Pulmonary langerhans cell histiocytosis (PLCH) is a diffuse lung disease that primarily affects young adults, with cigarette smoking playing a significant role in developing the disease [1,2]. Patients with PLCH present with characteristic CT chest findings of small irregular nodules and upper zone cysts [3-5]. Previously, larger nodules greater than 10 mm and cavitation have only been reported a few times in the literature [6-9]. We describe the case of a 69-year-old male who presented with dyspnea, non-productive cough and weight loss, who was found to have multiple cavitary nodules on CT imaging of the chest. Histopathologic sampling of the lung revealed Langerhans cells which stained positive for S100 and CD1a, consistent with a diagnosis of PLCH. The patient was counselled to quit smoking as the mainstay of treatment. In 3-month follow-up his symptoms had largely resolved, with evidence of decreased nodule size on repeat CT imaging.
Pulmonary Langerhans Cell Histiocytosis (PLCH) is a rare interstitial lung disease that primarily affects young adults between the ages of 20-40 [1,2]. The cell type responsible for PLCH are Langerhans-like cells, which are found in large numbers and express CD1a, CD207 and S100 proteins characteristic of the disease [10]. Most patients with PLCH have high-resolution CT chest findings of small nodules (2-10 mm) and upper zone predominant cysts [3-5]. Here, we present the unique case of an elderly patient presenting with multiple non-cystic cavitary nodules up to 17.2 mm in size.
A 69-year-old male presented to the emergency department with a one-year history of dyspnea on exertion, chronic non-productive cough and weight loss of 30 lbs, referred by his primary care physician for further evaluation. He had a significant smoking history of 75-pack-years, and a rare copper deficiency for which he was treated with copper supplementation. He reported no other medications, allergies, recreational drug use or prior tuberculosis exposure.
The patient had normal vital signs but appeared cachectic. He had quiet breath sounds bilaterally, but no adventitious sounds. C-reactive protein was elevated at 13.7, but otherwise his blood work was unremarkable.
CT chest demonstrated multiple bilateral pulmonary nodules, more prominent in the upper lobes with cavitation (Figure 1 and Figure 2). The largest irregular nodule was in the left upper lobe and measured 17.2 mm. The largest nodule in the right upper lobe measured 12 mm. In addition, there was centrilobular emphysema in the upper lobes bilaterally. Given the concern for malignancy, the patient underwent a bone scan which was unremarkable, and a CT abdomen and pelvis, which showed mild asymmetric rectal thickening. As a result, the patient had a gastroscopy and colonoscopy, which were normal. Subsequently, a CT-guided core needle biopsy of the largest lung nodule was performed and the patient was discharged due to his stable clinical status.
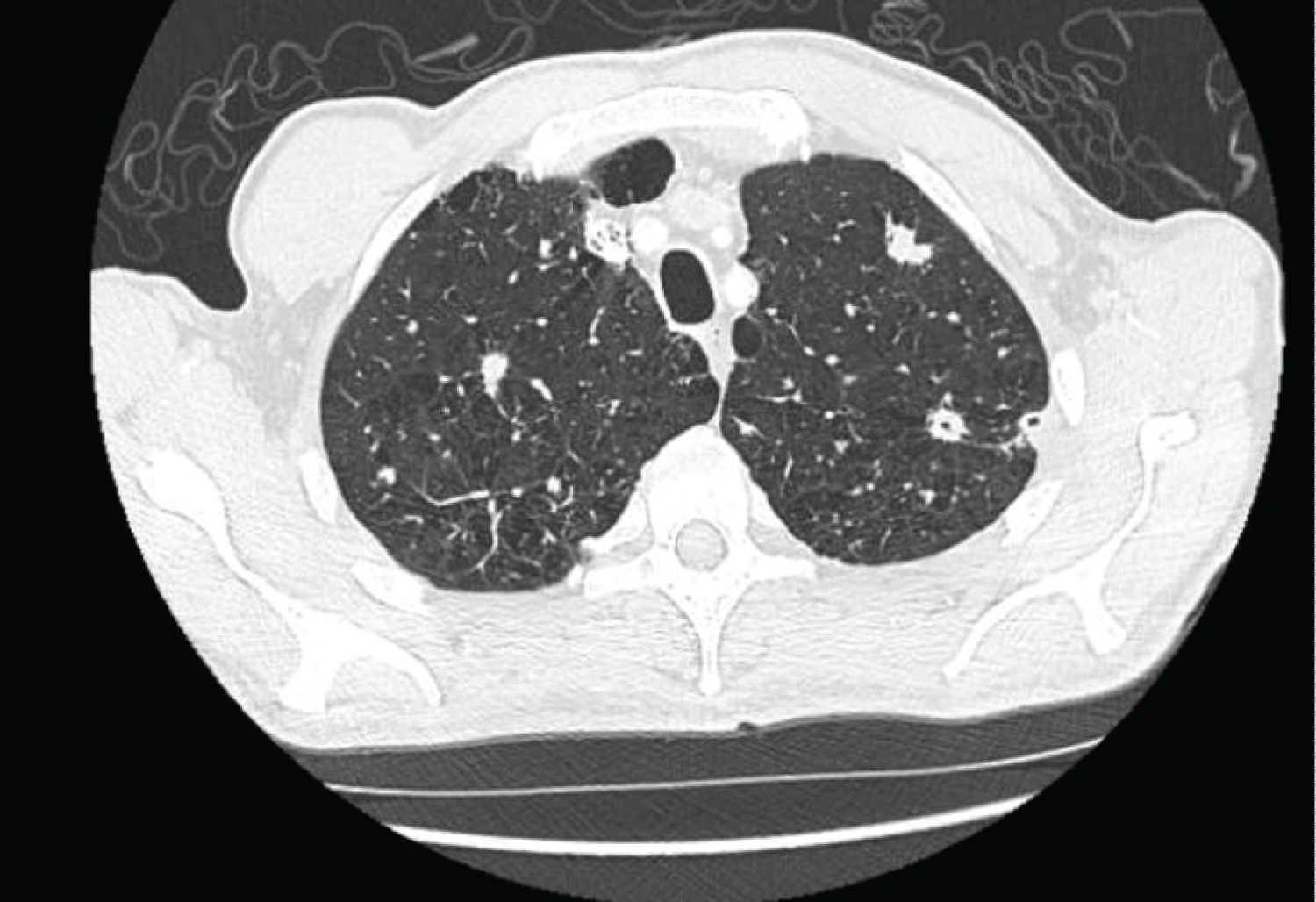 Figure 1: Presence of multiple large nodules in the upper lobes with cavitation, along with centrilobular emphysema.
View Figure 1
Figure 1: Presence of multiple large nodules in the upper lobes with cavitation, along with centrilobular emphysema.
View Figure 1
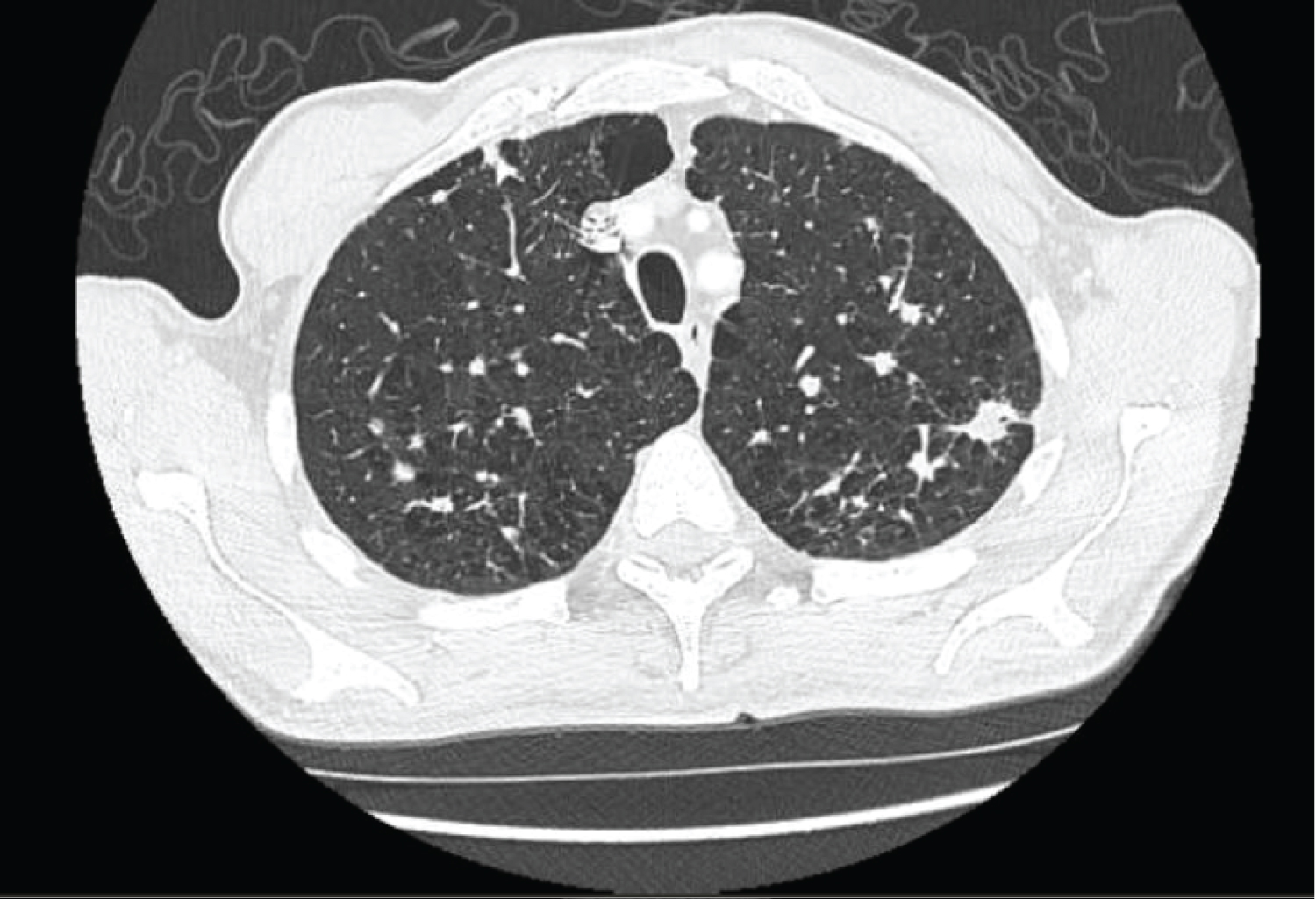 Figure 2: Presence of a large nodule in the left upper lobe measuring 17.2 mm in size with evidence of cavitation.
View Figure 2
Figure 2: Presence of a large nodule in the left upper lobe measuring 17.2 mm in size with evidence of cavitation.
View Figure 2
The patient was seen in follow-up pulmonology clinic and underwent bronchoscopy and pulmonary function testing (PFT). Bronchoalveolar lavage (BAL) did not reveal any malignant cells and contained sparse histiocytes which were negative for CD1a. PFT revealed FEV1/FVC ratio of 0.64, normal lung volumes and a diffusion capacity of 58%. A lung pathologist reviewed the histology demonstrating non-necrotizing granulomas with histiocyte-type cells consistent with Langerhans cells which stained positive for S100 and CD1a. The patient was counselled to quit smoking as the mainstay of treatment.
On 3-month follow-up, with smoking cessation, the patient had experienced near resolution of his symptoms. Repeat CT chest also demonstrated significant decrease in his pulmonary nodules, with the largest nodule measuring 10 mm in size (Figure 3).
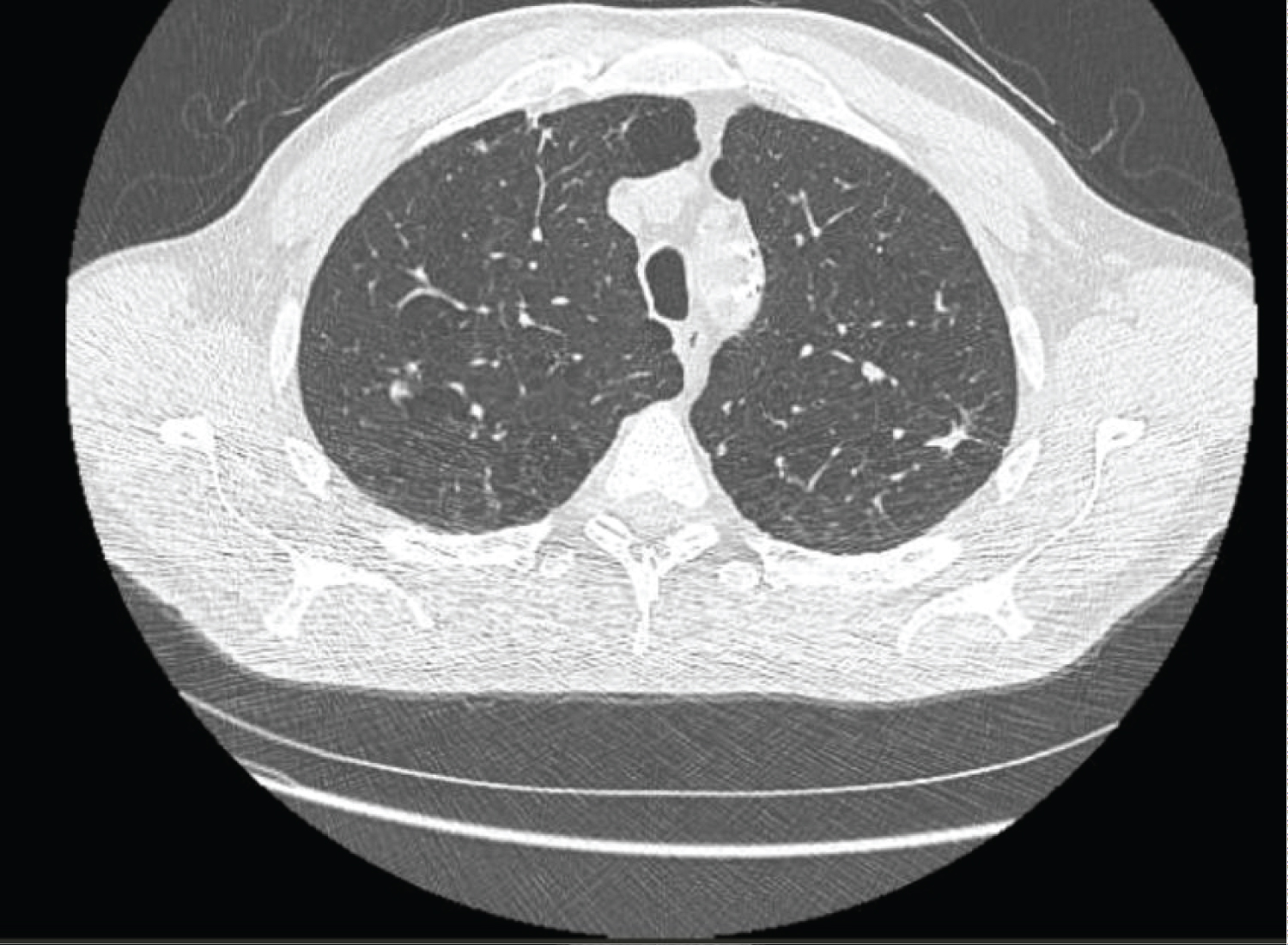 Figure 3: Follow-up CT scan 3 months after smoking cessation demonstrating a significant decrease in the size of the nodules, with the largest nodule in the left upper lobe now measuring 10 mm in size.
View Figure 3
Figure 3: Follow-up CT scan 3 months after smoking cessation demonstrating a significant decrease in the size of the nodules, with the largest nodule in the left upper lobe now measuring 10 mm in size.
View Figure 3
PLCH often presents with non-productive cough and dyspnea, and constitutional symptoms such as fever, fatigue and weight loss [11]. PFT often demonstrates normal lung volumes and limited obstruction [12]. BAL can support the diagnosis when it demonstrates greater than 5 percent Langerhans-like cells which are positive for CD1a and CD207.2 Lung biopsy results will show a characteristic cluster of Langerhans-like cells on histology, which stain for the CD1a, CD207 and S100 proteins [2]. Treatment of the disease is focused on smoking cessation [13]. In a case series of 40 patients, smoking cessation was the only treatment needed in 25 patients for resolution or stabilization of the disease [13].
Our patient presented with a case of PLCH that did not fit common diagnostic and clinical parameters, leading us to strongly consider malignancy as a potential diagnosis. Our patient had large cavitary nodules, up to the size of 17.2 mm. Larger non-cystic nodules greater than 10 mm and cavitation is common in malignancy but has only been reported in PLCH a handful of times [6-9]. The evolution of nodules on CT imaging appears to be that of smaller nodules progressing over time to cysts, but our patient had large non-cystic nodules on presentation [14]. Additionally, our patient was 69 years of age, which is uncommon, as most cases of PLCH occur between the ages of 20-40 [1].
Interestingly, our case did offer subtle clues that suggested an alternative diagnosis to malignancy on initial presentation. The patient's large cavitary nodules were in the upper lobes, whereas metastatic cancer would be most likely to present in the lower lobes due to greater perfusion in the lung bases. Additionally, although the patient reported significant weight loss and other constitutional symptoms, this is also a common finding in interstitial lung diseases such as PLCH [11]. Our case report therefore highlights a unique presentation of this disease and serves to encourage physicians to consider this PLCH in their differential diagnosis when faced with an atypical presentation.