempt to decrease the quantity of n-Heptane utilized as it is a comparatively expensive solvent. We found that n-Heptane:paraffin oil in concentration 2:3 v/v, for clearing, deparaffinizing & staining procedures is adequate for sectioning and staining of tissue.
Considering the comparable results obtained with both n-Heptane and xylene, n-Heptane appears to be a less hazardous alternative in clearing, deparaffinizing and staining in routine histological procedures.