Improvement of neonatal intensive care in the modern era has greatly changed the demographic image with evolving new generation of extreme preterm survivors. And indeed, evolving new co-morbidities that influence the short and long-term outcomes of these infants.
Periventricular-Intraventricular hemorrhage (PVH-IVH) is a significant health problem affecting the brain structures in a critical stage of development in preterm infants. Although the incidence has declined since the 80's of the last century, PVH-IVH continued to be a costly co-morbidity in this subset of infants. Life-long neurological sequelae especially cerebral palsy, seizures, coordination disorders, and developmental delay are major concern to health care givers and represent significant economic and social burden to the family. This review offers an update on the pathogenesis, diagnostic neuroimaging modalities, and the long-term neurodevelopmental outcomes of mild grades PVH-IVH in preterm survivors.
Cerebral palsy, Intraventricular hemorrhage, Neurodevelopmental outcomes, Preterm infant
PVH-IVH: Periventricular Hemorrhage-Intraventricular Hemorrhage; CP: Cerebral Palsy; WMI: White Matter Injury; cUS: Cranial Ultrasound; MRI: Magnetic Resonance Image
Intraventricular hemorrhage is considered as the most common central nervous system bleeding in preterm infants particularly very low birth weight neonates (VLBW) weighing less than 1500 g with gestational age less than 28 weeks. The incidence of PVH-IVH is inversely related to the gestational age of preterm infants being highest among extreme premature infants with extreme low birth weights [1]. It is estimated that the incidence of PVH-IVH reach up to 30% in infants born between the 23rd and 26th week of gestation [2]. Bleeding in brain ventricles occurs because blood vessels in the germinal matrix are quite fragile and susceptible to different noxious stimuli that eventually lead to rupture and bleeding [3]. Cranial ultrasonography remains the main diagnostic tool of IVH despite the limited ability to define white matter injuries and cerebellar lesions which are important determinants of the future neurocognitive performance among IVH survivors. The pathophysiology, classifications, common risk factors and clinical presentation of PVH-IVH in preterm infants will be reviewed. Furthermore, this review aims to directly assess the current neuroimaging techniques and how the lower grades IVH impact the future neurocognitive outcomes of IVH survivors.
PVH-IVH typically originates at the germinal matrix which is a highly cellular structure consisting of neuronal and glial precursor cells. It has extremely fragile blood vessels and a free communication between the capillary network and the venous system located mainly between thalamus and caudate nucleus at the level of foramen of Monro. Impaired cerebral autoregulation in preterm infants represents the second major determining factor in PVH-IVH pathogenesis. Fluctuation in systemic blood pressure is associated with significant similar swings in cerebral blood flow velocities leading to damage of the vulnerable fragile capillary network and bleeding in the germinal matrix when cerebral autoregulation is impaired. A comparison of the anatomical classification systems of PVH-IVH by Papile and Volpe is given in Table 1. Grades I-II IVH are considered as mild grades while grades III-IV are labelled as severe IVH [4,5].
Table 1: Comparison between Papile and Volpe Grading classification of PVH-IVH. View Table 1
The pathogenesis of PVH-IVH is multifactorial and complex. The Table 2 summaries the different risk factors for intraventricular hemorrhage in preterm infants. The large capillary network in the geminal matrix is very delicate and thin walled and lacks adequate anatomical support. It is highly sensitive to hypoxic insults and increased venous pressure, with increased permeability and easy bleeding in the sub-ependymal region that can progressively extend to the ventricular system [6]. The fragility of the matrix microvasculature comes from: 1) Insufficiency of pericytes, which are the mural cells or the vascular endothelial smooth muscle cells; 2) Fibronectin deficiency in the basal lamina: Fibronectin molecules beside collagen, heparan and laminin are important components of basal lamina which is an essential structure of the blood brain barrier (BBB); 3) Glial fibrillary acidic protein (GFAP) deficiency in astrocytes: GFAP is a nanofilament protein in the astrocytes processes (end-feet) that extend around blood vessels to provide structural and haemostatic support to the BBB.
Table 2: Schematic diagram of the common risk factors of PVH-IVH. View Table 2
Most cases of PVH-IVH in preterm infants take place in the first postnatal week although the occurrence maybe much later. The presentation of hemorrhage can be either a catastrophic event with collapse and neurologic deterioration that progresses within hours to respiratory depression and coma [7]. The second presentation often follows a subtler course with nonspecific easily overlooked signs such as hypotonia, pallor, respiratory distress and often diagnosed on routine cranial ultrasound examinations [8].
It is the most commonly used diagnostic tool with high sensitivity (96%) and specificity (94%). It is cheap, non-invasive and allows several bed side brain scans with comfort [9,10]. It is the method of choice to screen for and follow up the progression of PVH-IVH in preterm infants especially extremely low birth weight infants less than 1000 g (Figure 1). Cranial ultrasonography limitations include operator dependency, scanning difficulty of the posterior cranial fossa and subtle diffuse white matter abnormalities, and the limited prediction of the outcome in the high-risk preterm infants [11].
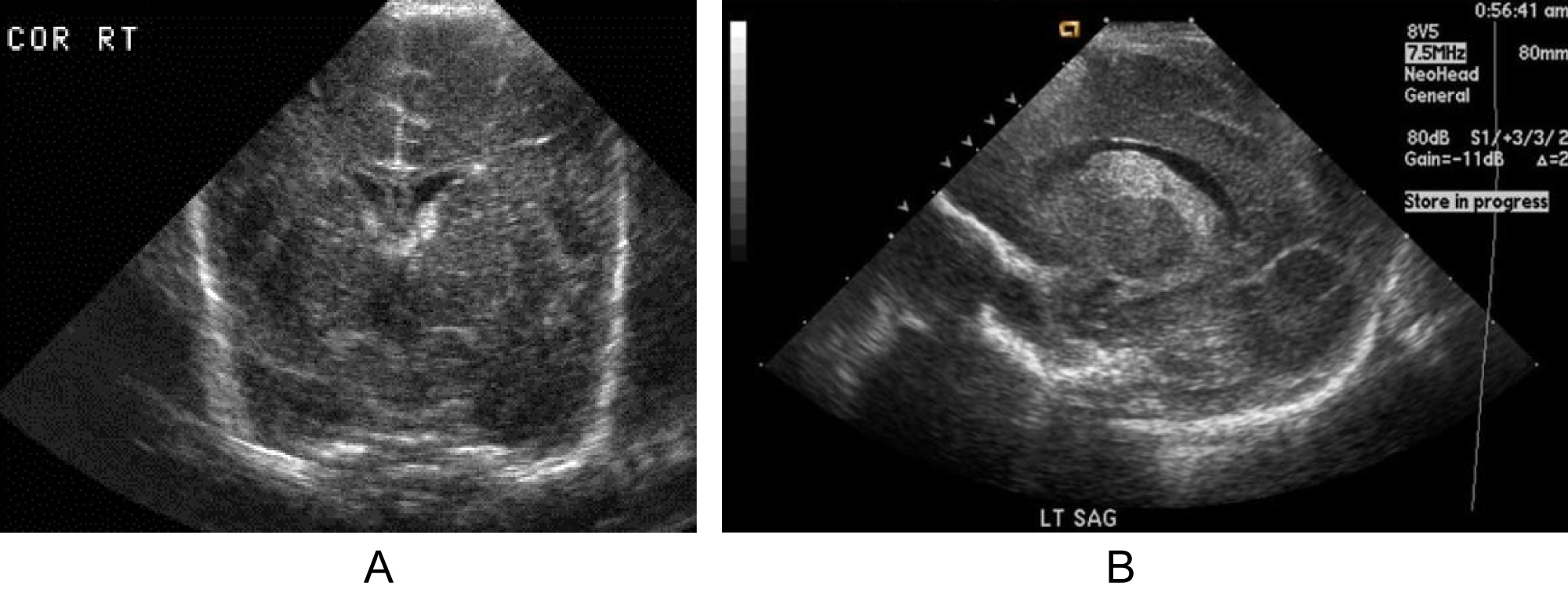 Figure 1: A) Grade I sub-ependymal hemorrhage in a 5- day old premature infant, Coronal view ultrasound; B) Grade II PVH-IVH in sagittal view.
View Figure 1
Figure 1: A) Grade I sub-ependymal hemorrhage in a 5- day old premature infant, Coronal view ultrasound; B) Grade II PVH-IVH in sagittal view.
View Figure 1
It is a powerful non-ionising tool that can effectively diagnose subtle brain lesions in preterm infants particularly diffuse non-cystic PVL, infarctions, and posterior fossa abnormalities (Figure 2). However, the need for transport to MRI scan, the long-time of examination, and the other logistic issues has limited its utilisation in neonatal units [12]. Unfortunately, the current neuroimaging tools including cUS and even the conventional MRI, cannot correlate accurately with the microscopic subtle neuropathological diagnoses like gliosis and neuronal-axonal injury which need micron and not millimetre working MRI resolution [13].
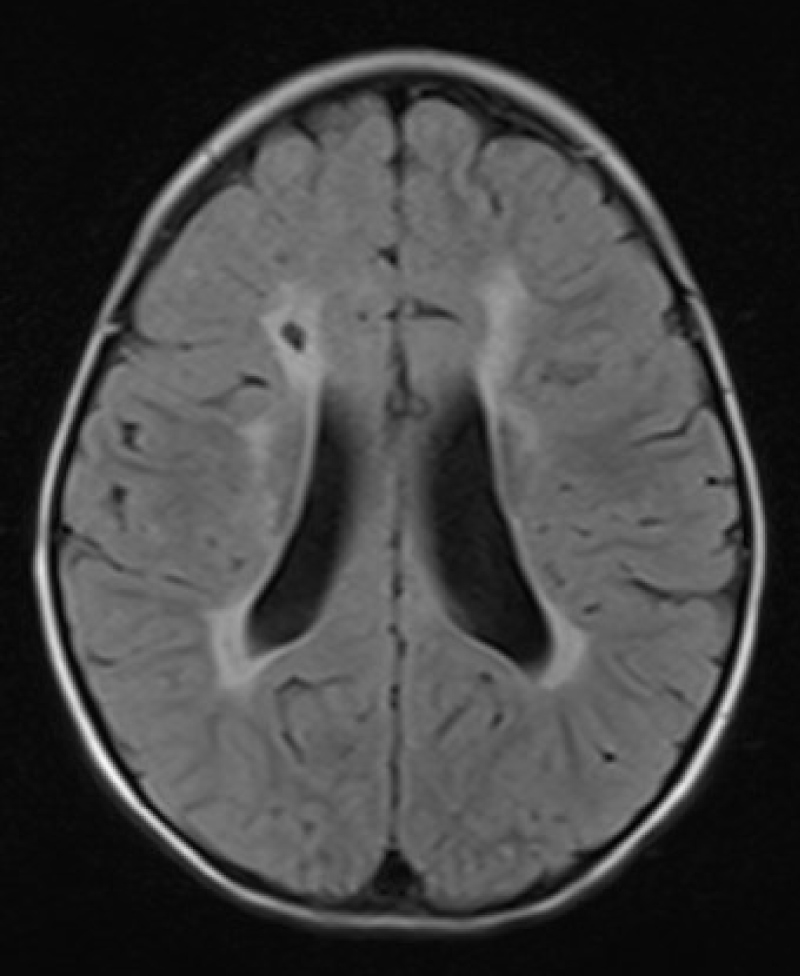 Figure 2: Brain MRI, Periventricular leukomalacia.
View Figure 2
Figure 2: Brain MRI, Periventricular leukomalacia.
View Figure 2
Through computational analysis of brain structure, connectivity, and function; quantitative brain MRI can analyse metabolic changes associating subtle perinatal brain injuries. Thus, with clinical correlation, it may establish cognitive and neurodevelopmental prognosis in preterm infants with PVH-IVH at earlier ages [14,15]. The reduction in cerebral, cerebellar, and basal ganglia volumes in preterm infants with white matter injury is well documented in previous studies and can be detected by the volumetric brain MRI imaging at term equivalent age [16,17].
It is an advanced computational MRI technique assessing changes in water molecule diffusion parameters in the brain tissues which are early indicators of neonatal brain damage (Figure 3). With assessing and quantifying the early WM microstructures integrity, it can predict future cognitive and motor outcomes of IVH survivors [18,19].
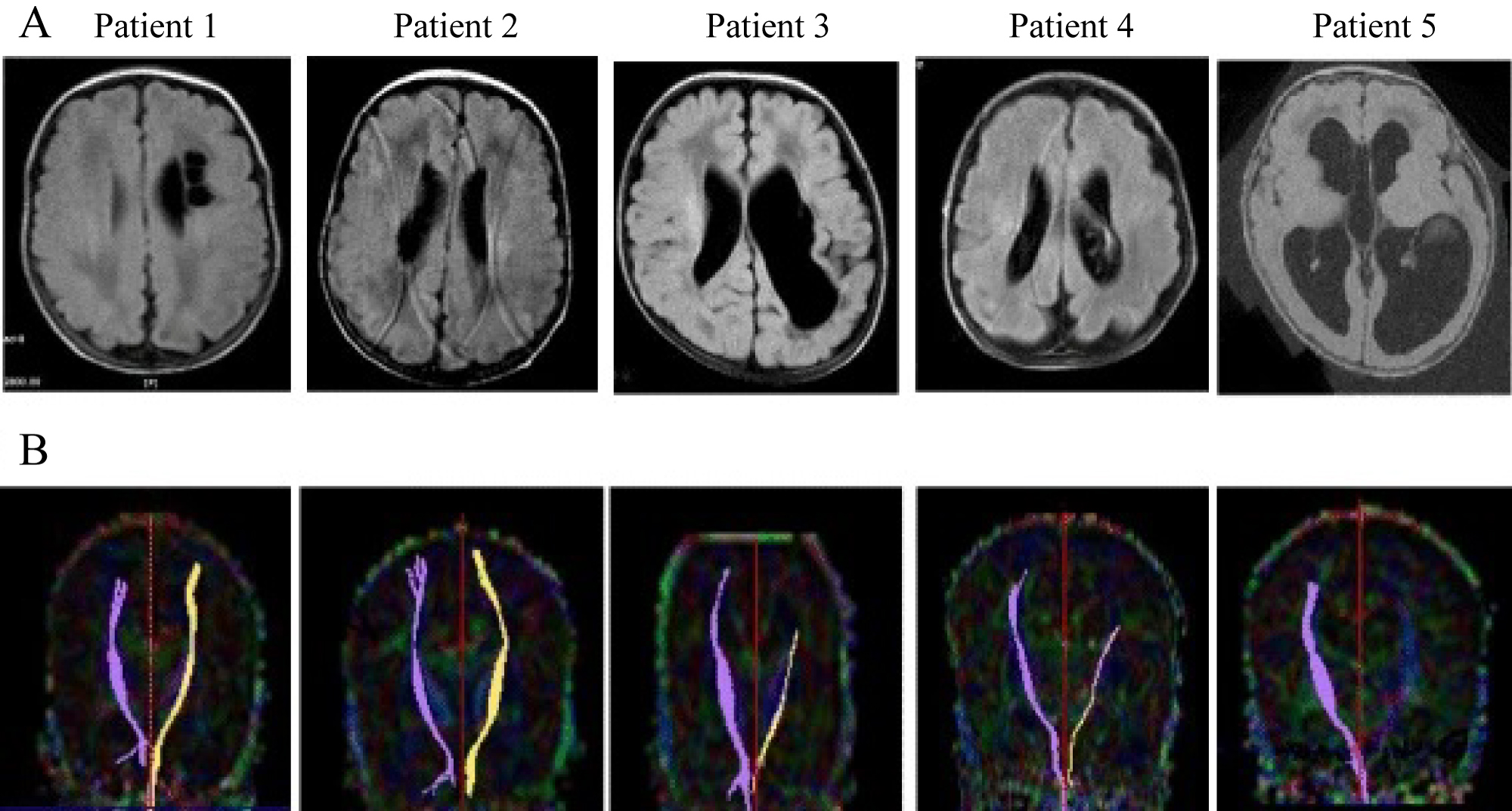 Figure 3: Diffusion tensor imaging (DTI). Preterm infants with WMI and CP. A) Axial images on T2 flair; B) DTI showing the trajectory of motor fibres normally in patients 1,2 and absent in patient 5 on left side.
View Figure 3
Figure 3: Diffusion tensor imaging (DTI). Preterm infants with WMI and CP. A) Axial images on T2 flair; B) DTI showing the trajectory of motor fibres normally in patients 1,2 and absent in patient 5 on left side.
View Figure 3
It is estimated that 35-40% of infants with IVH will suffer from periventricular leukomalacia (PVL), hydrocephalus, neurocognitive delay, cerebral palsy (CP), or seizures [20]. A spectrum of WMI has expanded greatly after brain MRI's widespread use and included unrecognised lesions commonly missed by cUS screening. PVL with diffuse necrosis and gliosis is commonly associated with PVH-IVH. IVH could be the pathogenetic mechanism for the development of PVL and other mixed patterns of encephalopathy of prematurity [21]. These complications worsen with increased severity of PVH-IVH and decreased gestational age. Mortality rate is directly proportional to the severity of IVH. Mortality rate reaches 4-10 percent in lower grades PVH-IVH, and up to 30%-40% for grades III and IV [22].
The prediction of neurodevelopmental outcome in preterm infants with PVH-IVH is dependent on three main criteria: 1) Clinical assessment; 2) Neuroimaging evaluation; 3) Neuropathological findings. Neurodevelopmental outcome is a composite term, and assessment typically includes neurologic, sensory, and cognitive evaluation preferably by an experienced researcher at 18-24 months corrected age. Bayley Scales for Infant and Toddler Development is a reliable tool for developmental assessment comparing results to a standardized norm [23]. In this scale, fine and gross motor development, cognitive development, and receptive and expressive language are assessed and scaled to a mean of 100 with a standard deviation (SD) of 15. A score less than 85 is classified as "at risk" of developmental delay and a score of less than 70 on any of the subscales is considered "delayed" [24]. Other tools involving parents as an active role player in neurodevelopmental assessment team are gaining popularity such as The Ages and Stages Questionnaire (ASQ). Grades III-IV PVH-IVH are associated with definite moderate to severe neurodevelopmental impairment in all domains compared with neonates who have no IVH as shown in many previous reports. However, there is a growing body of evidence that the long-term prognosis of preterm infants with severe PVH-IVH is more favourable than previously thought [25-27].
The details of search strategy to collect the required evidence of neurodevelopmental outcomes in preterm survivors with mild grades of IVH are illustrated in Table 3. The inclusion criteria included: 1) Studies which explored the relationship between PVH-IVH, and neurodevelopmental outcome reported at 18 months of corrected gestational age or later; 2) Studies in preterm infants less than 37 weeks of gestational age; 3) Articles in English language and human based studies; 4) And publication date after January 2000 reflecting relatively recent NICU practices.
Table 3: Details of search strategy. View Table 3
Based on the predetermined criteria of article selection, the electronic searches of the manuscript of the selected studies were investigated and reviewed for suitability of inclusion. The following data were extracted from each study: The authors, publication year, study design, methods, study population, risk of bias, follow up timing, the developmental outcome definitions, completeness of data at follow up, and applicability. For this review, we designed a modified Cochrane data collection form (Appendix 1) that was used for data extraction and management.
The selected articles were examined for the relevance to the posted purpose and were evaluated by means of a quality assessment tool, The Modified Newcastle-Ottawa Scale for cohort studies (NOS) [28]. NOS was developed by the University of Newcastle (Australia) and the University of Ottawa (Canada) for the assessment of the non-randomised trials included in systematic reviews. The scale consists of eight questions about the cohort selection, the comparability, and the assessment of the outcome. The initial database search identified 1650 articles using the mentioned keywords. After removal of duplicates and analysis of the title and abstracts, 29 potentially relevant citations were found to match the inclusion criteria. Twenty articles were excluded after revision of the full-text articles, and 9 articles were selected for inclusion (Figure 4). Variables of the selected studies including authors, population characteristics, control characteristics, age of assessment and follow up rates, primary objectives, and neurodevelopmental outcomes are illustrated in Table 4.
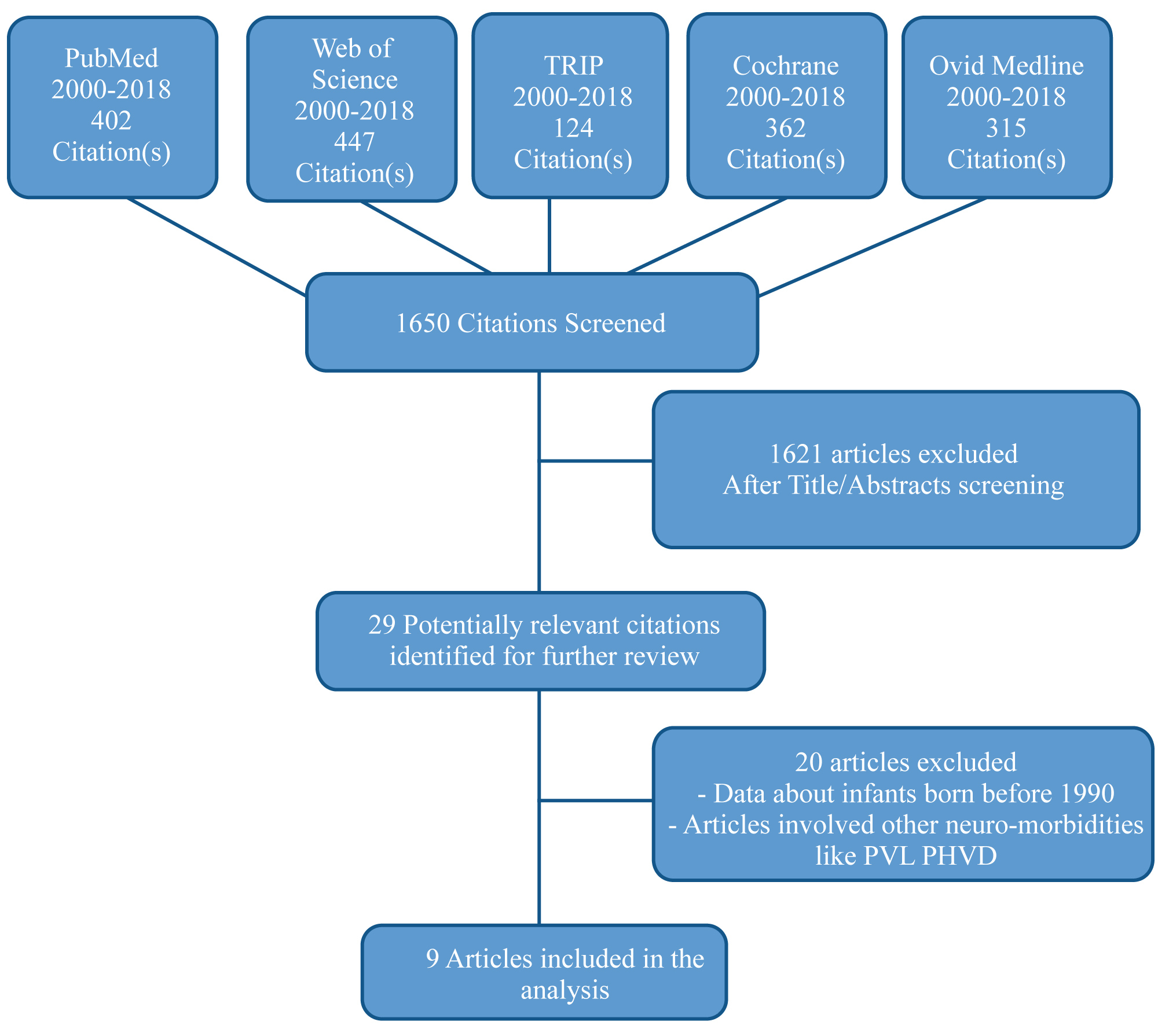 Figure 4: Flow diagram of study selection process.
View Figure 4
Figure 4: Flow diagram of study selection process.
View Figure 4
Table 4: Summary of the selected trials on neurodevelopmental outcome following IVH grades I-II. View Table 4
The impact of mild PVH-IVH on the neurodevelopmental outcomes remains elusive. As illustrated in Table 4, Some studies showed significant neurodevelopmental disabilities, CP and cognitive/language impairments in preterm infants with mild PVH-IVH compared to infants without IVH [29-33]. Other studies refuted any significant long-term neurodevelopmental disabilities with mild grades PVH-IVH particularly those utilised MRI as an adjuvant neuroimaging modality [34-37]. The conflicting results of these studies should be interpreted cautiously in view of different methodological limitations, cohort definition, evaluation methods, and the understanding of the developmental outcomes in each study. Furthermore, This may be explained by the bias in recruitment of preterm infants with low grade IVH who were evaluated solely with cranial ultrasound (cUS). Diffuse white matter injury (WMI) is commonly associated with PVH-IVH, even with the milder grades, hence, it potentially impairs cognitive, sensory and motor function development in premature infants [38,39]. Behaviour and cognitive deficits may only manifest late and present beyond school age [40]. In ELBW infants, Grade I-II PVH-IVH with no documented white matter injury is not innocent and is associated with moderate to severe neurosensory impairments, cerebral palsy, and deafness at 2-3 years corrected age [20,30,41]. A meta-analysis by Mukerji A, et al. [42] concluded that mild PVH-IVH was associated with higher odds of long-term moderate to severe neurodevelopmental impairment (NDI) but not cerebral palsy or cognitive delay at 18-24 months corrected age. It is obvious that studies assessed the neurodevelopmental outcomes at the age of 2-3 years by Klebermass, et al. [29], Bolisetty, et al. [30], and Patra, et al. [31] showed adverse neurodevelopmental outcomes compared to studies assessing the developmental outcomes at later age groups of 8, and 18 years by Sherlock, et al. [34] and Ann Wye, et al. [35] respectively that denied measurable effects of mild IVH on neurocognitive outcomes. This can be explained by the fact that postnatal period from 16 months to 2.5 years is associated with characteristic differentiation and maturation of CNS. A rapid synaptogenesis, myelination, and dendritic growth spurts occur in a high rate followed by a process of selective refinement giving a chance to the environmental factors to influence CNS structures [43]. The initial overproduction of synapses in this period provides a room for potential neuronal recovery after initial early brain insult [44]. The neurodevelopmental milestones acquisition is a dynamic process and not merely dependent on the perinatal period, despite its major impact, but represents complicated multifactorial processes. Other interacting factors such as environmental, social, and demographic characteristics of the families would greatly influence the eventual developmental outcome of preterm infants [45]. Maternal parity, education, age, Ethnity, medical insurance, and marital status are among the sociodemographic variables that correlate with cognitive outcomes of preterm infants. It seems that parental sociodemographic factors are not only important determinants of the occurrence of PVH-IVH, but also play a significant role in the long-term neurodevelopmental outcome among IVH survivors. Sociodemographic factors should be considered as a 'continuum' with an extended influence starting at the prenatal period of critical neuronal proliferation, migration and synaptogenesis, and then progressing into the postnatal period, adolescence, and early adulthood.
The issue of neurologic plasticity versus vulnerability and the possible recovery of neurologic insults even in later age should be considered when it comes to the long-term neurodevelopmental dynamic outcomes. Plasticity is the flexible and dynamic capacity of the central nervous system in early life to adapt in response to environmental stimuli through establishment of new neural connections [46,47]. This maybe a useful property in view of minimal functional specificity of the immature brain and the possible transfer of a function form a damaged area to another unaffected area [48]. This school is opposed by the immature brain vulnerability in face of different environmental, constitutional, and brain injury related factors. Hence, assessing the developmental outcome at a specific age and then generalising the outcome of certain grade of IVH is not fair. This observation is obvious in this review and may explain the conflicting developmental outcomes among survivors of mild PVH-IVH. Furthermore, the general intelligence notion is disputed by the theory of fluid and crystallised intelligence (Gf-Gc theory), where the fluid part Gf represents the influence of biological factors such as IVH on intellectual outcomes where the crystallised part Gc represents the influence of education and other demographic factors [49]. The peak capacity of fluid and crystallised knowledge is reached by early adulthood, hence highlighting the importance of extended follow up of cognitive developmental assessment beyond the previously thought the critical ages of development [50].
No doubt that even mild PVH-IVH can potentially interfere with the immature delicate physiological architecture of the germinal matrix and cerebral white matter. White matter injury (WMI) may result from hypoxic events that precede or concur with IVH or from the release of iron, a significant free radical, that impair the normal proliferation and migration of oligodendrocyte precursors. Uncomplicated cases of mild PVH-IVH are associated with significant impairment of cortical grey matter development when examined by brain MRI at term equivalent age (TEA) [51]. WMI maybe more important than IVH regarding the later neurodevelopmental outcome and maybe easily overlooked if only cUS is used without additional brain MRI for the evaluation of preterm infants with IVH. Tortora, et al. [19] used Advanced computational methods; the diffusion tensor imaging (DTI) modality to assess the early WM microstructures changes occurring in isolated mild PVH-IVH. They found distinct patterns of WMI that can be missed on conventional brain MRI and correlates with poor neurocognitive outcomes at 2-3 years corrected age. Accordingly, it is feasible from the pathological point of view- at least- to presume a spectrum of negative neurodevelopmental outcomes secondary to even subtle degrees of PVH-IVH.
Infants with moderate to severe IVH have a higher prevalence of neurodevelopmental handicaps than infants with low grade or no IVH. However, the neurodevelopmental outcomes among infants with lower grade PVH-IVH remain uncertain. The inconsistencies in neurodevelopmental outcomes among preterm infants with low grade IVH gives a clue to a wider spectrum of microstructural cerebral injuries that may be overlooked by cUS. Concomitant diffuse white matter injury of the brain is a major determinant of the neurodevelopmental outcome and can be detected by the newer brain MRI technology which is highly predictive for future neurocognitive disabilities. PVH-IVH may represent just the tip of iceberg of underlying cortical and subcortical microstructural pathologies that delineate the future neurocognitive outcomes. Further efforts to adopt advanced neuroimaging techniques and new classification system of brain injuries accompanying PVH-IVH based on the actual neuronal damage and not merely on the size or extent of the bleed are paramount, thus enhancing the prognostication process and family counselling. It is crucial to establish robust databases relating the new neuroimaging classification and the unified neurodevelopmental assessment scores to the long-term functional outcomes as well as mortality prediction.
No specific support.
Dr. Alshafei has disclosed no financial relationships relevant to this article. This commentary does not contain a discussion of an unapproved/investigative use of a commercial product/device.