A middle-aged African American male with a history of stage 2 chronic kidney disease status post double kidney transplant was admitted after developing symptoms of respiratory infection during the peak of COVID-19. The patient rapidly developed acute hypoxemic respiratory failure. Differentials for the patient included COVID-19, opportunistic infection such as mycoplasma, and legionella. During the admission, the patient had numerous infectious disease screening tests run including a COVID-19 test and Binax urine antigen test for legionella that all came back negative. The patient was started on Doxycycline, due to a recent Mycoplasma infection, Ceftriaxone, Lasix, and Methylprednisolone. Treatment was ineffective and the decision was made to step up investigations to a more invasive procedure. The patient then had bronchoalveolar lavage samples cultured returning a positive legionella sample. The patient was then successfully treated with Azithromycin with resolution of symptoms.
Legionella, Binax, Urine antigen test (UAT), Atypical pneumonia
EIAs: Enzyme Immunoassays; UAT: Urine Antigen Test; BUN: Blood Urea Nitrogen; BiPAP: Bi-level Positive Airway Pressure; Lp: Lipoprotein Assay
Legionella pneumophila is an aerobic, pleomorphic, flagellated, non-spore-forming, Gram-negative bacilli. Legionella is transmitted through aerosolized particles and can be found in surface water, drinking water, and tap water used on surgical wounds [1]. Legionella has been recognized as an important cause of community and hospital-acquired pneumonia. Legionella has fourteen different serogroups and diagnosis of Legionella can pose unique diagnostic challenges [1]. Enzyme Immunoassays are the most used initial diagnostic tool for detecting Legionella's serogroup 1; however, in rare cases these assays may not detect certain serotypes which must instead be diagnosed with more invasive methodology such as bronchoalveolar lavage and culture. Currently 97% of clinical diagnoses are obtained using a urinary antigen test [2]. The urine antigen test uses monoclonal antibodies to detect L. pneumophila serogroup 1 lipopolysaccharide antigens as described previously. The clinical sensitivities for pneumococcal pneumonia were 88.1 and 94.4% for the ImmuView and Binax assays, and 87.6 and 84.2% for the Lp assays, respectively [3]. Test specificities for pneumococcal pneumonia were 96.2 and 97.0% for the ImmuView and Binax assays and 99.6 and 99.1% for the Lp assays [4]. UAT produces results within 15 minutes which is clinically advantageous to culture when treatment must begin urgently [5]. This case report highlights a rare occurrence where a patient was legionella positive after a negative Binax urine antigen test. This case report also seeks to foster clinical suspicion in patients that have a high probability of infection with legionella despite a negative screening test.
A 44-year-old African American male with a past medical history of stage 2 chronic kidney disease post double kidney transplant on tacrolimus regimen presented to his primary care office with complaint of fever, and shortness of breath for three days. The patient had an oxygen saturation of 82% and was admitted to the hospital for acute hypoxemic respiratory failure and suspected COVID-19 infection. The patient returned three separate COVID-19 tests that were negative. After admission the patient described worsening shortness of breath, cough, myalgias, fatigue, and intermittent diarrhea. The patient denied headaches, chills, chest pain, abdominal pain, nausea, and vomiting. The patient denied smoking, drinking and any recent sick contacts. The patient lived with his wife. A review of symptoms was unrevealing. Of note, the patient was admitted one year prior with a similar presentation of atypical pneumonia. During that stay, he required intermittent BiPAP due to worsening hypoxemia. The patient had a positive mycoplasma antigen test and was placed on Doxycycline with resolution of symptoms. The patient's vitals showed tachycardia at 95 bpm, tachypnea at 31 breaths per minute, and oxygen saturation at 82% (Figure 1).
 Figure 1: Chest X-Ray taken upon admission.
View Figure 1
Figure 1: Chest X-Ray taken upon admission.
View Figure 1
On physical exam the patient showed respiratory distress with accessory muscle use and subcostal retractions. Lung sounds revealed decreased breath sounds diffusely with patchy areas of dullness to percussion. All other systems had insignificant findings. The primary differential diagnosis was COVID-19 versus atypical pneumonia. This patient presented at the height of the pandemic and it was shown that COVID-19 had gastrointestinal involvement in the setting of atypical pneumonia. The patient was also immunosuppressed and showed signs of opportunistic infection.
A complete blood count and comprehensive metabolic panel were both obtained once admitted. Preliminary white cell counts were at 9.4 K/μLand the patient showed mild hyponatremia at 134 mmol/L. The patients BUN increased to a maximum of 62 mg/dL with a creatinine of 2.5 mg/dL (1.5 mg/dL baseline). Platelet counts were mildly below normal ranges at 145 K/uL on admission. The patient's hospital course was complicated by this acute renal failure and renal function continued to deteriorate during the admission. All other labs were within normal ranges (Figure 2).
 Figure 2: Lab results from Infectious screening panel.
View Figure 2
Figure 2: Lab results from Infectious screening panel.
View Figure 2
A chest X-ray demonstrated bilateral infiltrates consistent with COVID-19 or an atypical pneumonia. A CT scan showed multifocal airspace opacities throughout both lungs. Doxycycline 100mg BID, Ceftriaxone 1 g BID, Lasix 40 mg BID, and Methylprednisolone 40 mg TID were commenced for broad spectrum coverage and diuresis. Oxygen saturation of 93% was maintained on 4-5 liters nasal cannula after medications were given. Blood cultures, COVID-19, Clostridium difficile toxin, acid fast bacilli culture, fungus culture, cryptococcal antigen, and a Legionella urine antigen tests were all negative. Consulting physicians from infectious disease and pulmonology then decided that a more invasive procedure was needed to obtain direct respiratory samples for culture. A bronchoscopy with lavage was performed to obtain respiratory cultures. His oxygen saturations declined after the bronchoscopy procedure and he was subsequently placed on intermittent BiPAP. The cultures from the Bronchoscopy with alveolar lavage returned a positive sample for Legionella. This was a peculiar finding since the urine antigen came back negative. In the setting of an immunosuppressed patient with pneumonia, other pathogens such as pneumocystis carinii or mycoplasma would be suspected.
At discharge it was determined that the patient required home oxygen as he failed a 6-step test. The patient was sent home with a 21-day course of Azithromycin 500 mg once daily for treatment of Legionella. Two weeks later the patient followed up at an outpatient infectious disease clinic. The patient had and improvement of symptoms, showed a resolved chest X-ray, and had labs return to baseline values. The patient no longer required home oxygen and no further treatment was required. The patients transplant was also deemed unaffected by the admission (Figure 3).
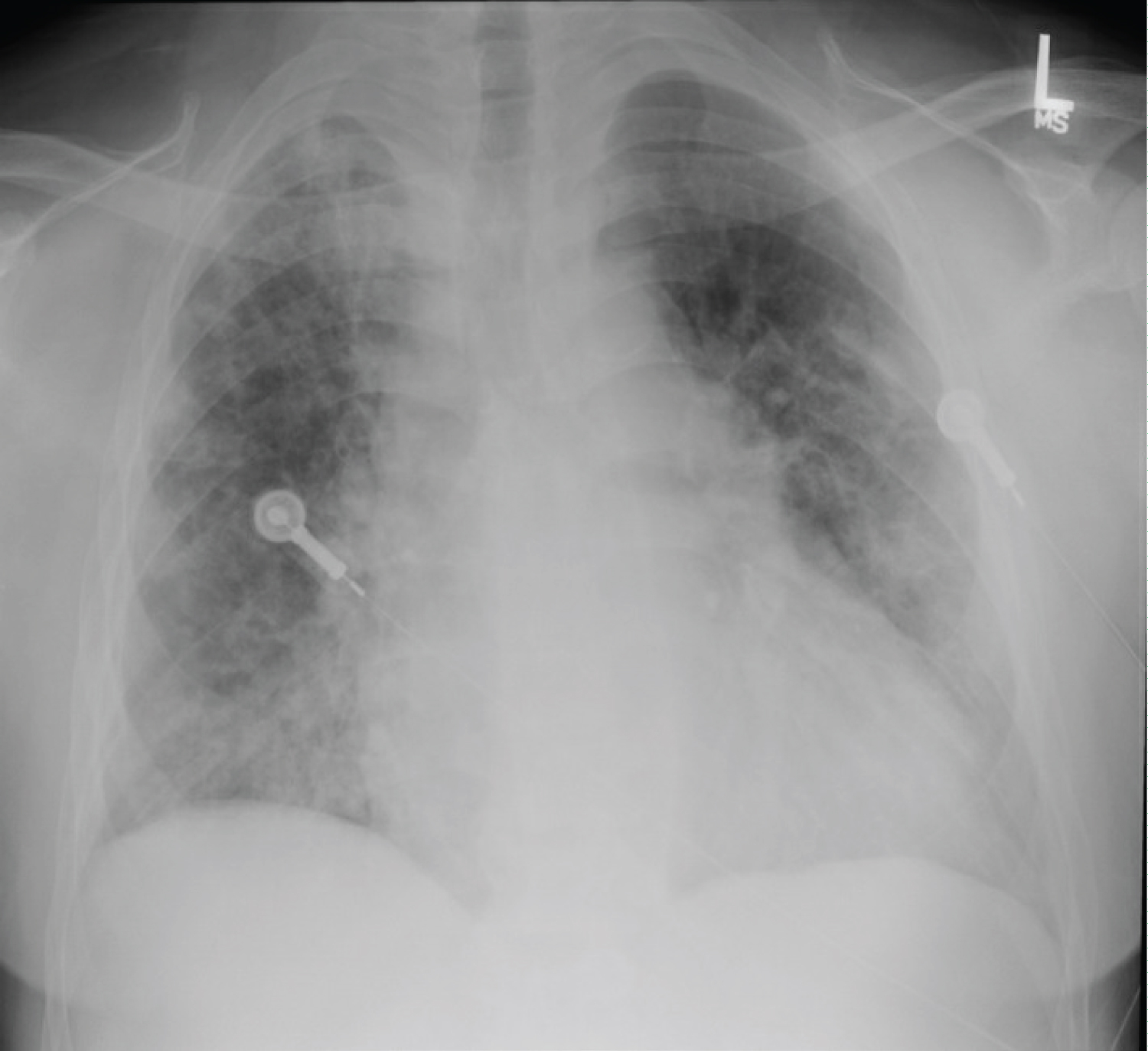 Figure 3: Chest X-Ray taken at 2-week follow-up appointment.
View Figure 3
Figure 3: Chest X-Ray taken at 2-week follow-up appointment.
View Figure 3
The use of enzyme immunoassays (EIAs) for the detection of Legionella, specifically the Binax Legionella Urinary Antigen EIA, is well established and widely used [4]. While this test is reportedly sensitive and specific, it is only intended for L. pneumophila serogroup 1. Although 84% of isolates associated with Legionnaires' Disease are from serotype 1, Legionella should be maintained on the differential in patients who test negative but are in high-risk populations, or for patients with a suspicious clinical picture.
In transplant patients, opportunistic atypical pneumonias should be on the shortlist of a differential diagnosis; however, the clinical picture should be weighted similarly to clinical investigations. An atypical pneumonia presentation with myalgias, diarrhea, and hyponatremia should not single out legionella, but this constellation of symptoms should put it in the top of differentials to consider. A confounding factor to this diagnosis is the specter of COVID-19 that was prevalent when this patient was admitted. COVID-19 had shown gastrointestinal involvement with an atypical pneumonia picture. It was prudent in this case to again consider alternate infectious organisms when multiple COVID-19 tests returned negative. Overall, the clinical picture that a patient presents with should hold as much weight as clinical investigations such as screening tests like the Binax Urine Antigen Test. These tests have been shown to have high sensitivities but are not able to capture the gambit of serogroups that Legionella has.
When considering treatment and long-term outcomes for the patient, prior hospitalizations can be a viable resource for determining where to start in an uncertain clinical picture. This patient was recently infected with Mycoplasma and had successful treatment with Doxycycline. During the admission for Legionella the patient was started on Doxycycline without resolution of symptoms, so Mycoplasma could be placed lower on the differential. This patient eventually had confirmatory testing that showed Legionella infection, but a keener clinical suspicion using the clues mentioned could have prompted clinicians to begin a Macrolide antibiotic sooner in the hospital course. Accurate timely antibiotics leads to decreased mortality and decreased length of stay in patients with sepsis or who are heading towards sepsis [6].
• Despite the high sensitivity and specificity of screening tests for infectious organisms, serogroup variations can render these tests ineffectual.
• The clinical picture of a patient should drive treatment options when the diagnosis is unclear from initial screening tests.
• More invasive procedures will have to be performed when all screening tests come back negative, and the bronchoalveolar lavage with lavage culture is a gold standard confirmatory test.
• Early effective antibiotic treatment based on clinical suspicion and prior hospitalizations should be a key contributor to the development of the initial antibiotic regimen.
The authors in this Case Report contributed equally to the research, summarization, and publication of this article.