Almost 60% of normal healthy term babies and 80% of preterm babies develop visible jaundice in the first week of life. Although the outcome of the majority is benign, newborns with untreated severe hyperbilirubinemia can develop short and long-term sequelae of bilirubin encephalopathy.
This study was designed to evaluate the impact of exchange transfusion on various biochemical parameters and to assess the need for routine calcium gluconate supplementation during the procedure.
We studied 84 newborn double volume exchange transfusions and recorded various biochemical parameters of sodium, potassium, calcium, ph and blood sugar during the procedure. Mostly fresh donor blood was employed for the procedures. Nearly 74% transfusions were performed with donor blood aged less than 2 days.
We observed no clinically significant alteration in recorded biochemical parameters during and after exchange transfusion. Routine intravenous calcium gluconate was not given to patients during the procedure. Though we observed a fall in serum calcium level during transfusion, but the concentrations remained within normal physiological range. Insignificant variation in recorded parameters and zero mortality were attributed to mostly using fresh blood for the exchange transfusions.
We concluded that there was no significant alteration in serum electrolytes during and after exchange transfusion. Further, in contrast to the earlier reports of higher incidence of hyperkalemia during exchange transfusion, we observed a paradoxical fall in serum potassium during and after transfusion, although within normal range.
Neonatal hyperbilirubinemia, Exchange transfusion, Biochemical parameters, Calcium gluconate, CPDA blood
Jaundice is one of the most common clinical problems in neonatal medicine [1]. Almost 60% of normal healthy term babies and 80% of preterm babies develop visible jaundice in the first week of life [1]. A significant proportion of these neonates develop pathological hyperbilirubinemia, which is defined as hyperbilirubinemia that requires treatment during first week of life [1,2]. Neonatal jaundice is considered as one of the most common causes of readmission after early discharge from post-natal ward [3]. Premature babies have much higher incidence of neonatal jaundice requiring therapeutic intervention, than term newborns [4]. Although the outcome of the majority is benign, newborns with severe hyperbilirubinemia can develop acute bilirubin encephalopathy which could lead to higher mortality or kenicterus spectrum disorders, later on in life [5,6]. In order to prevent bilirubin induced neurologic dysfunction in acute hyperbilirubinemia, especially due to Rh or ABO incompatibility or G6PD deficiency where rate of bilirubin rise is very high, the exchange blood transfusion (EBT) is indicated to rapidly decrease the free bilirubin and the antibody load [6]. Although it is considered to be a safe undertaking, but changes may take place in various serum biochemical parameters, plasma osmolality and electrolyte profile in the infant's blood, which may be significant at times, giving rise to various complications including hyponatremia, hypoglycemia, and hyperkalemia, after the exchange [7-10].
This prospective study was conducted to evaluate the impact of fresh CPDA-1 anticoagulated blood on various biochemical parameters during exchange transfusion and to assess routine use of intravenous calcium gluconate for prevention of hypocalcemia during the procedure.
The study was conducted in the Neonatal intensive care unit, Department of Pediatrics and Neonatology, Sher-Kashmir Institute of Medical Sciences Srinagar Kashmir. The department has its own laboratory facility for biochemical analysis with semi-automatic Gem 3000 analyzer for Blood gases, electrolytes, lactate, bicarbonate and ionized calcium. The study was conducted over a period of two years from January 2014 to December 2015. The indications for exchange transfusion were different for preterm and term babies. Based on our current protocol, critical values for exchange transfusion in preterms include: Infants < 1 kg at 10 to 12 mg/dl, infants 1-1.5 kg at 12-15 mg/dl, infants 1.5-2 kg at 15-18 mg/dl and infants 2-2.5 kg at 18-20 mg/dl [11]. Indications for exchange transfusion in term infants were based on the consensus guidelines of American academy of pediatrics Subcommittee [12] (Figure 1). We excluded babies with shock, renal failure and those with underlying biochemical and electrolyte abnormalities.
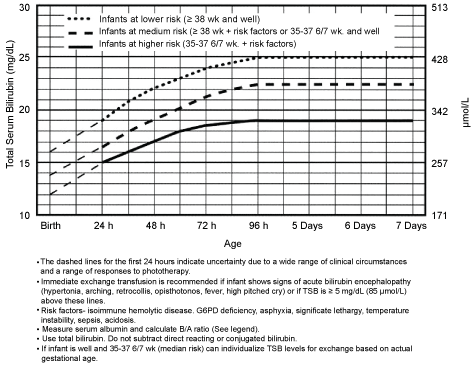 Figure 1: Guidelines for exchange transfusion in infants 35 or more weeks' gestation.
Figure 1: Guidelines for exchange transfusion in infants 35 or more weeks' gestation.
• The dashed lines for the first 24 hours indicate uncertainty due to a wide range of clinical circumstances and a range of responses to phototherapy.
• Immediate exchange transfusion is recommended if infant shows signs of acute bilirubin encephalopathy (hypertonia, arching, retrocollis, opisthotonos, fever, high pitched cry) or if TSB is ≥ 5 mg/dL (85 µmol/L) above these lines.
• Risk factors- isoimmune hemolytic disease. G6PD deficiency, asphyxia, significate lethargy, temperature instability, sepsis, acidosis.
• Measure serum albumin and calculate B/A ratio (See legend).
• Use total bilirubin. Do not subtract direct reacting or conjugated bilirubin.
• If infant is well and 35-37 6/7 wk (median risk) can individualize TSB levels for exchange based on actual gestational age.
View Figure 1All EBTs were performed under standard aseptic conditions via umbilical catheterization or via peripheral route using radial artery (for removing blood) and a peripheral vein (for fresh blood infusion). Blood exchange was done in aliquots of 5 ml for < 1.5 kg, 10 ml for 1.5-2.5 kg, 15 ml for 2.5-3.5 kg and 20 ml > 3.5 kg babies. The amount of blood exchanged was double the infant's total circulating blood (85 ml/kg × 2). The selection of blood for transfusion was done depending upon the blood group of babies as well as mother and was selected as per the following criteria:
• Fresh < 7 days old irradiated whole blood or reconstituted whole blood (hematocrit 40-50%) made from packed RBCs and fresh frozen plasma collected in citrate phosphate dextrose.
• In Rh hemolytic disease, if blood was prepared before delivery type O Rh negative cross matched against mother's blood was used.
• In ABO incompatibility the blood type O Rh negative or Rh compatible with the mother and infant was used. Usually type O used with AB plasma to ensure that no anti A or anti B antibodies were present.
• In non-immune hyperbilirubinemia blood typed and cross matched against the plasma and RBCs.
• Continuous monitoring of hemodynamic parameters (heart rate, respiratory rate, capillary refill time and blood pressure) was done during the procedure. Assessment of serum levels of sodium, potassium, calcium, Ph and glucose was done at different intervals i.e, before, during, immediately after exchange and at 12 hours post-exchange, using 1 ml of peripheral blood for each analysis. Babies with shock and underlying biochemical and electrolyte abnormalities were excluded.
The study was approved by the institutional ethics committee and written informed consent were obtained from the parents/guardians of the patients.
The observed data was entered in the computer to analyze with the help of MS, Excel and SPSS version 20 for windows. The values of biochemical parameters were presented as mean and SD and statistically significant difference was evaluated using one-way analysis of variance (ANOVA). Pre and post-exchange variables were compared using Chi square test. A p value of < 0.05 was considered as significant.
Out of a total of 106 EBTs performed over the study period, 84 were included. Transfusions were performed via umbilical catheterization in 76(90.4%) and via peripheral route in 8(9.6%) patients. Regarding the baseline characteristics-59.5% were females and 40.5% were males, 76.2% and 23.8% were term and pre-term respectively. We observed that 19% patients had Rh incompatibility, 47.6% had ABO incompatibility and in 33.3% cases the cause of hyperbilirubinemia was unexplained. The direct coombs test was positive and negative in 35.7% and 64.3% patients respectively (Table 1). The mean age of the donor blood in our study was 2.24 days (Table 2). The mean serum glucose before, during and after exchange was 68.2 ± 25.3 mg/dl, 95.7 ± 31.6 mg/dl and 101.5 ± 40 mg/dl respectively. The rise in serum glucose during exchange was statistically significant (p < 0.05). However, serum glucose decreased to levels comparable to pre-exchange values at 12 hours after exchange (p > 0.05). The mean blood ph before, during and after exchange was 7.40 ± 0.04, 7.41 ± 0.03 and 7.36 ± 0.03 respectively. Though the fall in ph during exchange was statistically significant, normal ph comparable to pre-exchange values got restored at 12 hours post exchange. There was no significant change in serum sodium during exchange transfusion. Mean serum potassium of patients before exchange was 4.29 ± 0.70 mEq/L, during exchange was 3.95 ± 0.51 mEq/L and at completion of exchange was 3.93 ± 0.63 mEq/L. The decrease in potassium during exchange was statistically significant (p < 0.05). As compared to pre-exchange levels serum potassium was persistently low (3.69 ± 0.38 mEq/L) even at 12 hours post exchange. We observed a significant fall in serum calcium of patients during exchange transfusion. Serum calcium decreased from a pre-exchange level of 9.22 ± 0.61 mg/dl to a post-exchange level of 8.59 ± 0.74 mg/dl. The serum calcium values remained low (8.34 ± 0.59 mg/dl) even at 12 hours after exchange (Table 3).
Table 1: Baseline patient characteristics. View Table 1
Table 2: Age of donor blood with mean serum potassium and ph. View Table 2
Table 3: Patient serum parameters of calcium, sodium, potassium, glucose and ph. View Table 3
In our study ABO incompatibility was the most common indication for exchange transfusion in neonatal jaundice (47.6%). Sgro M, et al. and Chen, et al. also observed ABO incompatibility to be the most common cause for exchange transfusion [5,8]. There was no death, nor any serious untoward event reported during any of the procedures. J Craig Jackson in their study observed that at least 2 (2%) of the 106 patients died of complications probably attributable to exchange transfusion and both deaths were reported in very sick neonates [13]. Sick newborns and those with hemodynamic instability were excluded from our study. The mean pre-exchange transfusion serum sodium level observed was on higher side of normal range (145 ± 7.81 mEq/L), likely due to decreased intake of breastmilk and physiological loss of extracellular fluid, which characterize the first few days of life [14,15]. Furthermore, there was no significant change in serum sodium levels possibly because the donor mean sodium level of 143 mEq/L was almost equal to that of the patient before EBT. These observations differ from those of earlier workers who have reported transient but significant hypernatremia after exchange by hypernatremic donor blood [16], but consistent with the findings of Patra K, et al. [9]. Clinical hyperkalemia resulting from RBC transfusions has been recognized as a transfusion complication for decades and the risk increases with the duration of blood storage and irradiation of donor blood. The rise in patient potassium after transfusion is usually transient due to the redistribution of the potassium load within the body [10,16]. In contrast to above discussion we observed a statistically significant fall in mean serum potassium during and in post exchange period, though within the normal physiological range. The fall in serum potassium persisted even at 12 hours post-exchange. The lower serum potassium levels may be because we mostly used fresh irradiated donor blood for exchange transfusion, which mitigated chances of significant hyperkalemia. Further the low serum potassium coincided with higher serum pH (alkalosis), which is known to cause shift of serum potassium into intracellular compartment. Relatively higher serum pH may be attributed to higher serum bicarbonate concentration in freshly CPDA treated blood. The concentration of serum bicarbonate in CPDA treated blood usually starts decreasing after 3 days of storage [17].
We observed a statistically significant decrease in serum calcium during transfusion, though within physiologically normal range. Such fall occurs due to calcium chelating properties of citrate which is present in high concentration in CPDA treated donor blood [17,18]. Singh M, et al. in their study on effect of exchange transfusion on bilirubin and calcium level in neonatal hyperbilirubinemia observed a transient rise in serum calcium in post-exchange period which was attributed to routine intravenous administration of 10% calcium gluconate after each 100 ml of blood exchange in order to counteract any fall in serum ionized calcium levels [18]. In our study we did not employ routine use of i.v. calcium gluconate during exchange transfusion. There was a significant rise in post exchange serum glucose, attributed to high dextrose content in the donor blood along with anticoagulants for the preservation [13]. Although normal mean blood sugar was observed at 12 hrs post-exchange, one needs to be cautious as rebound hypoglycemia may occur during the initial hours. So, monitoring of blood glucose becomes mandatory and continuous glucose input may be needed for next few hours either by iv infusion or orally. Most current guidelines for exchange transfusion recommend donor blood which is as fresh as possible (preferably less than 5 days old) to avert any possible complications [19]. In our study we did not observe any physiologically significant change in biochemical milieu during and after exchange transfusion. The possible explanation is that we mostly employed fresh donor blood (mean age 2.24 days) for exchange transfusion.
The study demonstrated changes in serum glucose, potassium and calcium levels which were not clinically significant and resolved spontaneously within 12 hours. However, in contrast to the earlier reports of higher incidence of hyperkalemia during exchange transfusion, we observed a paradoxical fall in serum potassium during and after transfusion, although within normal range. The lower serum potassium levels were attributed to use of mostly fresh irradiated blood for transfusion and possible intracellular shift due to concomitant relative metabolic alkalosis due to CPDA treatment of donor blood. Further, we reported that routine intravenous calcium gluconate is not needed if fresh blood is employed for exchange transfusion.
Dr. Syed Wajid and Dr. Bashir Ahmad Charoo conceptualized and designed the study, drafted the initial manuscript, coordinated and supervised data collection and approved the final manuscript as submitted. Mr Mohd Ibrahim and Dr. Mohd Rafiq carried out the initial analyses, reviewed and revised the manuscript, and approved the final manuscript as submitted.
Dr. Mudasir Nazir and Dr. Roumissa Lone designed the data collection instruments, and coordinated and supervised data collection, critically reviewed the manuscript, and approved the final manuscript as submitted.
The authors have no financial relationships relevant to this article to disclose.
The authors have no conflicts of interest relevant to this article to disclose.