Background: The study report the results of a quality improvement (QI) program implemented to improve viral suppression (VS) in a cohort of children living with HIV who had viremia (viral load [VL] ≥ 1,000 copies/mL) despite being on effective antiretroviral therapy (ART).
Methods: The Meeting Targets and Maintaining Epidemic Control (EPIC) project's quality assurance/quality improvement team monitors quality across HIV/AIDS thematic areas. Epic collaborated with the state pediatric task force supporting pediatric ART management to address VS. QI activities involved assessment of each child's needs by the pediatric task team, implementation of an individualized package of care together with enhanced adherence counseling (EAC), situation-room meetings to monitor overall implementation, and follow-up VL testing to determine suppression. We compared routine patient data across 49 health facilities from prior to the high VL event (July 2020-December 2020) to data collected after 6 months of QI activity implementation in July 2021. Segmented regression using the generalized estimating equation logistic model was conducted to determine the effect of the QI intervention on various service delivery components. We used logistic regression to determine factors associated with persistent viremia.
Results: Overall, 312 children were evaluated. The median age was 7 years (IQR: 4-10), 50.6% were males, median duration on ART was 19 months. After 6 months of QI implementation, 98.1%, 94.9%, and 92.3% had completed 1, 2, and 3 EAC sessions, respectively. We observed significant improvements from baseline in uptake of prophylaxis for TB opportunistic infection (p < 0.001), enrollment in age-appropriate adherence model groups (p < 0.001), HIV status disclosure (p = 0.002), multimonth drug dispensing (p < 0.001), and transition to the optimal regimen (p < 0.001). Post-EAC VL testing uptake was 84.6% and viral suppression was 77.2%. Males and children on ART for longer than 12 months were more likely to have persistently high VL (p = 0.009 and p = 0.044, respectively).
Conclusions: Our QI intervention was associated with improved viral suppression in this cohort of children. Additional measures are needed to address the needs of males and children who have been on ART for longer periods.
High viral load, Children, HIV/AIDS, Quality improvement, Patient-centered, Pediatric task team
AIDS: Acquired Immuno-Deficiency Syndrome; ART: Antiretroviral Therapy; ARV: Anti-Retrovirals; CLHIV: Children Living with HIV/AIDS; COVID-19: Coronavirus Disease 2019; DTG: Dolutegravir; EAC: Enhanced Adherence counseling; EFV: Efavirenz; HIV: Human Immunodeficiency Virus; IEC: Information Education Communication Material; LAMIS: Lafiya Management Information System; LGAS: Local Government Area; LPV/r: Lopinavir/ritonavir; MMD: Multi-Month Dispensing; OTZ: Operation Triple Zero; PEPFAR: President's Emergency Plan for AIDS Relief; QA/QI: Quality Improvement/Quality Assurance; SIDHAS: Strengthening Integrated Delivery of HIV/AIDS Services; TB: Tuberculosis; VL: Viral Load; WHO: World Health Organization
Ending HIV and AIDS by 2030 requires countries to diagnose 95% of all HIV-positive individuals including children 0-14 years, provide antiretroviral therapy (ART) for 95% of those diagnosed, and achieve viral suppression for 95% of those treated. However, on a global scale in 2019, only 53% of children living with HIV (CLHIV) were receiving ART compared to 68% of adults. Similarly, the proportion of children 0-14 years with suppressed viral load (VL) was significantly lower (40%) than that of adults (67%). These differences reflect age-specific challenges across the treatment cascade [1]. Achieving viral suppression is necessary to decrease morbidity and mortality associated with HIV treatment failure among children and adults [2-4].
Several barriers exist to the achievement of viral suppression among children. These include poor adherence due to the side effects and nonpalatability of some available formulations [5,6], dependence on caregivers for ART pick-up at a health facility or to administer medication [7], confidentiality around the HIV status of the child, food requirements for some regimens [8], nondisclosure, forgetfulness [9], and transport to health facility [10]. While adherence interventions have been found to improve VL suppression in up to 70% of children failing first-line regimens [11-13], only a limited number of studies have been conducted in Nigeria - a country with one of the highest numbers of children living with HIV-to assess the impact of targeted interventions among this population. Children living with HIV require a continuum of care and treatment regimens proven to improve health outcomes.
The USAID-funded Strengthening Integrated Delivery of HIV/AIDS Services (SIDHAS) project (2011-2021) provided support for comprehensive HIV services in Akwa Ibom State, which has the highest HIV burden of the 36 states in Nigeria. By the end of December 2020, 2,504 children 0-14 years were receiving ART in the 21 local government areas (LGAs) supported by SIDHAS in Akwa Ibom State (68% of all LGAs in the state). The comprehensive ART services included support for children to adhere to medication to achieve and sustain viral suppression. Viral suppression among children enrolled in the program was 84% in December 2020 compared to 93% among adults. This led to the development and implementation of a quality improvement (QI) program using the assess-implement-evaluate approach to improve and maintain viral suppression among CLHIV in Akwa Ibom State and meet the Joint United Nations Programme on HIV/AIDS (UNAIDS) standards by July 2021.
The analysis presented in this paper compared data from before - after the QI program and determined the impact of the program on achievement of VL suppression among children living with HIV who had failed the first-line ART regimen in Akwa Ibom State, Nigeria.
The study was conducted in the 21 SIDHAS-supported LGAs in Akwa Ibom State in southern Nigeria.
Routine program data was collected and analyzed for cohort of virally unsuppressed (VL ≥ 1,000 copies/mL) children (0-14 years) living with HIV who were on a first-line antiretroviral (ARV) regimen as of December 2020 in 49 SIDHAS-supported comprehensive HIV treatment centers (29 primary, 19 secondary, 1 tertiary) in Akwa Ibom that participated in the QI activity. Data were reviewed at baseline (January 2021) and 6 months post-intervention (July 2021).
Intervention description: The SIDHAS project has a quality assurance (QA)/QI team that monitors program quality across all thematic areas. The project's state pediatric task force team comprised of dedicated clinicians, adherence case managers, and clinical mentors from a tertiary health facility provide support for pediatric ART management. The 2 teams conducted a joint assessment of pediatric VL suppression from July through December 2020 in 49 health facilities. They used the assess-implement-evaluate approach to develop an individualized care plan for each child. The plans were implemented from January through July 2021 across the different facility types.
The assess-implement-evaluate approach focuses on systematic collection and assessment of data to provide useful feedback for informed decision-making. In the assessment phase, health needs are considered and a package of care is developed to address the needs (Table 1). Potential barriers and challenges are also identified and a detailed plan is developed to address each one. In the implementation phase, the plan is activated and may be adjusted based on changing needs. In the evaluation phase, the outcomes of the intervention for each child are discussed for shared learning and feedback to inform decisions for childcare and treatment support.
Table 1: The assess-implement-evaluate phases of the VL intervention among CLHIV in SIDHAS-supported LGAs in Akwa Ibom State, Nigeria. View Table 1
During the assessment phase, a baseline assessment was conducted in all 49 participating health facilities using a quality assessment checklist developed from standards prescribed in the national guidelines [14]. The checklist assessed quality of care using 4 parameters: Optimized ART (correct regimen and dose), care and support services (opportunistic infections prophylaxis, family/social support for disclosure and adherence, and multimonth dispensing [MMD] of ARVs), and enhanced adherence counseling (EAC) services, and VL monitoring.
The joint QA/QI and state pediatric task teams conducted a chart review of all 312 CLHIV with VL ≥ 1,000 copies/mL to identify gaps in the HIV continuum of care services for each child. This was followed by a brainstorming session involving caregivers and treatment supporters to gain a contextual understanding of each child's characteristics and develop a plan tailored to the child's needs. The clinical charts of the children identified were marked as priority cases with a sticker and tagged to receive needed services. Facility-level assessments were also performed to identify system-level challenges to address.
During the implementation phase, a package of care was introduced that included:
• Correcting inappropriate regimen/dose for weight and age based on the National Guideline on HIV Care and Treatment [14].
• Screening for opportunistic infections and initiation of applicable prophylaxis, with cotrimoxazole preventive therapy administered to all CLHIV to prevent opportunistic infections.
• Disclosure: Caregivers and health care workers were supported to provide age-appropriate disclosure to children and adolescents per the national guidelines [14]. "Partial disclosure" (i.e., providing information about the child's illness which may include information on immunity and the need to take medicines to keep illness at bay but no mention of HIV or AIDS) is recommended for children ages 5-8 years, while "full disclosure" (whereby children are given detailed information about HIV) is recommended for children and adolescents ages 9-12 years, with the goal of completing the process by ages 10-12 years.
• Enrollment into support groups and other differentiated service delivery models: CLHIV (> 10 years) were linked to the adolescent support program, Operation Triple Zero (OTZ), following caregivers' disclosure of the child's HIV status to each one. OTZ is an asset-based program that empowers adolescents and youth living with HIV to participate in the care of their own health [15]. The OTZ caregiver package included the provision of basic child-care training skills through psychosocial group discussions to support caregivers of CLHIV. CLHIV ages 0-17 years were also referred and enrolled into an orphans and vulnerable children program that uses community-based organizations to strengthen adherence at the household level and supports children and adolescents to overcome socioeconomic determinants of health.
• EAC was implemented and monitored as per the national guidelines, which recommend that individuals with VL at or above 1,000 copies/mL undergo 3 EAC sessions [14]. The EAC sessions were delivered monthly. During each session, barriers to optimal adherence to ART were identified and a mitigation plan was developed and tailored to the child's unique needs. Adherence was assessed retrospectively based on a 4-day recall. Patients who reported missing a dose were probed for the reasons, counseled on adherence, and guided to develop plans to mitigate the reasons for missing doses. If a patient was nonadherent after 3 EAC sessions, EAC was extended or repeated. A repeat VL test was conducted after 3 EAC sessions. If the repeat VL was suppressed (≤ 1,000 copies/mL of blood), the patient continued with the same ART regimen and the next VL test was scheduled for 12 months later. If, however, the repeat VL was ≥ 1,000 copies/mL despite optimal adherence, the patient was switched to the second-line ART regimen as recommended in the national guidelines (Table 2).
Table 2: Baseline characteristics of virally unsuppressed CLHIV on ART, Akwa Ibom State, Nigeria, December 2020 (N = 312). View Table 2
• Clinic appointment dates were streamlined to ensure that children and their caregivers had the same appointment dates at the clinic to limit transport and scheduling barriers.
• Adherence case managers conducted daily follow-up with caregivers of targeted CLHIV through calls, SMS reminders, or weekly home visits to track medication adherence.
Facility-level plans included training health care workers on the management of CLHIV on ART, printing and distributing IEC materials and job aids, patient listing, and identifying children identified as having service delivery gaps for prompt action and tracking. A high VL register was introduced in these sites to document implementation of key activities during EAC and was maintained until viral suppression was achieved. The pediatric task force and QA/QI team also met virtually on a weekly basis to monitor implementation progress.
In the evaluation phase, quality-of-care parameters were measured. These included being on optimal ART (prescription of the right regimen and dose), access to care and support services (uptake of prophylaxis for opportunistic infections, access to family/social support for disclosure and adherence, and MMD), EAC service provision, and VL monitoring.
As with other health services, data from routine HIV services are documented in patient folders after each clinic encounter and entered in an electronic medical record, the Lafiya Management Information System (LAMIS). The data of children ages 0-14 years with unsuppressed VL (≥ 1,000 copies/mL) from July through December 2020 were extracted from LAMIS at baseline (December 2020) and compared to data at 6 months after implementation (July 2021).
The data elements extracted were weight, sex, HIV disclosure by caregiver (for children ages 8 years and older), prophylaxis for opportunistic infections, date of HIV diagnosis, date of ART initiation, adherence support provided (self-managed or treatment support group), and ART regimen. In addition, whether the child had had EAC, the dates and outcomes of each EAC session, and the date and result of post-EAC VL testing were also collected.
Data cleaning was done using MS Excel 2016® and analyzed using SPSS version 26. Sociodemographic data were presented as frequencies for categorical variables and as medians for continuous variables.
Segmented regression was conducted using a generalized estimating equation logistic model to analyze the before- and aftereffects of the intervention on various service delivery components (HIV status disclosure, prophylaxis coverage for opportunistic infections, regimen optimization, enrollment into age-appropriate adherence model, MMD of ARVs) at the end of the sixth month.
The uptake of services in the EAC intervention cascade was also analyzed and summarized as proportions by CLHIV age groups. This included EAC enrollment after notification of high VL (timepoint of January 2021), EAC uptake of at least 3 sessions (timepoint during January 2021-July 2021), post-EAC VL testing uptake (timepoint during April 2021-July 2021 after completion of EAC), and post-EAC VL testing outcome (timepoint during April 2021-July 2021).
We summarized post-EAC VL testing outcome as the proportion of the cohort with undetectable VL (≤ 50 copies/mL), suppressed VL (50 to ≤ 1,000 copies/mL), or unsuppressed VL (≥ 1000 copies/mL).
Finally, we fitted the variables into a logistic regression model to analyze factors associated with high post-EAC VL (i.e., persistent high VL of ≥ 1,000 copies/mL) and expressed output using coefficients and 95% confidence intervals. The p-value for level of statistical significance was set at < 0.05.
Ethical approval for retrospective collection of routine program data and dissemination of the study results was obtained from the Office of International Research Ethics (reference no. 1790435-1), which gave the study a non-research determination. Informed consent from patient was not required, as only routine, de-identified, operational monitoring data were collected and analyzed.
Of the 2,504 CLHIV on ART who were eligible for a VL test as of December 2020, 1,979 (79%) were tested and received results. Of those who received VL results, 315 (15.9%) had a VL of ≥ 1,000 copies/mL, 2 were reported as deceased, and 1 had interrupted treatment, and all were excluded from further analysis (Figure 1). Overall, 312 children were enrolled into the program.
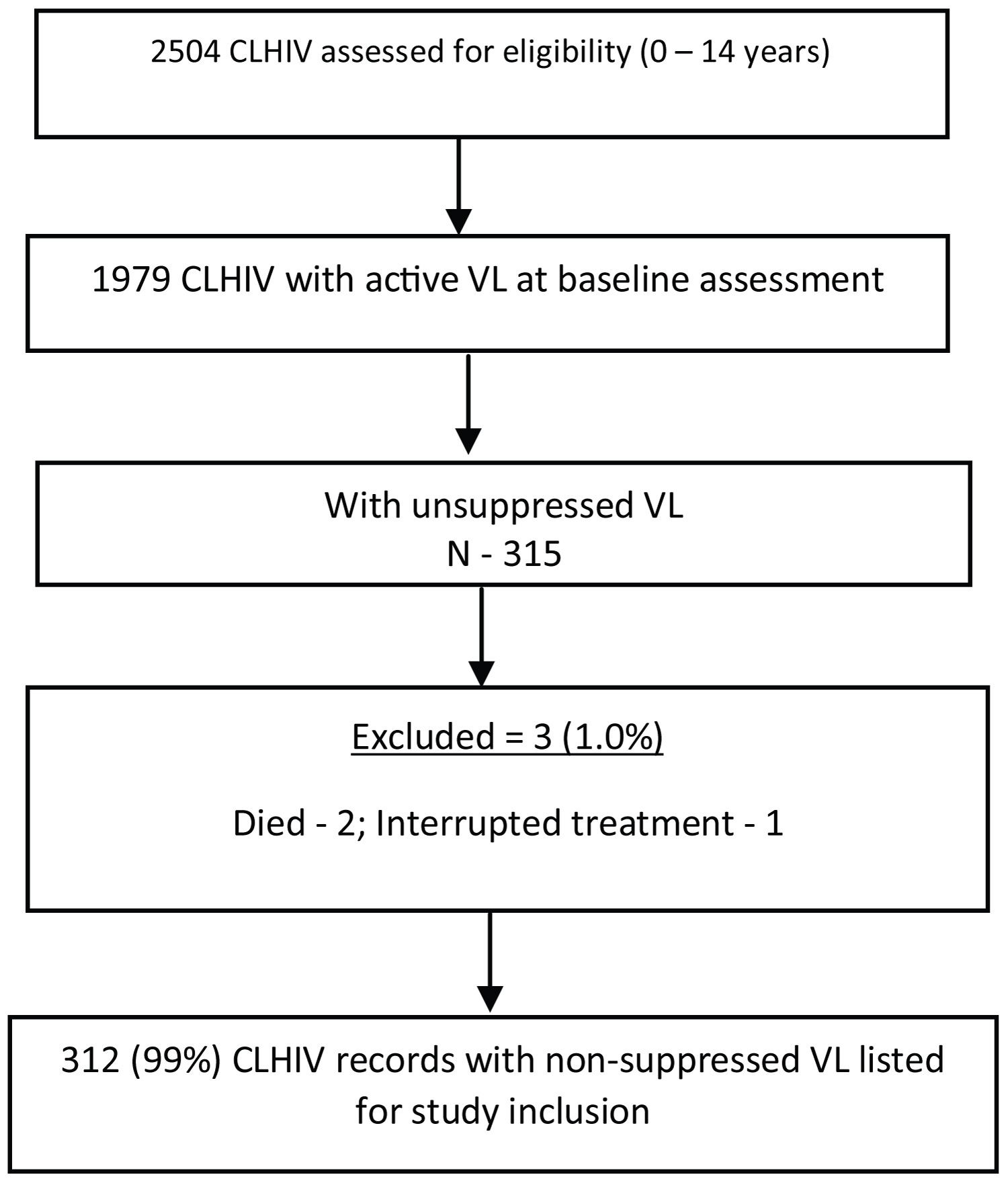 Figure 1: Baseline audit results.
View Figure 1
Figure 1: Baseline audit results.
View Figure 1
The median age was 7 years (IQR: 4-10), 50.6% (158/312) were male, and 60.3% were receiving care in secondary and tertiary health care facilities. The majority (62.2%; 194/312) had been on ART for less than 12 months, and 18.3% (57/312) for longer than 3 years (Table 2).
The disclosure rate among older children (11-14 years) increased from 20% to 46.7%, while coverage for tuberculosis (TB) preventive therapy across all age groups increased from 44.6% (139/312) to 100% (312/312) by the end of the 6-month intervention period. NVP-based and EFV-based regimens were eliminated and fully replaced with LPV/r-based and DTG-based regimens post-intervention. By the end of the intervention, all children were enrolled in a treatment support group. There was a positive trend in enrollment in MMD of ARVs, with the highest MMD rates among children 7 years and older (183, 71.5%) (Table 3).
Table 3: Number and proportion of children receiving various HIV service components before and 6 months after the QI intervention in Akwa Ibom State, Nigeria, December 2020-July 2021 (N = 312). View Table 3
In a segmented regression analysis to determine the effect of the intervention on selected intermediate outcome variables, we found significant improvement post-implementation for disclosure, opportunistic infection prophylaxis uptake, regimen optimization, enrollment into an age-appropriate adherence model, and ART dispensing schedule (Table 4).
Table 4: Segmented regression analysis showing the effect of the QI intervention on service delivery components, Akwa Ibom State, Nigeria, December 2020-July 2021. View Table 4
EAC uptake and retention are summarized in Table 5. Of the cohort of 312 children with unsuppressed VL, 98.1% (306/312) had commenced EAC at the end of the review period, 94.9% (296/312) had completed 2 EAC, while 92.3% (288/312) had completed all 3 EAC sessions, and 84.6% (264/312) had had a repeat VL test during the study period.
Table 5: Uptake and completion of EAC and post-EAC VL testing among the cohort of children with unsuppressed VL by age group, Akwa Ibom State, Nigeria, December 2020-July 2021. View Table 5
Of the 312 children in our cohort, 241 (77.2%) achieved a VL less than 1000 copies/mL and were considered suppressed according to national and World Health Organization (WHO) standards. Among those who were suppressed, 58.7% (155/264) had a VL of less than 50 copies/mL (the lowest limit detectable by the VL assays used in Nigeria) and were considered undetectable, while 32.6% (86/264) had a VL of 50-999 copies/mL. Viral suppression was higher among children 0-3 years, females on ART for a longer duration, children receiving care in a primary health facility, and those on MMD (Table 6).
Table 6: Post-EAC VL outcomes for the children who had a VL test after EAC by CLHIV characteristics, Akwa Ibom State, Nigeria, December 2020-July 2021 (N = 264). View Table 6
In the bivariate analysis to determine the effects of age, gender, facility type, ART refill schedule, and ART duration on the achievement of viral suppression post-EAC, only gender (p = 0.01) and duration on ART (p = 0.003) were statistically significant in predicting viral resuppression.
In a multivariate analysis, males (p = 0.009), as well as children on ART longer than 12 months (p = 0.044) (Table 7) were more likely to have persistent high VL.
Table 7: Logistic regression to show factors associated with persistent high VL for children who had a VL test after EAC, Akwa Ibom State, Nigeria, December 2020-July 2021. View Table 7
This paper analyzed data before and after a QI program and determined the impact of these interventions on achievement of VL suppression among CLHIV who had failed ART in Akwa Ibom State, Nigeria. Our intervention, which included assessment of child needs by a pediatric task team, implementation of an individualized package of care including EAC, and weekly situation room meetings to track and evaluate performance, was associated with achievement of viral suppression among 77.2% of CLHIV considered to have failed first-line ART. During the intervention period, significant improvements were observed in the uptake of prophylaxis for opportunistic infections, access to family/social support for disclosure and adherence, MMD, and the elimination of ineffective regimens.
We attribute these achievements to the implementation of tailored packages-developed following systematic assessments and customized to each child's needs and to each facility. At every phase, the project involved clinical, support staff, and caregivers of CLHIV in decision-making with a goal of improving each child's treatment outcomes. Interventions were designed to be centered on the individual and tailored to patient needs. Studies to measure the impact of multi-level stakeholder involvement on outcomes for PLHIV have been limited [16]. Our findings suggest that a multidisciplinary team approach can lead to better decision-making and improved health outcomes. In our program, 98.1% of virally unsuppressed children commenced the EAC sessions, 92.3% of whom completed EAC by the end of the study's review period. These rates are higher than published studies in the region. For instance, in a similar study in Uganda, only 77% of the enrolled cohort completed all 3 EAC sessions [17].
Several studies have cited the role of adherence case managers in optimal service delivery, increased care engagement, and treatment adherence, as well as in achieving better outcomes among PLHIV. In our study, adherence case managers ensured continuous adherence support and sent appointment reminders to caregivers and children through phone calls and SMS reminders to caregivers or weekly home visits to track medication adherence. They also reviewed routine patient data to ensure that EAC and associated packages were being documented. This active monitoring of the delivery of all planned services likely contributed to the outcomes we observed.
We note that VL testing coverage among children in this cohort was low. Post-EAC VL testing uptake was only 84.6% despite intense monitoring within this group. Our QI intervention was implemented at the peak of the COVID-19 pandemic in Nigeria and highlights the challenges pandemic related movement restrictions and lockdowns and enforced social distancing on access to facility based services including VL testing services.
We also note that EAC completion was quite high, although in a few cases completion of EAC extended beyond the three months recommended by WHO. This delay was to ensure adherence prior to repeating VL measurement. Viral suppression among those who completed EAC was 77.2%, highlighting the role of counseling in the achievement of optimal outcomes. This finding implies that nonadherence to ART could have been the most common reason for treatment failure in CLHIV in the study setting. It also partly explains why suppression rates in our study were higher than among cohorts with lower EAC completion rates, as evaluated in Uganda (23%) [17], Zimbabwe (31.2%) [18], and Ethiopia (66%) [19]. The current findings also support the WHO recommendation to address suspected virologic failure (VL of ≥ 1,000 copies/µl) through EAC and repeat measurement of VL before considering switching to a second-line drug [4,14], including for children. EAC interventions can preserve use of the first-line treatment regimen even among CLHIV. Our intervention likely prevented premature switching of regimens in the majority of CLHIV.
Our analysis had some limitations. First, because our study methodology was based on review of patient records, our data analysis and interpretation were limited to the variables routinely collected from patients and documented in the patient records. Thus, our study was unable to account for the influence of other factors, for example adherence, that could have played a major role in viral resuppression. Second, all interventions were implemented simultaneously, and it was not possible to breakdown impact of each aspect of the intervention. These limitations could be addressed by a prospective study. Nonetheless, our findings are important, as they measured the impact of interventions implemented across multiple ART health facilities.
We achieved a high viral suppression rate among CLHIV with the package of interventions we implemented in Akwa Ibom. However, access to VL testing was low among children who completed EAC. Also, we found the achievement of viral suppression to be more challenging among males and those with a longer duration on ART. These subgroups may need additional measures if ending AIDS is to become a reality.
All authors declare no competing interests.
UA, KU, IE, and EN conceptualized the study. UA, AI, OT, and TB conducted the data analysis with the guidance of OS, PN, HK, CW, and MB. All authors contributed to data interpretation, writing of the manuscript, and approval of the final version.
The authors acknowledge all those who were involved in the SIDHAS project in Nigeria, particularly the technical and strategic information staff members based at the various facilities.
This work resulted in part from data collected during the implementation of the PEPFAR-funded SIDHAS project in Nigeria (Cooperative Agreement Number: AID-620-A-11-00002).
• Involving all clinical service providers, treatment supporters, and caregivers of CLHIV is key to improving treatment outcomes among children.
• Delivery of targeted interventions should be person-centered.