Introduction: Sickle cell disease is a genetic disease that is common in Madagascar with serious complications. The cerebral vasculopathies are among the most dreadful complications by its morbid mortalities. In Madagascar, these cerebral vasculopathies in children with sickle cell disease are not sufficiently studied, and the risk factors are still unknown, hence the goal of our study.
Methods: We conducted a case-control multi-center study over a 24-month period (from March 2016 to March 2018), on children with sickle cell of 2 months to 15-years-old.
Results: Sixty-four children were included with 32 cases and 32 matched controls by age. Twenty-nine cases had clinical cerebral vasculopathy with stroke, and 3 had accelerated cerebral arterial velocities with TCD ≥ 200 cm/s. The risk factors found were leukocytosis p = 0.014 IC 95% OR 3.63(1.27-10.37), thrombocytosis p = 1.9e-05 IC 95% OR 10.35 (2.5-42.3) and a regular influenza vaccination p = 0.02 IC 95% OR 0.16 (0.04-0.7).
Conclusion: The cerebral vasculopathies of sickle cell children are complications that can be easily prevented if the risk factors are identified.
Cerebral vasculopathy, Child, Risk factor, Sickle cell disease, Transcranial doppler
Sickle cell disease is a genetic disease with autosomal recessive transmission, in which normal hemoglobin A (α2β2) is replaced by hemoglobin S (α2β2s).
In Madagascar, according to the terms of the National Policy document developed in 2011 on the Management of Sickle Cell Disease in Madagascar (PNLD), the overall prevalence of sickle cell disease varies according to the regions with a high endemicity in the South-East region or Atsimo Atsinanana where it is 18.5% [1,2].
Neurological complications such as cerebral vasculopathies are formidable in this pathology. Clinical manifestations may be neurological focusing (convulsion, hemiparesis/hemiplegia, aphasia) disorder of consciousness up to coma, or behavioral disorder, secondary to a stroke; but may also be subclinical on an acceleration of blood flow (speed ≥ 200 cm/s) to the transcranial doppler (TCD). Heavy sequelae responsible for high mortality are possible [3]. These complications mainly affect sickle cell patients aged 2 to 10-years-old and are ischemic in 75% of cases. The risk factors identified in the literature are low baseline hemoglobin, a history of acute chest syndrome, hyperleukocytosis and thrombocytosis [4].
Transcranial Doppler (TCD) plays an important role in the prevention and prediction of these neurological complications. A speed of ≥ 200 cm/s at the TCD predicts a risk of stroke of 40% at 3 years.
In Madagascar, TCD screening is not a common practice in the monitoring of sickle cell children because of its cost, and few studies have been carried out on cerebral vasculopathy in sickle cell children.
The objective of this study was to identify risk factors for cerebral vasculopathy in sickle cell children in Madagascar. This is in order to improve the functional but also vital prognosis of these patients.
A retrospective, case-control, multicenter analytical study was carried out.
The place of study was the Center for the Fight against Sickle Cell Disease of Madagascar Antananarivo (LCDM), the surgical resuscitation department of the Joseph Raseta Andrianavalona Antananarivo University Hospital (CHUJRA), the pediatric department of the Analakininina Toamasina University Hospital, over a period from March 2016 to March 31, 2018.
The children included were sickle cell children aged from 2 months to 15-years-old, i.e., of homozygous SS, heterozygous SC or thalassemic S phenotype β. A case defined by a major sickle cell child, who has had a clinical vascular accident with hemiplegia or hemiparesis or dysarthria objectified during hospitalization, or who has an acceleration of cerebral arterial velocity ≥ 200 cm/s in the Transcranial Doppler (TCD) with a known baseline hemoglobin level. A control defined by a major sickle cell child without clinical stroke, and with a TCD of ≤ 169 cm/s during the study period.
Cases of seizures and cognitive impairment not documented by imaging are not included.
Those who died during the study period or without regular follow-up with TCD and without baseline blood count are excluded.
A total of 32 cases were identified matched to a control by age.
The variables studied were demographic characteristics (age, gender, ethnicity), clinical aspect of sickle cell disease (phenotype, age of discovery, mode of discovery, presence of stroke, presence of history of acute chest syndrome, presence of other documented associated vasculopathies), paraclinic aspect (baseline blood count excluding acute attacks, TCD), and current treatment (daily folic acid, hydroxyurea, monthly intravenous hyperhydration, followed by a transfusion program, anti-infective prophylaxis by antibiotic prophylaxis and vaccination).
The data was processed by epi-info 7 software. The quantitative variables were presented on average, the qualitative variables in numbers and percentages. The multivariate regression test determined the factors associated with mortality. Differences were considered significant for p < 0.05 and OR > 1.
This study had a selection bias: the "case" patients were all hospitalized children with brain imaging and the "controls" were those who had regular TCD.
Four hundred and seventy-seven sickle cell children were identified in the 3 sites during the study period, including 93 children at the Analakinina University Hospital, 5 in surgical resuscitation and 379 at the LCDM.
Ninety-six children met the inclusion criteria.
Twelve passed away during the study period, 7 had unexplored seizures, and 13 did not have annual systematic follow-up at the TCD.
In total, the final study population consists of 64 children, or 32 cases retained for 32 controls.
Of the cases, 5 were hospitalized in the surgical resuscitation ward of the HJRA, 15 hospitalized in pediatrics of the Analakininina Toamasina University Hospital and 12 identified within the LCDM center. Sickle cell children, whose stroke diagnosis was made in clinical haematology of HJRA during their consultations, were treated in surgical resuscitation to initiate early management (Figure 1).
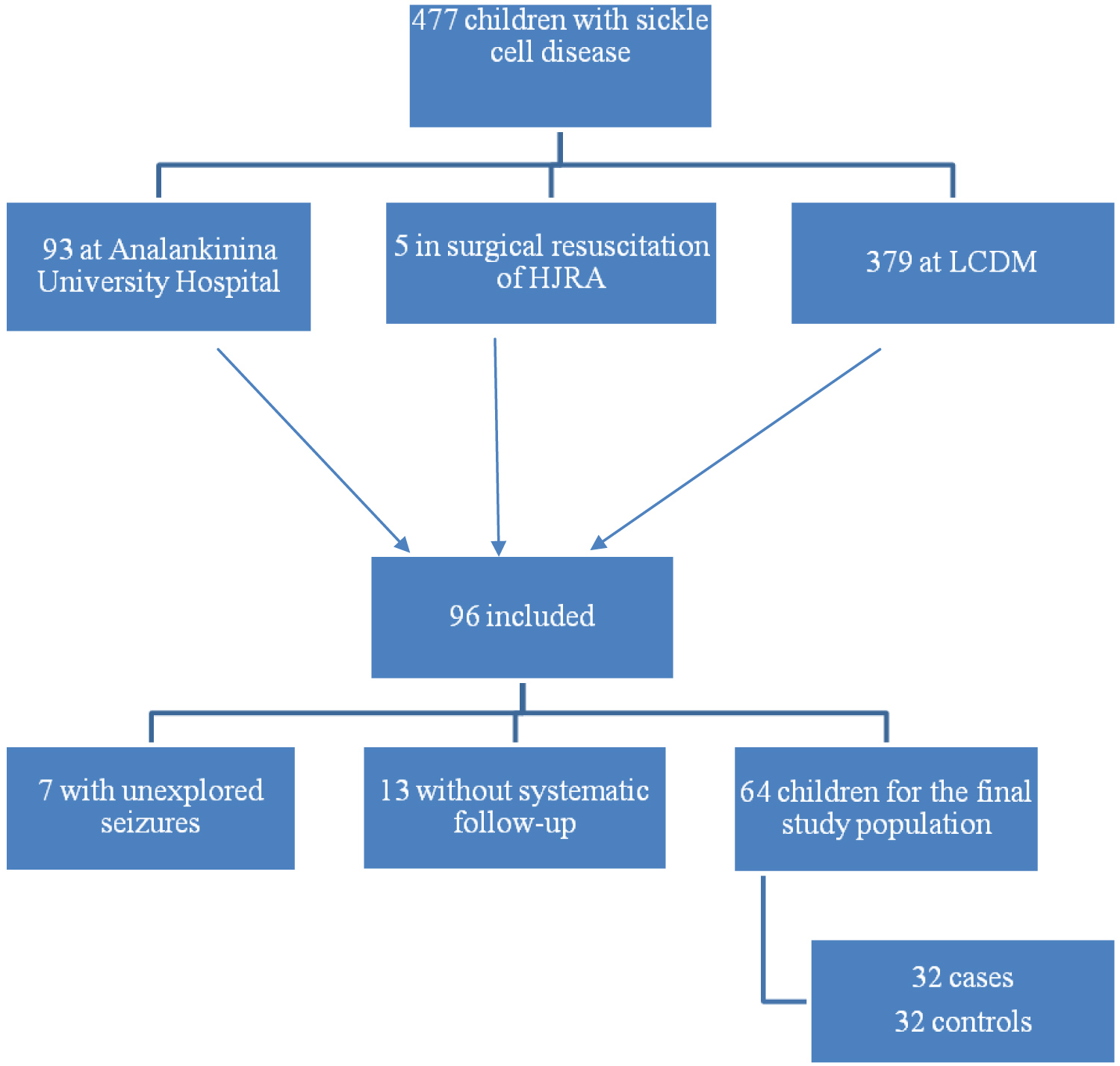 Figure 1: Flowchart.
View Figure 1
Figure 1: Flowchart.
View Figure 1
The witnesses were counted at the LCDM center.
The average age of the study population was 8.4-years-old ± 2.5 with extremes of 2 and 14-years-old. A female predominance was found with a sex ratio of 0.81.
The disease was diagnosed before the age of 36-months-old in 38 children in the study population, and 89% of cases (n = 57) were of hospital discovery. The discovery circumstances were vaso-occlusive seizures for 60.9% of the children (n = 39) and a stroke chart was the telltale mode in a child.
Six of the 64 children had a history of acute chest syndrome. Five of the 64 children had non-cerebral vasculopathy; this was cardiomyopathy discovered during hospitalization and confirmed by a cardiac echodoppler.
Fifteen out of 64 children had a low baseline haemoglobin level < 7 g/dl. Hyperleukocytosis for age was found in 38 children and thrombocytosis was found in 29 children.
The annual systematic implementation of the TCD was monitored by 55 children.
Folic acid was taken by 95.31% of children while the transfusion program was followed by only 28.1% of children. Hydroxyurea has only been received by 18.75% of sickle cell patients, those who have more than 6 painful attacks per year. Fifty-one percent of the children received pneumococcal antibiotic prophylaxis with ORACILLINE. Vaccinations against influenza viruses, pneumococci, and salmonella were followed in 65.62%, 59.37%, and 34.37% of the study population, respectively.
On the other hand, parents are not up to date with vaccine boosters, for lack of means.
In children with cerebral vasculopathy, the age group 4 to 10 years was most represented with extremes of 3 and 11 years. According to ethnic origin 11 out of the 18 ethnic groups of Madagascar were represented with 21 children of Merina ethnicity.
Hemiplegia was the most represented sign of vasculopathy (20 out of 32 children), followed by hemiparesis (8 out of 32 children) and dysarthria (1 out of 32 children). Children with these motor disorders received brain imaging by CT scan.
Three children presented with cerebral vasculopathy at TCD without clinical signs.
Of the cases, 9 children have never done a TCD in their lives. The most common pathological artery found at TCD was the right anterior cerebral artery (RACA) as shown in Figure 2.
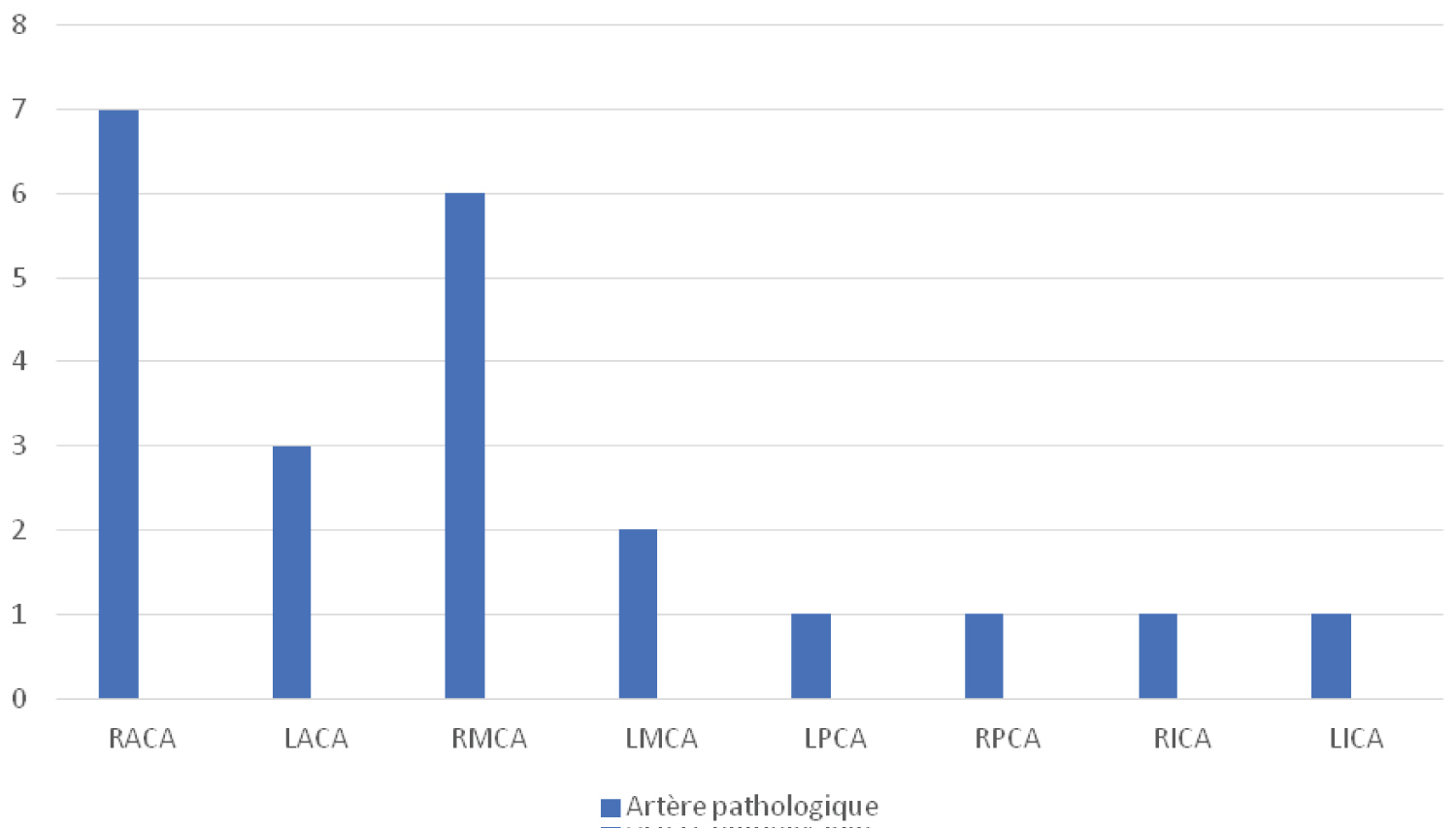 Figure 2: Distribution of sickle cells by pathological artery.
Figure 2: Distribution of sickle cells by pathological artery.
RACA: Right Anterior Cerebral Artery; LACA: Left Anterior Cerebral Artery; RMCA: Right Middle Cerebral Artery; LMCA: Left Middle Cerebral Artery; LPCA: Left Posterior Cerebral Artery; RPCA: Right Posterior Cerebral Artery; RICA: Right Internal Carotid Artery; LICA: Left Internal Carotid Artery
View Figure 2
The risk factors identified in this study were hyperleukocytosis p = 0.014 OR = 3.63 95% CI [1.27-10.37], thrombocytosis p = 1.9e-05 OR = 10.35 [2.5-42.5] and influenza vaccination p = 0.02 (Table 1).
Table 1: Risk factors for cerebral vasculopathy. View Table 1
During this study, stroke occurred in more girls than boys 20/12 or 62% with a sex ratio of 0.6.
This differs from the results found by Arouna Ouattara in Mali and Moustapha Ndiaye in Senegal who find a male predominance [5,6].
According to Ohene-Frempong K, there was no difference in the two types. This female predominance in this work could be explained by the number of births by gender. In Madagascar, twice as many girls are born as boys.
Children aged 4 to 10-years-old are the most affected in this study: 43.75% (n = 28) with an average age of 8.4 years.
This age range is similar to what Arouna Ouattara observed: 4 to 10 years [5] and to what Ohene-Frempong K reported 5 to 10 years [7].
A study in Nigeria found an average incidence of stroke at 6 years [8] and in Senegal this average incidence was 7 years [9].
According to a study by Verlhac, the age of onset of the first stroke was between 1.7 and 16.5 years [10]. Absence of clinical or imaging stroke before 10 years, reduces the risk of occurrence of vascular event in adolescence [10,11].
This predominance before adolescence is explained in the literature by the increase in blood circulatory speeds in the cerebral arteries between 3 and 12 years with a maximum peak between 7 and 8 years. Circulatory acceleration is independent of any stenosis due to chronic anemia through tissue hypoxia which is responsible for decreasing peripheral resistance, increasing cardiac output and cerebral blood flow [12].
Sickle cell children are 300 times more likely to develop cerebral vasculopathy than other non-sickle cell children of the same age. The peculiarity of strokes in sickle cell children is that they can occur without a prodrome or as a result of a vaso-occlusive attack or too large a blood transfusion increasing blood viscosity [13].
According to our study 20 children or 69% were hospitalized for hemiplegia diagnosed as stroke.
Studies in Senegal reported hemiplegia as the main telltale sign (84%) followed by partial seizures (37%) [6]. In Nigeria hemiplegia is the main sign of stroke in 45.5% of cases, coma in 18.1% of cases, dysarthria in 9% of cases [14].
According to Arouna Oattara, seizures were the most common clinical signs (46.2%) followed by hemiplegia (23.1%) [5]. Chung B in China, has objectified 52% of cases of convulsions and 34% of hemiplegia [15].
Chronic arterial disease with paradoxical decrease in arterial stiffness and reflection waves, as well as heart failure and PAH are frequently encountered in sickle cell disease. These heart attacks begin as early as 2 years of age [16,17].
In 5 children, cardiac echo-doppler-documented heart disease was identified. But the indication of this imaging was in front of the presence of signs of cardiac decompensation. As a result, undecompensated heart disease may not be objectified in the children in our study.
This is the only predictive examination of stroke in sickle cell children. Mean velocities > 200 cm/s are predictive of the occurrence of cerebral vasculopathy of 40% within 3 years [18,19].
American studies have found a risk of stroke of 10% per year in sickle cell children with an average speed greater than or equal to 200 cm/sec, whereas this risk was only 0.5 to 1% at normal speeds (< 170 cm/sec). This risk is intermediate from 2 to 5% in the case of limit speeds between 170 and 199 cm/sec [20,21].
In Madagascar, TCD is recommended in the sickle cell disease management program at least once a year for monitoring. If an anomaly is detected, the transfusion exchange program is immediately started. A velocimetric profile concerning the average speed of the intracerebral arteries of Malagasy children with sickle cell disease and not, has already been established in a previous study [22].
Systematic screening with TCD is not yet commonplace because of the cost of this examination, yet it is the subject of international recommendations [20].
This study found that TCD was routinely performed in only 9 of the 32 children with clinical cerebral vasculopathy.
Among the remaining 55 children, routine TCD screening revealed cerebral vasculopathy by accelerating blood flow in 3 children. These children immediately entered a transfusion program.
The results of the TCDs carried out by these children revealed a predominance of the involvement of RACA isolated or associated with the involvement of another artery, namely an involvement of the anterior territory (carotid artery and its branches).
According to Adams, et al. there is involvement of the large vessels in 72% of cases, by occlusion of the anterior cerebral arteries (ACA), middle cerebral arteries (MCD) and internal carotid arteries (IC) and junctional accidents in 31% [19]. This is similar to what has been found in our study and observed by other authors, but the explanation is still unknown [21].
Our study demonstrated two biological risk factors which are: hyperleukocytosis and thrombocytosis with respective p and OR of p = 0.014 OR 3.63 (1.27-10.37), and p = 1.9e-05 OR 10.35 (2.5-42.3). The results are similar to those found by Ohene - Frempong K who found a significant link between cerebral vasculopathy and hyperleukocytosis p = 0.026 OR = 1.94 (1.73-2.18) [7].
This is explained physiopathologically by the activation of erythrocytes, leukocytes, and platelets, by hypoxia related to chronic anemia, oxidative stress, and chronic inflammation [23]. They will be responsible for a rheological change.
In Madagascar, the whole blood count for the detection of hyperleukocytosis and thrombocytosis is systematically done at least every 6 months and in the face of worrying clinical manifestations. However, this only applies to children who are regularly monitored within the LCDM.
After statistical analysis, children with cerebral vasculopathy are mostly vaccinated and vaccination against influenza virus has been found as a risk factor for occurrence of cerebral vasculopathy with a p = 0.02 OR 0.16 (95% CI: 0.04-0.7). Our hypothesis is that children with cerebral vasculopathy are those who have a poorly managed disease with several complications, including repeated infections, thus requiring vaccination to limit infectious episodes.
As described in the literature, our results showed that gender and age are not risk factors for vasculopathy in sickle cell patients [5-11].
We found no significant association between cerebral vasculopathies and a history of acute chest syndrome p = 0.12 OR 5 [95% CI: 0.54-62.64], whereas Ohene-Frempong K, et al. found a significant association with a p-value = 0.005 OR 2.39 [95% CI: 1.27-4.48] [12]. There is an association between acute thoracic syndrome and cerebral vasculopathies because it seems that endothelial damage to the cerebral vessels is precipitated by chronic hypoxia secondary to pulmonary micro-embolizations [7].
This study did not establish this significant association because of the size of the sample. And it is possible that some children with acute chest syndrome may have died without being hospitalized.
Although a low haemoglobin level has been proven in some studies to be a risk factor for the occurrence of cerebral vasculopathy, this was not found in our study p = 1 OR 1.04 (95% CI: 0.38-2.28). Kossorotof, Ohene-Frempong K found a significant association between low hemoglobin and cerebral vasculopathies with respective p and OR values of p = 0.038 OR 1.59 (95% CI 1.92-2.44) [7] and p = 0.013 OR 1.59 (95% CI 1.03-2.44) [7].
Our study claimed that cerebral vasculopathy is a real complication (13.4%) that can be responsible for heavy morbidity such as motor and/or cognitive sequelae.
Hyperleukocytosis, thrombocytosis and influenza vaccination were identified as risk factors. A prospective, multi-center study could highlight other factors.
The possibility of easy access to brain imaging could also make it possible to detect these vasculopathies early; and the knowledge of these risk factors makes it possible to initiate primary prevention early.
None.
We would like to express our deep appreciation for the work done:
• The "Centre de Lutte contre la Drépanocytose" for the important contribution to the monitoring of sickle cell children, the realization of transcranial dopplers and access to medical records.
• The head of the Department of Surgical Resuscitation of the Joseph Raseta Andrianavalona Hospital for access to the records of children hospitalized in resuscitation.
• The Head of Haematology Department who allowed us to follow the children during their visits to the day hospital.