Background: Basal-like breast cancer (BLBC) is identified by gene expression profiling studies and is associated with worse overall survival. The identification of possible precursors of BLBC is important to gain insight into tumorigenesis and therapeutic targets. This retrospective study evaluated the characteristics of possible precursors of BLBC, including basal-like atypical ductal hyperplasia (bADH) and basal-like ductal carcinoma in situ (bDCIS); basal cytokeratin (CK) expression in bADH and bDCIS; and the frequency of bADH and bDCIS in patients with BRCA1/2 mutations.
Methods: 15 patients diagnosed with triple-negative BLBC and associated bDCIS and 2 patients with bADH were included. additional 3 bADH were identified in prophylactic mastectomies of 105 patients with BRCA1/2 mutation. The immunohistochemical expression of CK5/6, CK14, and CK17 were tested.
Results: All BLBCs were histologic grade 3, with basal-like morphologic features. The associated bDCIS had a trabecular or solid pattern and high nuclear grade. CK5/6 staining was positive in all 15 BLBC and bDCIS cases. bADH shows multiple cell layers, with thick proliferation of rounded hyperchromatic cells, an increased nuclear to cytoplasmic ratio, intermediate-grade nuclei, and nuclear molding. CK5/6 was diffusely strongly positive in all 5 bADH cases.
Conclusions: Our observations provide strong circumstantial evidence for a precursor basal- like ADH lesion. Importantly, there is an atypical intraductal basal-like lesion with diffuse CK5/6 staining; in the context of atypical hyperplastic lesions with basal morphologic characteristics, diffuse staining with CK5/6 should not confirm benignity. A relatively high prevalence of ADH, including bADH, was observed in patients with BRCA1/2 mutations.
Basal-like atypical ductal hyperplasia, Basal-like ductal carcinoma in Situ, Basal- like breast cancer, Cytokeratin CK 5/6, CK 14, CK17, BRCA
BLBC: Basal-Like Breast Cancer; bADH: basal-like Atypical Ductal Hyperplasia; bDCIS: basal-like Ductal Carcinoma In Situ; CK: Basal Cytokeratin; ER: Estrogen Receptor; PR: Progesterone Receptor; Triple-negative: HER2-negative
Gene expression profiling analyses have sub classified breast carcinoma into 4 groups: luminal A, luminal B, HER2-enriched, and basal-like groups [1-4]. Basal-like breast cancer (BLBC) comprises 15% of invasive breast cancer cases and is estrogen receptor (ER)-negative, progesterone receptor (PR)-negative, and HER2-negative (triple-negative) [5-7]. BLBC is associated with a significantly worse overall and disease-free survival than most other subclasses [5-7]. One or more basal cytokeratins (CKs), including CK5/6, CK14, and CK17 and epidermal growth factor receptor, are typically expressed in BLBC [5,8,9].
Little is known about the development of basal-like breast tumors. The identification of possible precursors of BLBCs is important to gain insight into tumor genesis and to develop potential therapeutic targets. Possible precursors of BLBC include basal-like atypical ductal hyperplasia (bADH) and basal-like ductal carcinoma in situ (bDCIS) [8,9]. Few cases of bDCIS associated with BLBC have been reported in the literature [10-12]. However, bADH has not been characterized to date; therefore, the frequency, clinical, pathologic, and immunohistochemical features of bDCIS and bADH are largely unknown.
Additionally, breast tumors secondary to heritable BRCA1 mutation are clinically triple- negative and demonstrate BLBC pathologic features in 80% of cases [13,14]. While BRCA1/2 mutations have been identified in up to 8% of patients with breast cancer, germline BRCA1 mutation has been found in up to 40% of patients with familial breast cancer [15]. A high prevalence of BRCA1 dysfunction was also identified in patients with sporadic BLBC without heritable mutations [16,17]. BRCA1/2 gene regulates diverse cellular functions, including cell cycle checkpoint activation, DNA repair, and tumor suppression [18-20]. However, the frequency of bADH and bDCIS in patients with BRCA1/2 mutations or dysfunction carriers is unknown.
This study evaluated the characteristics of possible precursors of BLBC, including bADH and bDCIS, and the significance of basal CK5/6, CK14, and CK17 expression in bADH and bDCIS. In addition, this study assessed the frequency of bADH and bDCIS in patients with BRCA1/2 mutations.
We retrospectively reviewed our institute database between 2018 and 2020 at The University of Texas MD Anderson Cancer Center and included 105 female patients with BRCA1 or BRCA2 mutation who had undergone prophylactic mastectomy were reviewed. Clinical and pathologic information were collected, including patient age, gender, histologic diagnosis, grade, tumor size, and disease stage and ER, PR, HER2, and Ki67 status. Among the 105 patients, 1 patient had been diagnosed with invasive ductal carcinoma, 1 with DCIS, and 14 with ADH. Through review of the histologic slides from the 14 patients with ADH, 11 were luminal type ADH with secondary lumen formation and 3 were bADH; the 3 bADH cases were included in this study.
In addition, we also included 15 patients who had been diagnosed with BLBC and associated bDCIS, and 2 patients diagnosed with bADH were included in this study. These 17 cases were obtained through a review of 626 sequential breast pathology specimens including 195 biopsies, 271 outside cases and 160 surgical resections obtained during a six-month period in 2021, representing 2.2% of breast pathology cases reviewed at a tertiary care cancer hospital. The 2 bADH cases were identified by MRI-guided biopsy showing non mass enhancement and one patient was upgraded to bDCIS in subsequent mastectomy.
Conventional atypical ductal hyperplasia is of low nuclear grade, which has some, but not all the features of ductal carcinoma in situ and is quantitatively and qualitatively insufficient for the diagnosis of DCIS. The term bADH is proposed for the lesions described herein with limited involvement of the acinus and intermediate grade nuclei that are quantitatively insufficient for the diagnosis of bDCIS.
Whole tissue sections of the 5 bADH and 15 BLBC cases were evaluated in this study.
The immunohistochemical expression of ER, PR, HER2, and CK5/6 was determined in all cases; CK14 was tested in 9 BLBC and 8 bDCIS cases. CK17 was tested in 14 BLBCs and 13 bDCIS, as the area of interest disappeared in deeper sections, and in 4 bADH cases. This study was approved by the institutional review board at University of Texas, MD Anderson Cancer center.
Immunohistochemical staining was performed in a Leica Bond Max autostainer system (Leica Biosystems, GmbH, Nussloch, Germany) following standard automated protocols. The following antibodies were used: ER (clone 6F11, Leica, Buffalo Grove, IL), PR (clone 6, Leica, Buffalo Grove, IL), anti-HER2/neu (clone 4B5, Ventana, Indianapolis, IN), CK5/6 (clone D5/16B4, 1: 50 dilution, Dako, Santa Clara, CA), CK14 (clone LL002, BioGenex, Fremont, CA), and CK17 (clone E3, 1: 20 dilution, Dako, Santa Clara, CA).
The immunoreactivity was reviewed by two pathologists. Cytoplasmic staining of CK5/6 and 17 was counted as positive. Immunoreactivity scores for CK expression were evaluated by staining distribution (focal, < 30% of cells stained; patchy, 30%-70%; and diffuse, 71%-100%) and intensity (weak +, moderate ++, and strong +++). ER and PR results were interpreted as positive (≥ 10% nuclear staining), low positive (1-9% staining), or negative (< 1% staining) according to modified American Society of Clinical Oncology (ASCO)/CAP 2020 guidelines [21,22]. HER2 over- expression and amplification were evaluated according to the ASCO and CAP 2018 guideline [23].
Statistical analyses of the difference in basal CK expression in BLBC and bDCIS were performed using Fisher’s exact test for categorical variables. All tests were 2-sided, and P values < 0.05 were considered statistically significant.
15 female patients who had been diagnosed with BLBC and associated bDCIS were included in the present study (Table 1). Their mean age of BLBC patients was 48 years (range, 33 to 59 years). The mean tumor size was 30 mm (range, 6 to 66 mm). 7 patients had a known family history of breast cancer. Lymph node metastasis and distant metastasis were observed in 6 (40%) and 5 (33%) of the 15 BLBC cases. 5 patients received chemotherapy after resection. 5 patients had clinical stage IV disease after a mean follow-up period of 1.2 years (range, 0.3 to 6 years).
Table 1: Clinical features of bDCIS and bADH cases. View Table 1
Morphologically, all BLBC (100%) cases were histologic grade 3 and showed basal-like morphologic features (Figure 1). Metaplastic carcinomas or heterologous elements were not observed in our case cohort. The associated DCIS showed a trabecular or solid pattern and high nuclear grade. ER, PR, and HER2 were negative in the BLBC and DCIS components of all 15 cases (100%).
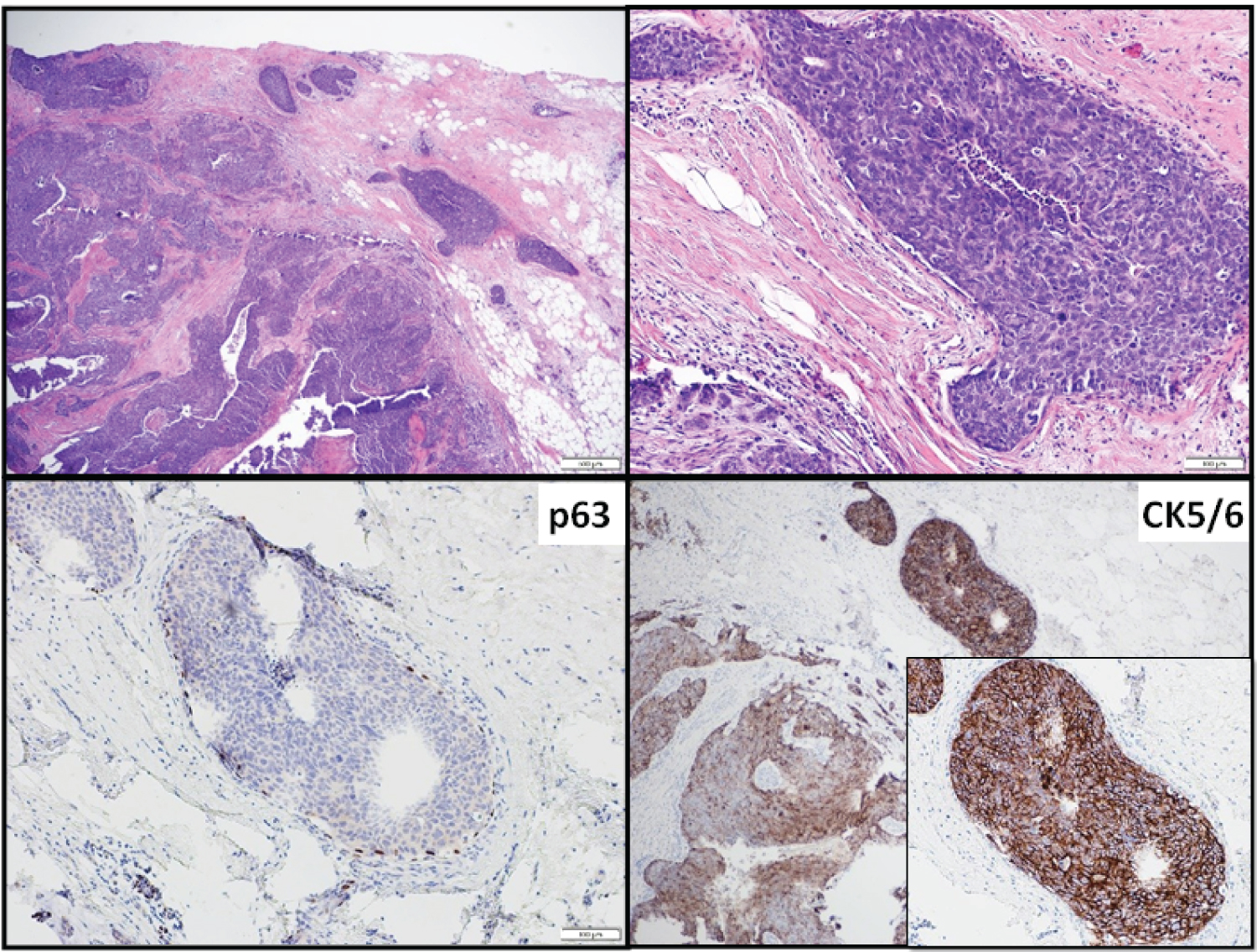 Figure 1: BLBC associated with bDCIS, case 11.
View Figure 1
Figure 1: BLBC associated with bDCIS, case 11.
View Figure 1
Immunohistochemically, in invasive carcinoma components, CK5/6 staining was diffusely strongly positive in 11 cases and patchy positive in the remaining 4, while CK17 was diffusely strongly or moderately positive in 9 of 14 cases; the remaining 5 had patchy staining (Table 2). In bDCIS components, CK5/6 staining was diffusely strongly positive in 11 cases and patchy in 4. CK17 was tested in 14 BLBC and 13 bDCIS cases. CK17 was diffusely strongly positive in 6 and patchy positive in 4 of 10 bDCIS cases; the remaining 3 cases had negative staining.
Table 2: Pathological features of bDCIS and bADH cases. View Table 2
14 of 105 female patients with BRCA1 or BRCA2 mutation and subsequently prophylactic mastectomy were diagnosed with ADH. Upon re-review, 3 of 14 females who met the criteria of basal ADH were included in the present study and 2 other bADH cases encountered in 2021 was included as well. The bADH patients’ mean age was 49 years (range, 44 to 58 years).
Morphologically, bADH showed luminal cell proliferation of multiple cell layers, devoid of secondary lumen formation. Luminal cells are hyperchromatic with an increased nuclear to cytoplasmic ratio exhibiting a rounded to cuboidal shape, intermediate-grade nuclei, and nuclear molding (Figures 2, Figure 3). Immunohistochemically, CK5/6 was diffusely positive in all 5 bADH cases. CK17 was tested in 4 cases and was diffusely positive. ER and PR were negative in 3 cases and weakly low positive in 2 cases.
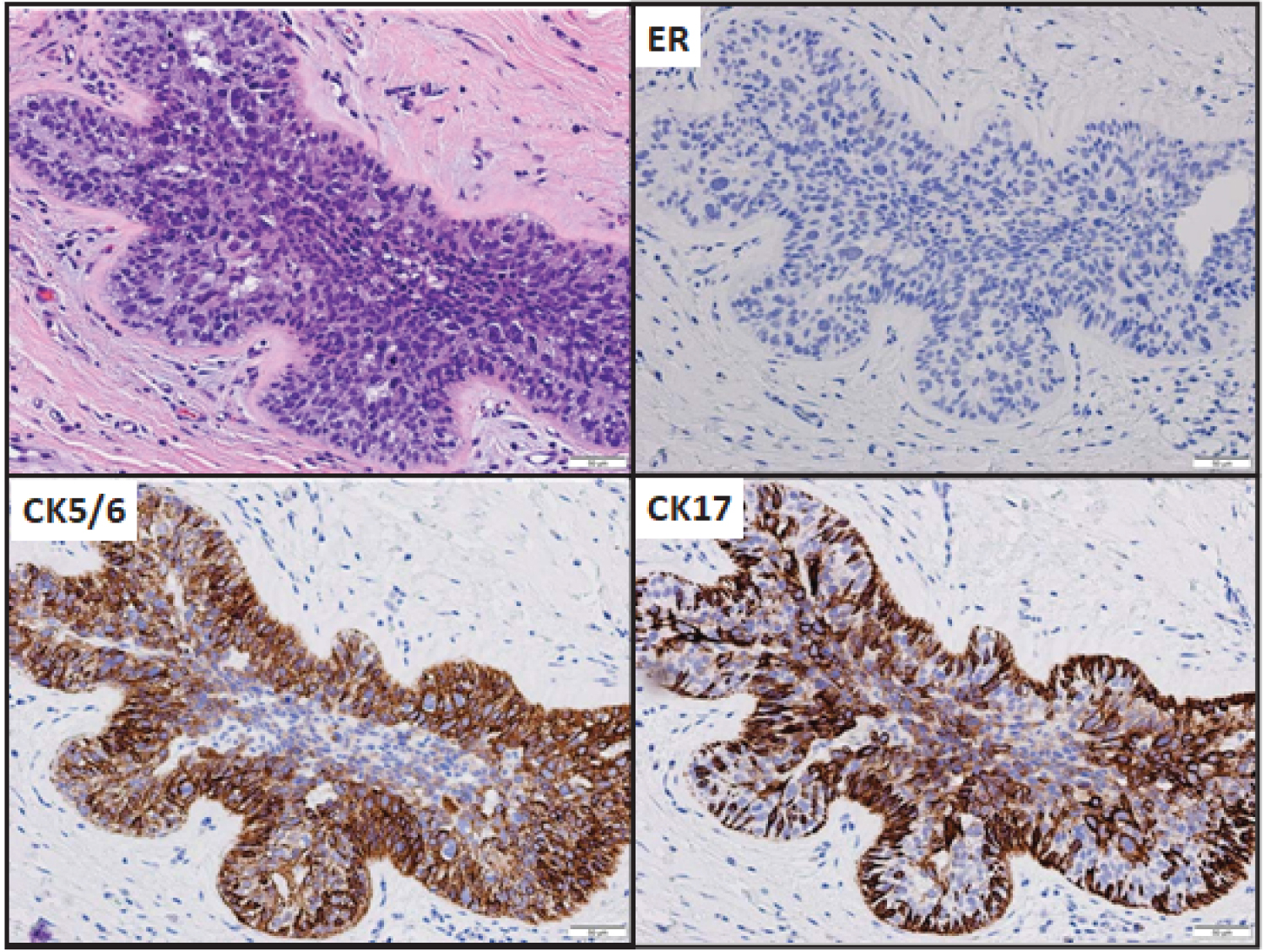 Figure 2: bADH (case 16) with ER and CK expression, original magnification, 200X.
View Figure 2
Figure 2: bADH (case 16) with ER and CK expression, original magnification, 200X.
View Figure 2
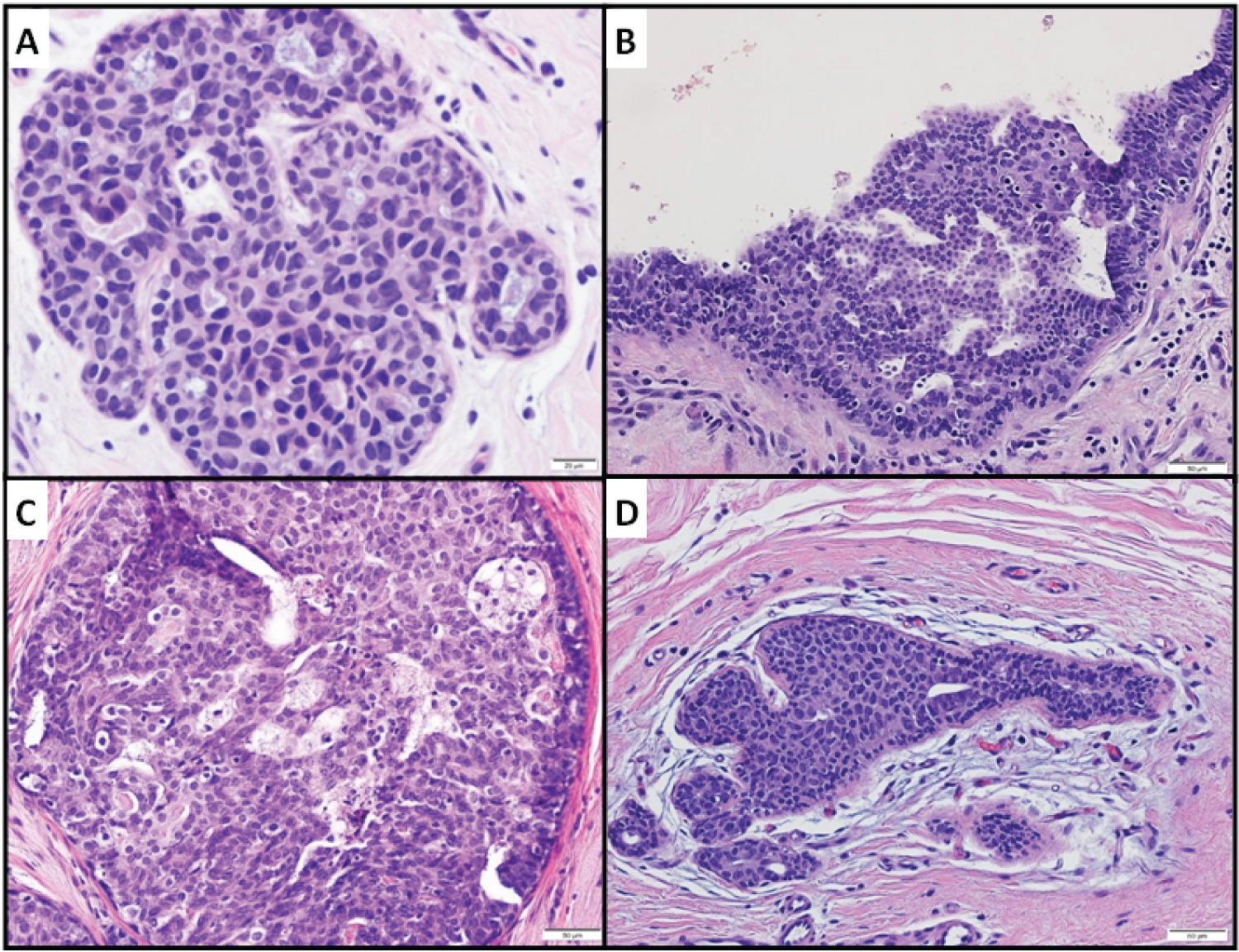 Figure 3: bADH. A, Case 17, H&E magnification x400; B, case 18, 200X; C, case 19, 200X; D, case 20, 200X.
View Figure 3
Figure 3: bADH. A, Case 17, H&E magnification x400; B, case 18, 200X; C, case 19, 200X; D, case 20, 200X.
View Figure 3
CK5/6 showed a significantly (p = 0.03) more sensitive and stronger staining pattern in both BLBC and bDCIS than did CK14 and CK17 (Table 3). CK5/6 showed positive expression in all 15 BLBC and bDCIS cases. 11 of15 (73%) BLBC and 11/15 (73%) bDCIS cases showed diffusely strong CK5/6 staining. In comparison, CK14 was positive in 4 of 9 (44%) BLBC and 5 of 8 (62%) bDCIS cases. 3 of 4 (75%) BLBC cases and all 5 (100%) bDCIS cases showed diffusely strong CK14 staining. CK17 was tested in 14 BLBC and 13 bDCIS cases. All 14 (100%) BLBC cases and 10 (73%) bDCIS cases were CK17 positive. 9 (64%) BLBC cases and 6 (60%) bDCIS cases showed diffusely strong CK17 staining.
Table 3: Basal cytokeratin expression in BLBC and bDCIS. View Table 3
BLBC is triple-negative, high-grade invasive breast cancer that typically expresses 1 or more basal CKs [5,8,9]. CK5/6, 14, and 17 are common basal markers that are expressed in BLBC [6,8,9]. This study demonstrated that CK5/6 is expressed in all BLBC cases, which is consistent with the findings of previous studies. The bDCIS identified in this study are associated with BLBC and shared the same hormone receptor status and HER2 status (ER- negative, PR-negative, and HER2-negative), high histological grade, and the same basal-like immunohistochemical profile as the associated BLBC. These findings suggest that bDCIS represents a precursor lesion of BLBC.
A high prevalence of BLBC has been observed in breast tumors, secondary to heritable BRCA1 mutations, and a high prevalence of BRCA1 dysfunction is identified in sporadic BLBC patients without heritable mutations [13,14,16,17]. The identification of early precursor lesions to BLBC in patients with germ line BRCA mutations or in BRCA carriers is crucial to managing this group of patients and is an important first step in designing targets for BLBC chemoprevention [24]. A stepwise progression of early precursor lesions with cellular atypia to carcinoma in situ and invasive breast cancer is the current model of human breast cancer evolution [25,26]. ADH is considered as an early precursor in the low-grade pathway of breast cancer evolution. In this study, we identified ADH in 13% of patients with BRCA1/2 mutation and bADH in 2% of patients with BRCA1/2 mutation. These findings support the uncommon nature of bADH, even among high-risk cohorts, and suggest that bADH and bDCIS is the precursor lesion of BLBC. A small portion of ADHs, especially in this high-risk population, may be involved in high-grade triple- negative breast cancer progression. However, this hypothesis does not exclude the possibility that some BLBCs progress from CK5/6-positive stem cells or progenitor cells [27,28].
Different types of CK are expressed in the luminal epithelial cells and myoepithelial cells of normal mammary ducts and acini. Low–molecular weight keratin CK7, 8, 18, and 19 are expressed in luminal cells, while high–molecular weight keratin CK5, 14, and 17 are found in myoepithelial and basal cells [4]. However, a small number of luminal epithelial cells have been identified that were positive for CK5/6 but negative for CK8, 18, and 19 and SMA. These cells show the morphologic features of stem cells that have the capacity to differentiate towards the luminal or basal phenotype [29]. As demonstrated in this study, bADH may represent an intraductal proliferative lesion of CK5/6-positive stem cells from the luminal basal area into the lumina, while low-nuclear grade ADH represents the proliferation of CK5/6-negative differentiated luminal cells. CK5/6, 14, and 17 positivity in BLBC and in bDCIS associated with BLBC suggests that some BLBCs progress from CK5/6-positive stem cells and progenitor cells.
Among the 3 basal CKs we tested, CK5/6 is the most sensitive marker, showing 100% positivity in both BLBC and bDCIS. CK17, with 100% positivity in BLBC and 77% in bDCIS, is more sensitive than CK14, which showed 44% positivity in BLBC and 62% in bDCIS cases. Our finding of CK 5/6, CK14 and CK17 positivity in in situ lesions (bADH/bDCIS) has also been reported by Thike, et al. [12] CK5/6, together with ER, are commonly used makers to differentiate usual ductal hyperplasia from ADH and DCIS. The results of previous studies indicate that CK5 staining is absent or only focally positive in 97% of ADH and all low-grade DCIS cases, while CK5 staining exhibits a strong, mosaic pattern in usual ductal hyperplasia cases [30,31]. However, our study demonstrated that a small portion of ADH cases exhibited strong CK5/6 positivity and ER negativity or low positivity. Strongly positive CK5/6 expression was observed in bDCIS associated with BLBC as well. It is important to highlight that there is an atypical intraductal proliferation lesion exhibiting basal-like morphologic features and diffuse CK5/6 staining. In particular, an atypical intraductal proliferative lesion with basal-like morphologic characteristics should not be classified as benign, only on the basis of diffuse staining with CK5/6. The weak to low positivity of ER in 2 cases of bADH suggests that as basal-like neoplastic cells progress from atypia to in situ to invasive carcinoma, they lose functional expression of ER.
We observed atypical intraductal basal-like proliferations of rounded cells with intermediate nuclear grade and multiple cell layers thick that were ER-negative and CK5/6 positive; these findings are strong circumstantial evidence for a precursor basal-like ADH lesion. Importantly, there is an atypical intraductal basal-like lesion with diffuse CK5/6 staining; in the context of atypical hyperplastic lesions with basal morphologic characteristics, diffuse staining with CK5/6 should not be used to confirm benignity. A relatively high prevalence of ADH, including bADH, was observed in patients with BRCA1 or BRCA2 mutation. Awareness and identification of possible precursors of BLBC is important to gain insight into tumorogenesis and the development of potential therapeutic targets.
We would like to thank Ann Sutton from the Department of Research Medical Library, at The University of Texas MD Anderson Cancer Center for her assistance in editing this document and Kim-Anh Vu from the Department of Pathology for her help with graphic images.