Cerebral Palsy (CP) is a heterogeneous group of conditions that cause non-progressive childhood disabilities, characterized by varying degrees of motor and intellectual dysfunction. To date, no fully restorative treatment for CP has been developed, however, a few clinical trials conducted in the past have implicated the potential benefit of Mesenchymal Stem Cell (MSC) therapy for the patients with CP.
Here, we present a case report of a 14-year-old, underweight, male child diagnosed with spastic cerebral palsy with a severe manifestation of scoliosis. Patient's low body weight posed a substantial challenge, as it precluded him from having an elective scoliosis surgery. We employed multiple doses of autologous, Hope-Biosciences adipose-derived MSCs (HB-adMSCs) for the treatment, with a goal to improve patient's weight to enable him to undergo his scoliosis surgery and ultimately improve his quality of life. Thirty-two (32) recurrent intravenous infusions (each with ~ 50 × 106 cells) were administered over the course of almost 3 years.
Multiple infusions of HB-adMSCs resulted in significant improvements in the patient's well-being. Importantly, patient's weight got significantly improved, which allowed him to undergo a much-needed scoliosis surgery. Additionally, recurrent therapy also resulted in improvements in the patient's neurological state. No serious adverse events occurred during the entire treatment period.
HB-adMSCs may offer a safe and effective treatment option to improve the quality of life in patients with CP. Larger clinical studies are needed to confirm these findings.
Mesenchymal Stem Cells, Adipose derived, Autologous, Cerebral palsy, Stem cell therapy, Case report
Ad-MSCs: Adipose Derived Mesenchymal Stem Cells; BMI: Body Mass Index; CBC: Complete Blood Count; CMP: Comprehensive Metabolic Panel; CP: Cerebral Palsy; EOS: End of Study; FDA: Food and Drug Administration; FU: Follow-up; HB-adMSCs: Hope Biosciences Adipose Derived Mesenchymal Stem Cells; MSCs: Mesenchymal Stem Cells; SAE: Serious Adverse Event; SVF: Stromal Vascular Fraction; WIRB: Western Institutional Review Board
Cerebral Palsy is a severe childhood motor disability affecting function and development, with the incidence reported as 1-2 per 1000 live births in developed countries [1]. The etiology of CP involves prenatal, perinatal, or postnatal complications including birth asphyxia, preterm birth, trauma, low birth weight, infection or inflammation that may lead to fetal or infant brain damage during development [2]. CP is a non-progressive disorder characterized by a heterogeneous motor disability, with the clinical manifestations that range from motor impairment of a single body part to inability of walking, accompanied by a range of other neuromotor impairments, such as intellectual and behavioral abnormalities, neuropsychological disorders, cognitive and visual impairment, epilepsy, and movement disorders [3]. Other secondary disorders like poor growth and malnutrition are also common in children with CP, severely affecting their quality of life [4]. Based on what parts of brain are affected, CP can be categorized into - spastic, ataxic or dyskinetic with the prominence of spasticity in ~70% of the affected children [5]. CP patients have reported incidence of scoliosis ranging from ~20-25% [6,7], however, there is an increased incidence risk of 64% in patients with total body involvement. Research has shown that spastic CP population typically have manifestation of scoliosis with the highest incidence in the spastic quadriplegics [8].
Owing to their immunomodulatory and regenerative capabilities, MSCs have been emerging as a promising therapeutic approach to treat several diseases. MSCs are multipotent with their ability to inhibit inflammation through secretion of several bioactive molecules, thereby stimulating recovery of injured cells [9]. Both preclinical and clinical studies have implicated therapeutic potential of MSCs in treatment of patients or animal models of CP [10,11]. Previously, clinical trials using MSCs derived from multiple sources (bone-marrow, umbilical cord blood or umbilical cord) provided evidence for beneficial effects of MSCs in treatment of patients with CP [12-15]. Patient-derived stem cells offer a safer treatment option with a feasibility of multiple transplantations, since these do not induce immune rejection, which may arise in case of allogeneic stem cell transplantations. Here, we report noticeable improvements in body weight and serum leptin levels of a 14-year-old CP individual using fresh, culture-expanded adipose-derived mesenchymal stem cells. We used multiple doses of autologous, HB-adMSCs with a goal to treat our CP patient to gain sufficient weight to undergo surgery for his severe scoliosis and to ultimately improve his quality of life.
The patient was a 14-years-old non-verbal, male child with spastic quadriplegic CP, presented with severely affected quality of life, who has exhausted all traditional treatment options without any improvement in his conditions. He was born at 28 weeks, product of a monochorionic twin pregnancy, survivor recipient of a Twin-to-Twin Transfusion Syndrome (TTTS) stage V of Quinteros classification. Soon after birth the patient was diagnosed with Grade I Intraventricular Hemorrhage (IVH), Periventricular leukomalacia, Neonatal Respiratory Distress Syndrome and Retinopathy of prematurity. During the first year of life, his neurological development was severely affected, leading to a later diagnosis of spastic quadriplegic cerebral palsy and global developmental delay. He suffered constant pain due to the most severe scoliosis, one of the common manifestations of cerebral palsy. He was denied surgical intervention and was deemed a poor surgical risk due to his extreme malnutrition, poor body weight and failure to thrive. Despite continuous consultations with gastroenterologists and nutritionists as well as being fed table food by his mother with round the clock tube feedings via the jejunostomy port of his GJ feeding tube, he was unable to gain body weight. At the time of screening (baseline), he weighed 57.60 pounds (lbs.) and was cachectic.
His spasticity was poorly controlled with routine Botox injections to his hips and limbs as well as oral baclofen following three failed trials with an implanted baclofen pump in the past. He had a severe onset of sialorrhea at the age of 9, for which he was being treated with Botox injections in his salivary glands. He had very low baseline serum leptin levels of 0.5 ng/ml compared to the normal acceptable levels of 2.71 ± 0.19 ng/ml for his age [16].
After receiving authorizations from Food and Drug Administration (FDA) and Western Institutional Review Board (WIRB) on May 24, 2019 and June 06, 2019 respectively, the patient started receiving infusions of autologous HB-adMSCs with his first infusion administered on July 17, 2019. HB-adMSCs were isolated from 4.5 mL of adipose tissue from subcutaneous fat as the source of MSCs as opposed to Hope Biosciences' standard method using abdominal fat, since the patient was under-weight. The tissue was disinfected with 70% alcohol due to possible contaminants from the fat extraction procedure, followed by dissection and treatment with collagenase to separate the stromal vascular fraction (SVF). Cells from the SVF were plated in Hope Biosciences' HB-103 medium to establish a P0 culture. The resulting adherent cells were further cultured with HB-101, Hope Biosciences' growth medium. Cells were cryopreserved at passages #0, #1 and #2 to create a complete cell bank for the patient. For infusions, passage #2 cells were thawed, recovered in passage #3 and cultured to passage #4. A total of 32 infusions (manufactured from the initial cell bank created for the patient and freshly harvested from passage #4), each with 50 million ± 20% MSCs mixed in 250 ml of sterile saline were administered intravenously, over a course of ~3 years (from July 2019 to October 2021) (Table 1). Each product lot for infusion underwent cGMP-compliant quality control standard assessments, which included viability; appearance; sterility (USP71); gram staining; mycoplasma; endotoxin; and cell identity/purity as indicated by MSC defining surface markers (CD73+, CD29+, CD45- and CD31-), to ensure a standardized product is delivered for each treatment.
Table 1: Infusion details with MSC quality control metrics. View Table 1
Following the first few interventions, marked improvements in his weight became apparent, which culminated in a significant gain by the end of study after 32 infusions (Figure 1). The patient was able to meet a required goal of at least 80 lbs. to undergo surgery for his scoliosis and weighed 84.2 lbs. after 15th infusion on June 24, 2020, before he underwent his surgery on December 16, 2020 (post infusion 22), when he weighed ~100.8 lbs. The surgery went successful with the correction of his scoliosis. Of note, he gained a total of 60.4 lbs. (104.86% increase from baseline) during the whole conduct of the study. The increase in weight was statistically significant (p < 0.0001) as revealed by a simple linear regression analysis (Figure 1).
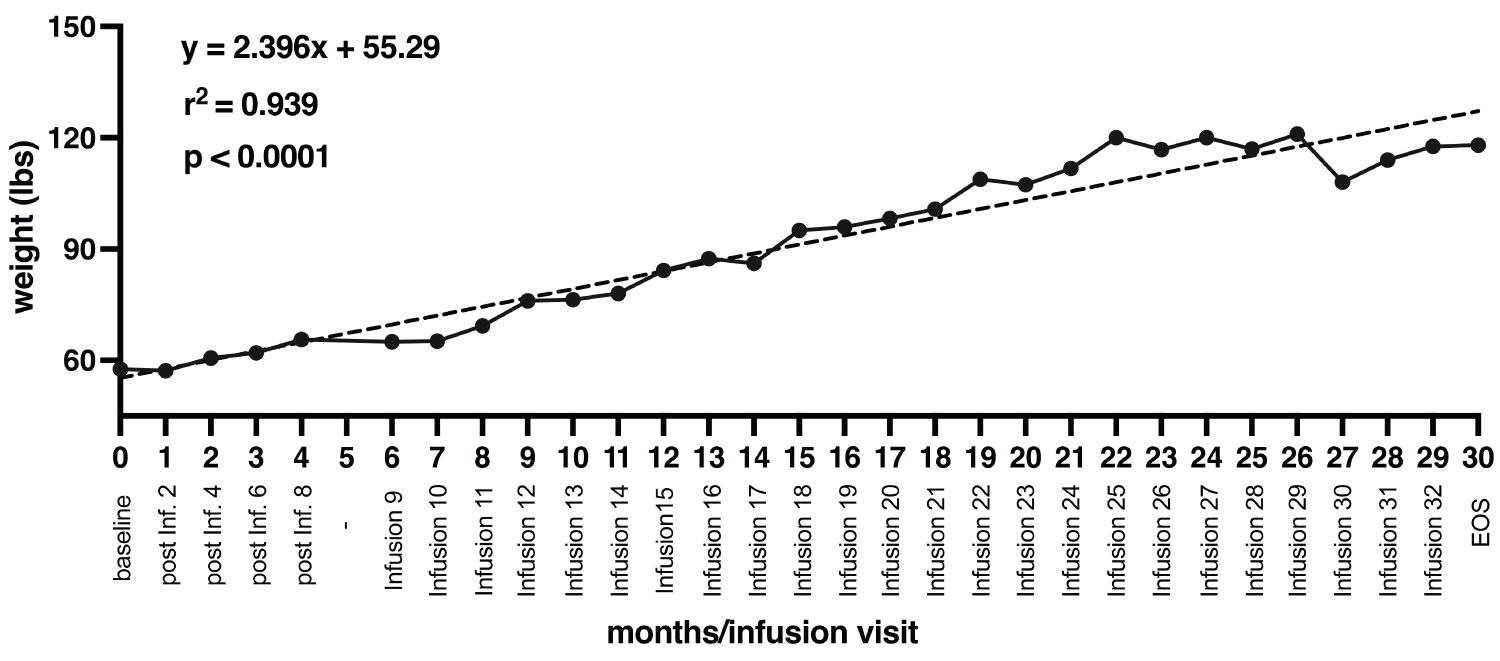 Figure 1: Changes in patient's weight with respect to each infusion over the course of entire trial (32 infusions total over the course of 30 months). The dashed black line represents simple linear regression analysis, showing a statistically significant positive correlation between changes in patient's weight with respect to number of infusions (p < 0.0001).
Figure 1: Changes in patient's weight with respect to each infusion over the course of entire trial (32 infusions total over the course of 30 months). The dashed black line represents simple linear regression analysis, showing a statistically significant positive correlation between changes in patient's weight with respect to number of infusions (p < 0.0001).
EOS: End of Study.
View Figure 1
As part of our safety assessment of the intervention, standard laboratory evaluations of complete blood count (CBC) and comprehensive metabolic panel (CMP) were performed at various timepoints. No unusual changes in patient's CBC or CMP components (WBC, Neutrophils, Monocytes, Lymphocytes, Basophils, Eosinophils, Granulocytes, Albumin, HGB, HCT, Protein, Bilirubin, BUN, Creatinine, Glucose, Carbon Dioxide, Chloride, Potassium, Sodium, Alkaline Phosphatase, ALT, AST) were observed, compared to baseline. No treatment-related adverse events or serious adverse events (SAEs) were reported during the whole course of treatment. Remarkably, his serum leptin levels were significantly increased over the course of the trial, with a 28-fold increase at the end of study visit (14 ng/ml) compared to baseline (0.5 ng/ml) (Figure 2; simple linear regression analysis, p < 0.0001).
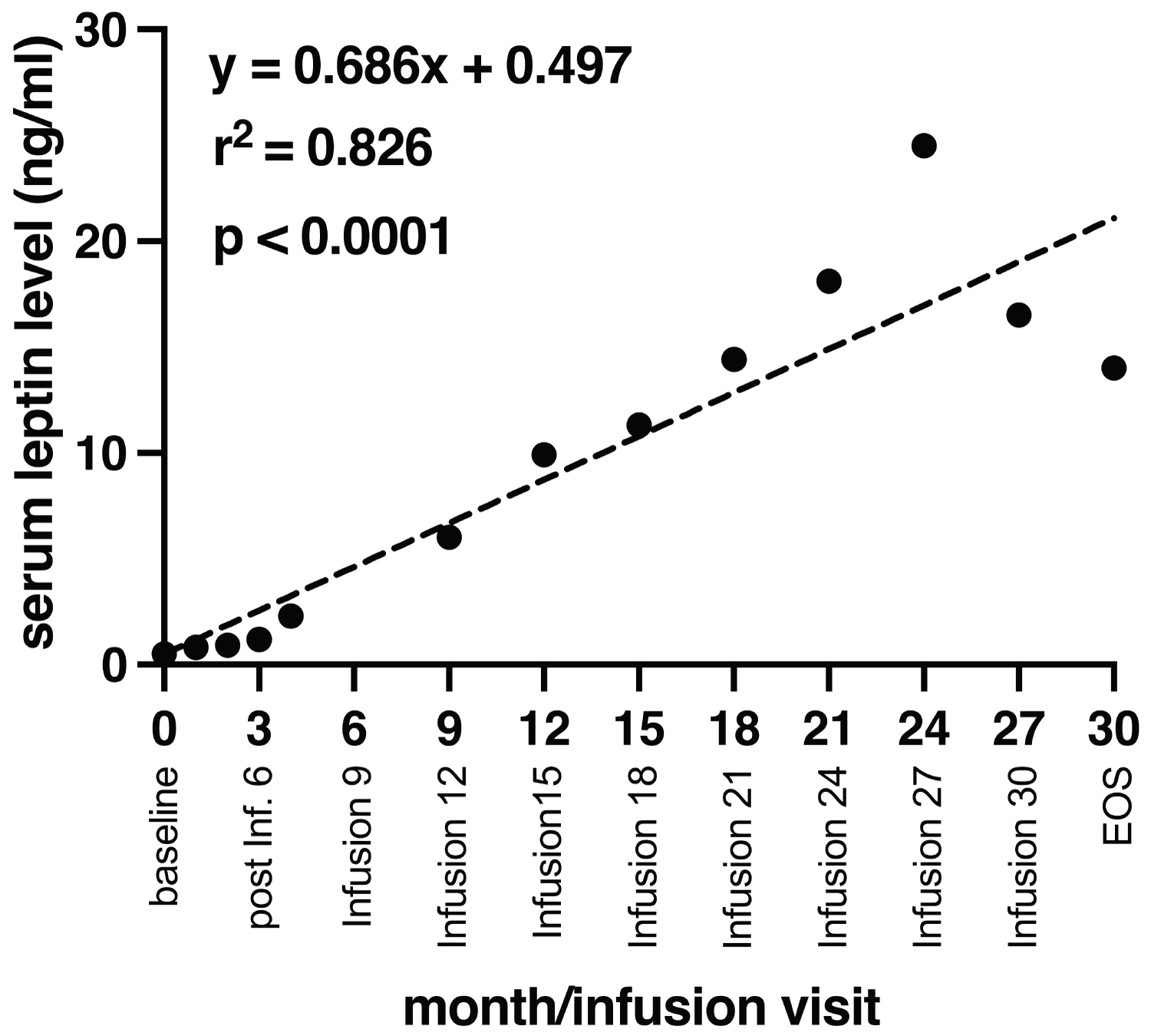 Figure 2: Linear regression plot for serum leptin with respect to infusion number. The dashed line represents regression line showing a positive correlation between levels of serum leptin and number of infusions with a statistically significant regression (p < 0.0001).
Figure 2: Linear regression plot for serum leptin with respect to infusion number. The dashed line represents regression line showing a positive correlation between levels of serum leptin and number of infusions with a statistically significant regression (p < 0.0001).
EOS: End of Study.
View Figure 2
We used the SARAH scale, a common motor-functional evaluation scale for children and adolescents with CP [17], to assess and evaluate post-intervention motor-functional and related assessments, compared to baseline. Although evaluation scale scores for either motor or functional dimension of SARAH did not show any significant changes, however, noticeable improvements were seen in patient's neurological state when compared to the baseline visit (Table 2). Amelioration of involuntary movement disorders along with other CP-associated disorders was observed after the administration of few infusions of HB-adMSCs, e.g., no indication of dystonia was noticed after only 6 infusions of HB-adMSC therapy (Table 2). Also, visual impairment was substantially rectified after the end of 9 infusions, in addition to noticeable improvements in neuropsychological disorders (post 12 infusions). Athetosis, another form of involuntary movement, also declined over the course of the study after 30 injections. With a prior medical history of gastroparesis since December 2010, the patient was also diagnosed with gastroesophageal reflux disease during the intervention period (indicated at month 9, infusion 15; Table 2). However, the condition was resolved after few infusions, well before the end of the study (Table 2).
Table 2: Changes in patient's neurological state (motor impairments, involuntary movements, and associated disorders) measured at baseline, intervention/follow-up (FU) visits and at the end of study FU visit. View Table 2
Due to their easy accessibility, multipotent ability, low immunogenicity, and immunomodulatory properties, MSCs have been previously employed in treatment of numerous studies with proven clinical efficacy. Functional efficacy of MSC therapy has also been implicated in the treatment of neurological and neurodegenerative pathologies because of their neuroprotective potential [18,19]. In the recent years, MSCs have emerged as a novel therapeutic paradigm for the treatment of patients with CP. Children with CP often exhibit poor growth, low weight, and nutritional dysfunction, along with their neuro-motor and functional impairments [20,21]. Previous studies using assessments of nutritional status have indicated the prevalence of underweight with low body mass index in patients with quadriplegic, low-functioning CP patients [22,23]. Many CP patients require elective surgeries to correct for various abnormalities; however, their poor body weight renders them high surgical risks. Weight gain was also a substantial challenge for this patient that had even precluded him to undergo a much-needed spinal surgery to correct his scoliosis. Here, in this study, we used multiple infusions of autologous HB-adMSCs with a goal to improve his weight so that he can undergo his surgery for the scoliosis. The patient started to gain weight beginning a few infusions with a significant increase over the entire course of study. Consequently, the patient was able to undergo a successful surgery with a correction of his scoliosis. Overall, our HB-adMSC therapy has demonstrated significant improvements in the patient's weight and serum leptin levels that also correlated with the rise in his body mass index (BMI), increasing from 12.5 to 21.6 over the course of the trial.
Since the start of HB-adMSC therapy, the patient experienced much less drooling, and he did not feel the need of Botox therapy for spasticity or sialorrhea, indicative of his improved resistance to the disease. Additionally, his language expression also strengthened with the treatment; for example, he would very clearly call out “mama” and looks for his mother or father when they are out of his sight. He also showed obvious improvements in perception. He could respond to simple questions with yes or no answers and started to demonstrate clear preferences for what he needed/liked or not. Post-treatment, he had started to communicate with a computer screen interface. Also, he had substantial improvements in his vision, as evidenced by his increased focus towards television screen.
Previously, a preclinical study using human umbilical-cord derived MSCs for wound regeneration reported that mice that received MSC therapy represented stability in body weight due to increase in the metabolic processes, that resulted in concomitant faster healing [24]. Here, in this study, the illustration of beneficial effects of HB-adMSC therapy has been presented that showed significant improvements in patient's weight. At the end of study visit (after 32 infusions) the patient had a healthy BMI with the total weight of 118 lbs. implicating the beneficial effects of the therapy.
In conclusion, the study results of HB-adMSC therapy for a severe quadriplegic CP individual showed that multiple doses of HB-adMSCs have significantly beneficial effects on patient's weight which led him to undergo his elective spinal surgery. Also, HB-adMSC therapy helped the patient enhancing his neurological state, as implicated by improvements observed in many CP-associated disorders, ultimately improving his quality of life. Based on the level of severity of the disease, HB-adMSCs may offer a safe and effective treatment to differentially improve the quality of life of CP patients. Further studies are needed to confirm these results.
We acknowledge Ranjan Prafull Shah, MD, and the clinical staff for conducting the screening visit as well as the first nine infusions for this study, as the employees of Clinical Trial Network, Houston, Texas. We also thank Rajiv Thakur, MD, the employee of Advanced Diagnostics Hospitals & Clinics, Houston, Texas, who served as a medical monitor for this study.
This study was approved by Western International Review Board, Inc. Washington, USA and was conducted in accordance with Good Clinical Practice guidelines and the Declaration of Helsinki. Ethical approval to report this case was not required. Written informed consents for participation in the study were obtained from patient's parents.
Written informed consent was obtained from the patient's legal guardian for publication of this case report. A copy of the written consent is available for review by the Editor-in-Chief of this journal.
The data that support the findings of this study are available from the corresponding author upon request.
RV, DL and TC declare no potential conflict of interests. DC, HK, and HP are employees and shareholders of Hope Biosciences LLC.
This study was funded in whole by Hope Biosciences Research Foundation and Hope Biosciences LLC provided the investigational drug.
RV wrote the manuscript and performed all the analysis. HK and HP prepared the investigational product, DL and TC conducted the study as principal investigator and sub-investigator respectively, DC provided financial and administrative support, reviewed the case report, and provided final approval for submission. All authors read and approved the final submission of this manuscript.