Parkinson's disease (PD) is a chronic, progressive, neurodegenerative disease with a multifactorial etiology, the predominant pathology of PD is the loss of dopaminergic cells in the substantia nigra. It is characterized by hallmark signs of bradykinesia, rigidity, tremor, and postural instability. Medical and pharmacological treatments for Parkinson's disease are limited to the symptomatic relief of patients, and has failed to prevent or slow down the process of neurodegeneration. Cell transplantation is a strategy with great potential for the treatment of Parkinson's disease, Mesenchymal stem cells are a great therapeutic cell source because they are easy accessible. They have trophic effects for protecting damaged tissues as well as differentiation ability to generate a broad spectrum of cells, including dopamine neurons, which contribute to the replenishment of lost cells in Parkinson's disease.
Parkinson's disease, Substantia nigra, Dopaminergic neurons, Stem cells, Mesenchymal stem cells
Definition: Parkinson's disease (PD) is a chronic, progressive, neurodegenerative disease with a multifactorial etiology. It is characterized by hallmark signs of bradykinesia, rigidity, tremor, and postural instability, it is superseded only by Alzheimer's disease as the most common neurodegenerative disorder [1]. PD exerts substantial burden on patients, families of patients, and caregivers [2] and is associated with a significant increase in morbidity and disability [3,4].
Parkinsonian symptoms can arise from either the neuropathologic condition of PD (idiopathic PD [iPD]) or other forms of parkinsonism. For neuropathologic PD, about 90% of cases are sporadic, with no clear etiology; an additional 10% have a genetic origin, and at least 11 different linkages with 6 gene mutations have been identified [5] Genetic forms of PD are seen more frequently in young-onset PD. A combination of environmental factors or toxins, genetic susceptibility, and the aging process may account for many sporadic cases [6] Secondary forms of parkinsonism can be caused by medications, the sequelae of central nervous system infection, toxins, or vascular/metabolic disorders (Figure 1). The only proven risk factor for PD is advancing age [7]. Other environmental or lifestyle risk factors associated with development of PD are rural living, exposure to pesticides and herbicides, well-water drinking, and working with solvents [6]. However, none of these factors unequivocally has been demonstrated to cause iPD [8].
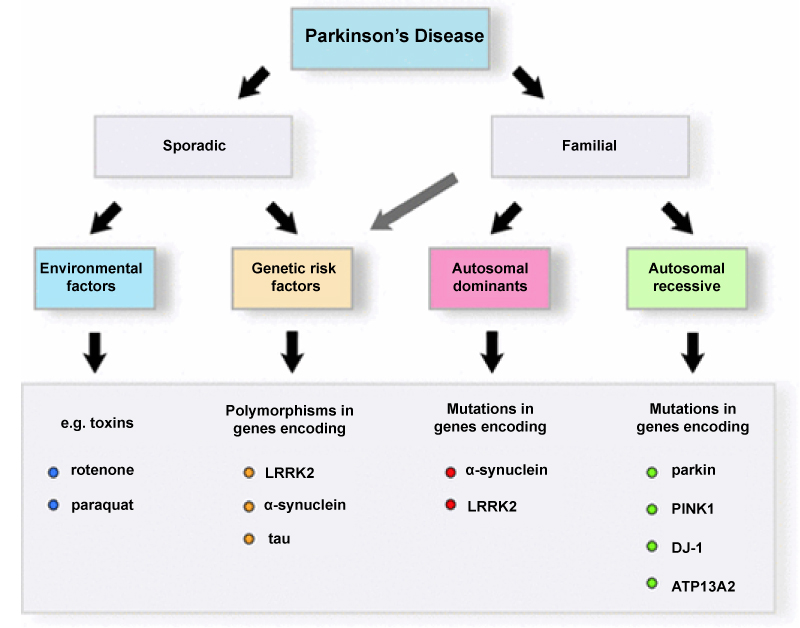 Figure 1: Etiology of Parkinson's Disease (PD).
View Figure 1
Figure 1: Etiology of Parkinson's Disease (PD).
View Figure 1
Basal ganglia motor circuitry: Parkinson's disease is predominantly a disorder of the basal ganglia, which are a group of nuclei situated at the base of the forebrain (Figure 2). The striatum, composed of the caudate and putamen, is the largest nuclear complex of the basal ganglia. The striatum receives excitatory input from several areas of the cerebral cortex, as well as inhibitory and excitatory input from the dopaminergic cells of the substantia nigra pars compacta (SNc). These cortical and nigral inputs are received by the spiny projection neurons, which are of 2 types: those that project directly to the internal segment of the globus pallidus (GPi), the major output site of the basal ganglia; and those that project to the external segment of the globus pallidus (GPe), establishing an indirect pathway to the GPi via the subthalamic nucleus (STN). The actions of the direct and indirect pathways regulate the neuronal output from the GPi, which provides tonic inhibitory input to the thalamic nuclei that project to the primary and supplementary motor areas [9].
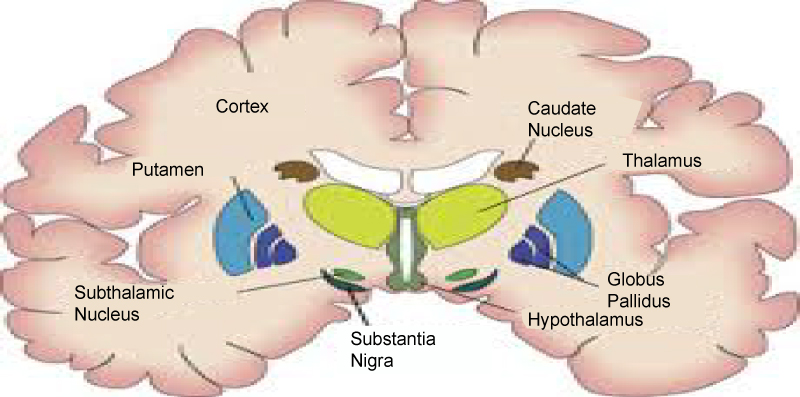 Figure 2: Anatomy of basal ganglia.
View Figure 2
Figure 2: Anatomy of basal ganglia.
View Figure 2
The basal ganglia motor circuit modulates the cortical output necessary for normal movement Signals from the cerebral cortex are processed through the basal ganglia-thalamocortical motor circuit and return to the same area via a feedback pathway. Output from the motor circuit is directed through the internal segment of the globuspallidus (GPi) and the substantia nigra pars reticulata (SNr). This inhibitory output is directed to the thalamocortical pathway and suppresses movement [10].
Two pathways exist within the basal ganglia circuit, the direct and indirect pathways (Figure 3), as follows:
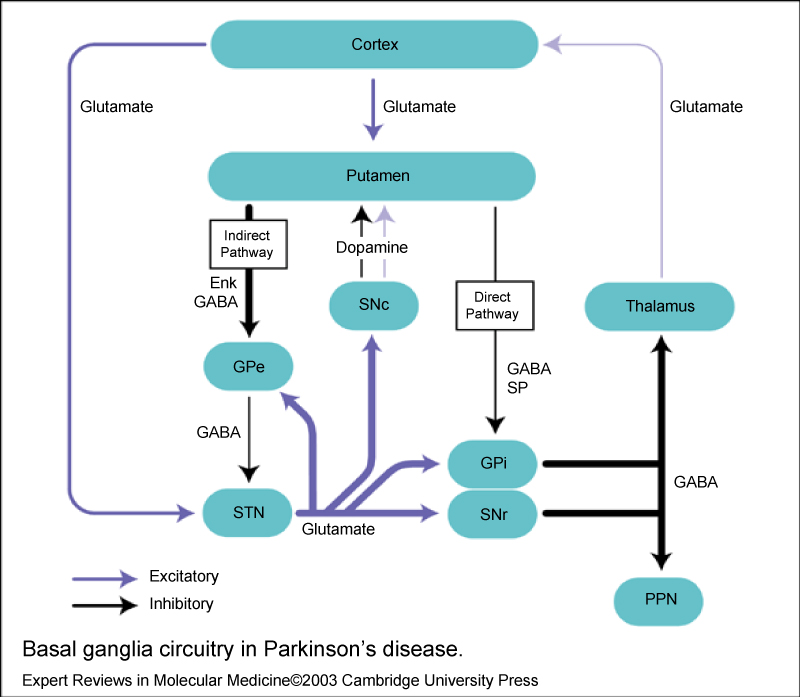 Figure 3: Basal ganglia circuitry in Parkinson's disease.
View Figure 3
Figure 3: Basal ganglia circuitry in Parkinson's disease.
View Figure 3
• In the direct pathway, outflow from the striatum directly inhibits the GPi and SNr; striatal neurons containing D1 receptors constitute the direct pathway and project to the GPi/SNr
• The indirect pathway contains inhibitory connections between the striatum and the external segment of the globuspallidus (GPe) and between the GPe and the subthalamic nucleus (STN); striatal neurons with D2 receptors are part of the indirect pathway and project to the GPe [9].
The STN exerts an excitatory influence on the GPi and SNr. The GPi/SNr sends inhibitory output to the ventral lateral nucleus (VL) of the thalamus. Dopamine is released from nigrostriatal (substantia nigra pars compacta [SNpc]) neurons to activate the direct pathway and inhibit the indirect pathway. In Parkinson disease, decreased striatal dopamine causes increased inhibitory output from the GPi/SNr via both the direct and indirect pathways. The increased inhibition of the thalamocortical pathway suppresses movement. The loss of dopaminergic neurons in the substantia nigra pars compacta, which results in a reduction in the level of dopamine in the striatum, leads to alterations in the activity of striatal output nuclei. This results in changes in the other nuclei basal ganglia, which can be summarized as following: (a) Degeneration of the nigrostriatal pathway, (b) The under activity of the GABA/dynorphinstriato-medial pallidal/SNr nigral pathway, (c) The overactivity of the GABA/enkephalinstriato-lateral-pallidal pathway, (d) the overactivity of the subthalamic nucleus, (e) The overactivity of the GABA medial pallidal/SNr (output regions of the basal ganglia) -thalamic projection [9]. The overactivity of basal ganglia output results in increased inhibition of excitatory glutamatergic projections from the thalamus to the cortex. Cortical motoroutputs are, thus, underactive leading to the movement paucity in Parkinson's disease [10].
Although the predominant pathology of PD is the loss of dopaminergic cells in the substantia nigra, however, there is also degeneration of other neurotransmission systems, such as cholinergic, noradrenergic, serotoninergic and peptidergic brainstem nuclei. Some of these alterations in neurotransmitters occur before the appearance of Parkinsonian symptoms. Noradrenaline (NA) is particularly implicated in certain symptoms of Parkinson's disease. Biochemical analysis revealed that 40-80% of the brain's content of NA is depleted in PD [9].
Neuropathological manifestation: Neuronal changes in parkinson's disease are widespread, however the principle neurological abnormalities that occur are: I) The loss of dopaminergic neurons in the substantia nigra pars compacta, led to reduced dopaminergic input to the striatum and accompanied by adaptive responses in the internal and external globuspallidus, subthalamus, thalamus and substantia nigra pars reticularis. II) Round, hyaline neuronal cytoplasmic inclusions called Lewy bodies (LBs) and enlarged aberrant neurites and threads are found in the Parkinsonian substantia nigra [10,11]. In addition to the substantia nigra, other nuclei are involved such as the locus ceruleus, reticular nuclei of the brain stem, and dorsal motor nucleus of the vagus, as well as the amygdala and the CA2 area of the hippocampus. LBs and neuritis (Figure 4) -pathological hallmarks of PD-are composed of aggregates of normal, misfolded and truncated proteins, and ubiquitin, all of which are stored in the cytoplasm as nondegraded by-products of the degenerative process [12-16]. The main component of LBs and aberrant neurites is α-synuclein (a protein suggested to have a role in DA synthesis, synaptic plasticity and vesicle dynamics) which is abnormally phosphorylated, nitrated and oxidized, has an abnormal crystallographic structure and abnormal solubility, and is prone to the formation of aggregates and insoluble fibrils [16-20]. III) The degeneration of melanin pigmented brainstem nuclei because melanin production depends on the auto-oxidation of dopamine. Low melanin content could be due to reduced dopamine synthesis or more efficient transport to the nerve terminal. Consequently neuromelanin has been considered of potential importance in PD pathogenesis as it is capable of binding toxic compounds. Additionally, demelanisation of the substantia nigra (SN) in PD is believed to result from selective death of the more heavily pigmented neurons [21]. The nigral toxin MPTP (1-methyl-4-phenyl-1,2,3,6 tetrahydropyridine), which causes a Parkinsonian syndrome in primates, destroys those nigral neurons containing the most melanin pigment [22] thus providing a possible analogy with PD (Figure 5).
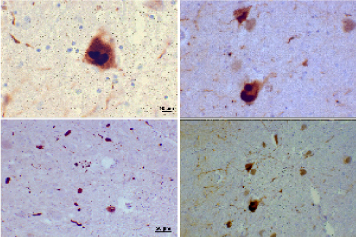 Figure 4: Photomicrographs of regions of substantia nigra in this Parkinson's patient show Lewy bodies and Lewy neurites in various magnifications.
View Figure 4
Figure 4: Photomicrographs of regions of substantia nigra in this Parkinson's patient show Lewy bodies and Lewy neurites in various magnifications.
View Figure 4
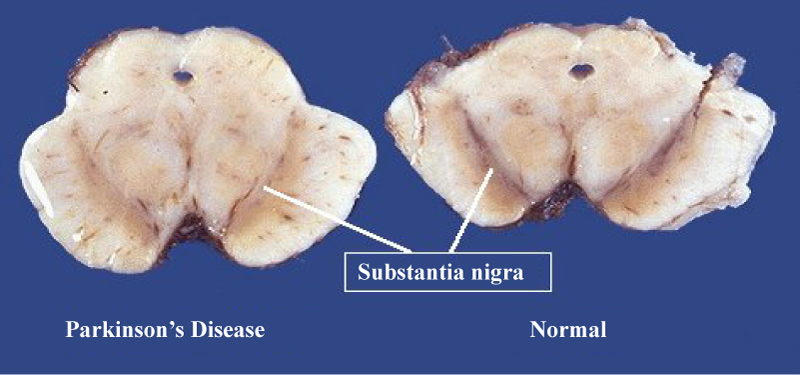 Figure 5: Left: Midbrain section showing loss of pigmented cells of the substantia nigra in Parkinson's disease. Right: Midbrain section showing normal substantia nigra. From: CNS Pathology.
View Figure 5
Figure 5: Left: Midbrain section showing loss of pigmented cells of the substantia nigra in Parkinson's disease. Right: Midbrain section showing normal substantia nigra. From: CNS Pathology.
View Figure 5
Top panels show a 60-times magnification of the alpha-synuclein intraneuronal inclusions aggregated to form Lewy bodies. The bottom panels are 20x magnification images that show strand-like Lewy neurites and rounded Lewy bodies of various sizes. Neuromelanin-laden cells of the substantia nigra are visible in the background. Stains used: Mouse monoclonal alpha-synuclein antibody; counterstained with Mayer's haematoxylin.
Cardinal manifestations: The onset of Parkinson's disease is often subtle and very gradual. There are however many hallmark signs of Parkinson's disease. These signs and symptoms are illustrated below in (Figure 6) and subsequently listed for more detail.
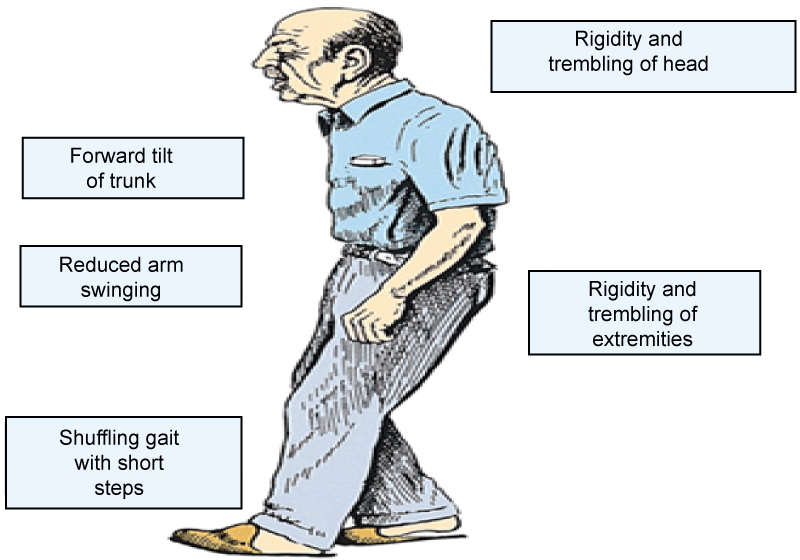 Figure 6: Classic presentation of Parkinson's disease.
View Figure 6
Figure 6: Classic presentation of Parkinson's disease.
View Figure 6
Common symptoms:
1. Tremor
a. Present when extremity/limb is at rest
b. Terminates with movement
c. Resting hand tremor is most characteristic (pill-rolling)
2. Bradykinesia
3. Rigidity of the extremities or trunk
4. Impaired posture and balance
5. Hypokinesia
a. May present itself in facial expression, decreased arm swing with gait
b. Leads to an increase in fall risk secondary to inability to generate adequate muscle force quickly and decreased ability to make postural adjustments/corrections in a timely fashion to avoid a fall
6. Visuoperceptive impairments
Secondary symptoms:
1. Micrographia
2. Decreased arm swing with gait
3. Foot drag resulting in shuffled gait
4. Freezing
5. Decrease in/loss of automatic movements (i.e. blinking, swallowing)
6. Cognitive impairment (present to some extent in up to 30% of individuals)
a. Memory loss
b. Impaired judgment
c. Poor planning
d. May preceed PD dementia
7. (Parkinson's) Dementia
a. Interferes with the following:
i. ability to plan
ii. ability to maintain goal orientation
iii. ability to make decisions
Other symptoms:
1. Depression
2. Pain
3. Anxiety
4. Psychosis (typically visual hallucinations)
5. Sleep disturbances
6. Autonomic dysfunction [23]
Disease progression:
Stage I
1. Signs and symptoms are unilateral
2. Mild tremor of a single limb
Stage II
1. Signs and symptoms are bilateral
2. Changes in posture and gait
3. Minimal disability noted overall
Stage III
1. Significant hypokinesia
2. Generalized dysfunction (moderately severe)
3. Deficits of equilibrium/balance affecting gait and standing
Stage IV
1. Ambulation limited
2. Rigidity
3. Bradykinesia
4. Unable to live alone
Stage V
1. Extreme weight loss
2. Unable to live without assistance/requires constant supervision
3. Cognitive deficits may be present and/or prominent (i.e. hallucinations and delusions) [24]
Medical and pharmacological treatments for Parkinson's disease are limited to the symptomatic relief of patients, and has failed to prevent or slow down the process of neurodegeneration. Due to the fact that dopamine deficiency is the main cause of PD, pharmacological treatments have been aimed at restoring the neurotransmitter levels of dopamine [25].
The underlying pathology in PD is a deficiency in dopamine in the basal ganglia so it was originally hypothesized that a substitution of dopamine would be an effective treatment [25]. However, dopamine does not cross the blood-brain barrier and as a result is ineffective as the majority of the drug is converted in the periphery. The immediate precursor to dopamine, dihydroxphenylalanine (levodopa) is able to cross the blood brain barrier through active transport. Once levodopa enters the brain it is transformed into dopamine by decarboxylation from the enzyme dopadecarboxylase. Levodopa is usually taken orally and is given a peripheral decarboxylase inhibitor such as carbidopa [26]. The inhibitor prevents levodopa from prematurely being converted to dopamine in the periphery before it reaches the brain. Any amount of levodopa that is converted to dopamine in the periphery will not be able to cross the blood brain barrier. Although levodopa therapy is considered the gold standard for PD treatment there are many side effects and difficulties with levodopa treatment.
Levodopa side effects: Levodopa has been associated with gastrointestinal problems, cardiovascular issues, dyskinesias, and diminished responses. One of the most serious problems in levodopa treatment is that the drug seems to lose its effectiveness if given for prolonged periods (continuously for 3-4 years) [27].
Dopamine agonists are also used in the treatment of PD. These drugs have a similar function to dopamine and are often used in conjunction with levadopa [25]. Bromocriptine (Parlodel), pergolide (Permax), and ropinirole (Requip) are a few dopamine agonists that are used to treat PD and do not have excessive adverse effects [26].
Dopamine agonist side effects: Dopamine agonists may produce nausea and vomiting and in some patients postural hypotension is also a problem. With prolonged use, these drugs can cause CNS-related side effects such as confusion and hallucinations [25].
Another form of pharmacological treatment for PD comes in the form of anticholinergic drugs. The deficiency of striatal dopamine causes excessive activity in certain cholinergic pathways in the basal ganglia so drugs that limit acetylcholine transmission have been found to alleviate PD symptoms including tremors and rigidity [25].
Anticholinergic side effects: Anticholinergics are associated with side effects including mood change, confusion, hallucinations, drowsiness, and cardiac irregularity. Other side effects are blurred vision, dry mouth, constipation, and urinary retention [26].
Amantadine: Amantadine is thought to work by blocking the N-methyl-D-aspartate (NMDA) receptor in the brain, thereby inhibiting the effects of excitatory amino acids such as glutamate. This is an evidence that excitatory neurotransmitters play a role in motor complications associated with advanced PD [25].
Amantadine side effects: The primary adverse side effects of Amantadine are orthostatic hypotension, CNS disturbance, and patches of skin discoloration on the lower extremities [25].
Selegiline: Selegiline (Deprenyl, Eldepryl) is a drug that strongly and selectively inhibits the monoamine oxidase type B (MAOB) enzyme. This enzyme is responsible for breaking down dopamine [25].
Selegiline side effects: Some side effects are dizziness, sedation, gastrointestinal distress, and headache [25].
Catechol-O-Methyltransferase inhibitors (COMT): COMT inhibitors work to prevent levodopa conversion in peripheral tissues, allowing more levodopa to reach the brain [25].
COMT side effects: The primary problem associated with COMT inhibitors is an initial increase in dyskinesias. Other side effects include nausea, diarrhea, dizziness, and muscle pain/cramps [25].
Neurotrophic factors (NTF): NTF are secreted proteins that play a critical role in the maturation and survival of neurons. Specific NTF act in the adult brain to support and protect mature neuronal populations. One NTF that has selective effects on dopaminergic neurons is glial cell line-derived neurotrophic factor (GDNF). GDNF has been shown to induce the dopamine synthetic enzyme, tyrosine hydoxylase, in fetal human rat cortical cultures. It has also been shown to promote the survival and differentiaton of dopaminergic neurons in vitro and protect these cells from the dopaminergic toxins [28].
Deep Brain Stimulation (DBS): One other approach to PD treatment being used today involves surgically implanting electrodes into deep brain structures such as the globus pallidus, thalamus, and subthalamic nucleus. High frequency stimulation of these structures may help normalize neuronal circuitry within the basal ganglia, and help resolve motor symptoms of PD [29].
At present, drug treatments and surgery can not fundamentally solve the problem. With the development of cell replacement therapy, medical professionals are exploring treatment by restoration of dopamine neurotransmitters in the neural circuits of patients with PD by getting dopaminergic neurons and cell transplantation.
Stem cells by definition have two essential properties, i.e. the capacity of self renewal, and the capacity to differentiate into different cell lineages. Under the right conditions, or given the right signals, stem cells can give rise (differentiate) to the many different cell types that make up the organism (Figure 7). Stem cell lineage determination is explained by several ideas, one among is focused on the stem cells microenvironment or 'niche'. A niche consists of signaling molecules, intercellular communication and the interaction between stem cells and their neighboring extracellular matrix.
 Figure 7: General hierarchy for the stem cell niche.
View Figure 7
Figure 7: General hierarchy for the stem cell niche.
View Figure 7
Stem cells are capable of performing three important functions with unique abilities: plasticity, homing and engraftment [30].
According potentiality of their differentiation: Potency is the potential of stem cells to develop into other cell types.
• Totipotent-the ability to differentiate into all possible cell types. Examples are the zygote formed at egg fertilization and the first few cells that result from the division of the zygote.
• Pluripotent-the ability to differentiate into almost all cell types. Examples include embryonic stem cells and cells that are derived from the mesoderm, endoderm, and ectoderm germ layers that are formed in the beginning stages of embryonic stem cell differentiation.
• Multipotent-the ability to differentiate into a closely related family of cells. Examples include hematopoietic (adult) stem cells that can become red and white blood cells or platelets.
• Oligopotent-the ability to differentiate into a few cells. Examples include (adult) lymphoid or myeloid stem cells.
• Unipotent-the ability to only produce cells of their own type, but have the property of self-renewal required to be labeled a stem cell. Examples include (adult) muscle stem cells [31].
According to the source of stem cells: Stem cells can be classified into four broad types based on their origin (Figure 8).
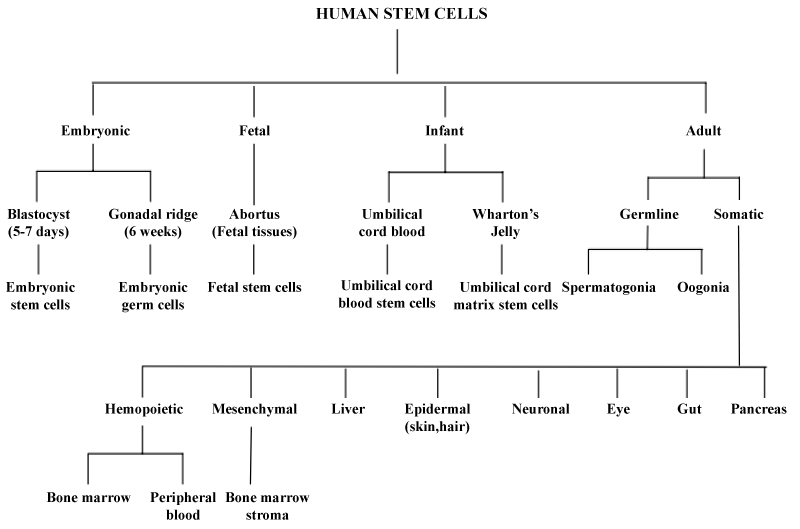 Figure 8: Classification of human stem cells.
View Figure 8
Figure 8: Classification of human stem cells.
View Figure 8
A) Stem cells from embryos, B) Stem cells from the fetus, C) Stem cells from the umbilical cord, D) Stem cells from the adult [30-32].
Stem cells have recently aroused a great deal of interest because of their potential to differentiate into dopamine neurons either by spontaneous differentiation or through certain induction protocols [33].
Embryonic stem cells (ES Cells): ES cells have attracted great attention as an alternative source for the generation of dopamine neurons because they can be continually expanded with high potential for differentiation. As they are pluripotent stem cells, they are able to form all three embryonic germ layer lineages following induced differentiation. Many studies have focused on optimizing the differentiation of ES cells into dopamine neurons. Among them, systematic and efficient induction systems for dopamine neurons have been reported by several groups [34-36], The prospect that ES cells can produce a sufficient number of dopamine neurons for transplantation therapy is particularly appealing, both for clinical and industrial use. At the same time; however, their clinical application is limited because of their ability to form tumors and the ethical problems surrounding the use of using fertilized human eggs to establish the ES cell lines [37].
Neural stem cell (NSCs): NSCs are an attractive source for cell replacement therapy for PD because they have the ability to differentiate into neurons, astrocytes, and oligo dendrocytes as well as into dopamine neurons [38]. NSCs can be isolated from different regions of the fetal brain as well as from the ventricular wall and the hippocampal dentate gyrus in the adult brain so that they can be harvested both from fetal and adult central nervous system tissues. As NSCs are able to self-renew, they can be maintained and expanded either as monolayers or as floating aggregates called neurospheres. Repeated expansion of NSCs is, however, reported to decrease their potential to differentiate into a variety of neuron subtypes, including dopamine neurons. In particular, adult NSCs have lower ability for differentiation than fetal NSCs [33]. Limitations in the use of NSCs include ethical and histocompatibility concerns for fetal NSCs and a limited supply of adult NSCs [39].
Induced pluripotent stem cells (IPS): Adult cells that have been genetically reprogrammed to an embryonic stem cell-like state by being forced to express genes and factors important for maintaining the defining properties of embryonic stem cells. Induced pluripotent stem (IPS) cells, whose properties are similar to those of ES cells, can be generated from adult human cells, such as dermal fibroblasts, by introducing genes such as Sox2, Oct3/4, Klf-4, and c-Myc [40]. They have thus attracted increasing attention as a new type of pluripotent stem cell without major ethical concerns. As is the case with ES cells, iPS cells can generate cells of all three germ layers and have unlimited proliferative activity, but their clinical application is limited by their tumorigenicity. IPS cells can be induced to form dopamine neurons, but the induction efficiency is generally lower than that of ES cells and the quality of the cells is not homogenous [40,41].
Mesenchymal stem cells (MSCs):
MSCs properties: MSCs are adult stem cells that belong to the mesodermal lineage and are traditionally found in the bone marrow together with haematopoietic stem cells which represents less than 0.05% of the total bone marrow but they have the potential to reconstitute all blood forming lineage [42]. MSCs can also be isolated from other mesenchymal tissues, such as umbilical cord, dermis, adipose tissue, and peripheral blood [43]. Morphologically, MSCs have long thin cell bodies with a large nucleus similar to fibroblasts Plastic adherence is a well-described property of MSC. As with some other tissue stem cells, MSCs have a high capacity for self-renewal while maintaining multipotency. In vitro derived MSCs express a panel of characteristics surface markers such as CD90, CD105, CD73, CD 29, and CD44 and are negative for hematopoietic markers as CD34, CD14 and CD45. The biologic property that most uniquely identifies MSC is their capacity for trilineage mesenchymal differentiation. In vitro MSCs differentiate into the cells of mesenchymal lineage such as bone, cartilage and adipose tissue [44]. Homing mechanism is an extraordinary ability of implanted MSCs cells to seek out the site of tissue damage [43]. MSCs secrete factors, including interleukin-6(IL-6), macrophage colony stimulating factor (M-CSF), interleukin-10(IL-10), hepatocyte growth factor (HGF), and prostaglandin-2 (PGE2), that promote tissue repair, stimulate proliferation and differentiation of endogenous tissue progenitors, and decrease inflammatory and immune reactions [44] (Figure 9).
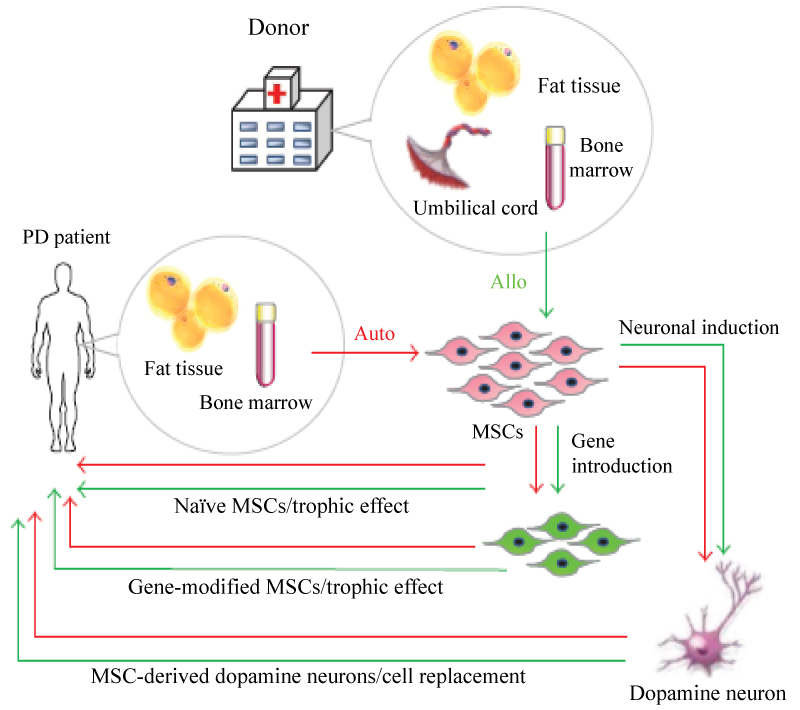 Figure 9: Strategy for MSC transplantation in PD patients.
View Figure 9
Figure 9: Strategy for MSC transplantation in PD patients.
View Figure 9
MSCs can be obtained from fat tissue or bone marrow aspirates of Parkinson's disease (PD) patients and are applicable for autocell transplantation. They can also be obtained from fat tissue, bone marrow aspirates, and umbilical cord of healthy donors for allocell transplantation. Naive MSCs can be directly transplanted into the striatum of PD patients, but this treatment exerts temporary trophic effects. Gene-introduced MSCs also have trophic effects for the replenishment of lost cells. MSCs are able to be induced into dopamine neurons that will contribute to the functional recovery of PD [44].
Among the many kinds of MSCs, bone marrow mesenchymal stem cell (BMSCs) are the most well studied. BMSCs can be cultivated from bone marrow aspirates as plastic adherent cells in vitro [45]. The great benefit of BMSCs is that they are easily accessible through aspiration of the patient's bone marrow, so that the use of BMSCs avoids ethical issues, facilitating their application both for auto- and allo-transplantation. BMSCs are also easily expanded on a large scale, which is very convenient for clinical use (e.g., 20 to 100 mL of bone marrow aspirate provides 107 BMSCs within several weeks) [46].
For cell-based therapy, MSCs have two major effects: a trophic effect that is mediated by the various types of trophic factors and cytokines produced by MSCs [47] and differentiation to generate a broad spectrum of cells for the replenishment of lost cells [48]. MSCs normally provide trophic factors to support hematopoietic stem cells in the bone marrow, thus their trophic effect is part of their normal function. MSCs are multipotent stem cells that are known to differentiate into osteocytes, chondrocytes, and adipocytes [45]. These differentiations are within the same mesodermal lineage, but recent reports demonstrated that MSCs show unorthodox differentiation into ectodermal and endodermal cells [49-53].
These findings stimulated the advancement of regenerative medicine aimed at the generation of desired cells from MSCs. To date, various cell types, such as mesodermal lineage cells (e.g., bone, cartilage, adipocytes, skeletal muscles, and cardiomyocytes), as well as endodermal lineage cells (e.g., airway epithelial cells, hepatocytes, and insulin-producing cells) and ectodermal lineage cells (e.g., neuronal cells and epidermal cells) have been induced from MSCs in vitro by the use of cytokines, trophic factors or gene introduction [53-58].
Naive Bone marrow mesenchymal stem cells (BMSCs): There have been many attempts to infuse BMSCs into a PD model aimed at ameliorating PD symptoms. BMSCs have trophic effects that are mediated by the various types of trophic factors and cytokines they produce. Therefore, naive adult BMSCs engrafted to the striatum induce partial but not drastic recovery of the dopamine pathway in a rat model of PD [59-63].
Findings from a human pilot study of autologous naive BMSC transplantation performed in PD patients and followed for up to 36 months indicated a certain degree of amelioration of symptoms with no tumor formation [64]. While BMSCs have advantages over some other stem cells regarding their safety, easy accessibility, and trophic effects. Naive BMSC transplantation has limitations for definitive care because most of the transplanted cells do not survive in vivo for a long time, and thus the trophic effects gradually decrease. In addition to naive BMSC transplantation, genetically modified BMSCs have been applied to the PD model. Cells genetically modified to produce L-DOPA or neurotrophic factors such as neurotrophins and glial cell line-derived neurotrophic factor (GDNF) are reported to be somewhat effective for the amelioration of PD symptoms [65-68].
For practical use, it would be more desirable to establish a specific system for inducing BMSCs to produce dopamine neurons prior to transplantation. There are several reports of the induction of dopamine neurons from BMSCs [66-68], but in these reports the effectiveness of the induced cells in vivo was not evaluated by transplanting them into a PD model. Another study reported that MSCs induced into immature neurons using basic fibroblast growth factor (bFGF), epidermal growth factor, platelet-derived growth factor, FGF-8, GDNF, or the reagents butylated hydroxyanisole and dibutyryl cAMP were transplanted into a PD model, but these immature neurons did not effectively ameliorate the PD symptoms [69,70]. In this manner, growth factor based methods allow MSC differentiation toward immature neuronal-like cells, but are not efficient in PD models.
Induction of functional dopamine neurons from BMSCs: A system to specifically induce dopamine neurons fro BMSCs was reported (Figure 10) [51]. This system first generates post mitotic functional neuronal cells with a very high efficiency without contamination by glial cells. The resulting neuronal cells are then further induced into dopamine neurons. The induction is achieved by lipofection of a plasmid containing a Notch1 intracellular domain (NICD) followed by the administration of a specific combination of trophic factors and cytokines [52,71]. BMSCs initially show little expression of markers for neuronal progenitor cells (NPCs). These findings suggest that the introduction of NICD into the cells induces BMSCs to acquire the characteristics of NPCs [51,72]. When NICD-introduced BMSCs are expanded and then stimulated with trophic factors (basic fibroblast growth factor (bFGF), for skolin, and ciliary neurotrophic factor (CNTF)) for several days, approximately 96% of the cells extend neurites and differentiate into post mitotic neuronal cells. These cells are positive for the neuronal markers MAP-2ab, neurofilament, suggesting that these induced cells are functional neuronal cells.
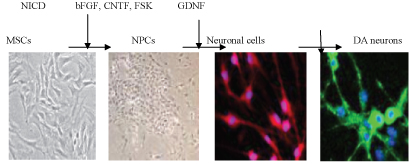 Figure 10: Production release of dopamine.
View Figure 10
Figure 10: Production release of dopamine.
View Figure 10
At this stage, cells positive for tyrosine hydroxylase (TH), a marker for dopamine neurons, accounted for ratios of only approximately 4%. After GDNF stimulation, the cells positive for TH substantially increased up to ̴ 60% Furthermore, other dopamine markers, were elevated in these TH-positive cells, Nurr-1 and Ptx3. The dopamine release upon depolarization in vitro measured by high-performance liquid chromatography indicated that the induced cells released dopamine into the culture media in response to high K+-depolarizing stimuli. These findings indicate that functional dopamine neurons can be efficiently induced from BMSCs [51].
(Figure10) Induction of dopamine neurons from MSCs. After NICD introduction, MSCs become similar to NPCs, expressing the NPC markers after cytokine stimulation (bFGF, CNTF and for skolin (FSK), cells become post mitotic neurons expressing neuronal marker. The administration of GDNF induces neurons to become dopamine neurons (TH), which are useful in the Parkinson's disease model.
Transplantation of BMSC-derived dopamine neurons into PD models: Induced dopamine neurons (1 × 105 cells) from either rodent or human (under the control of immunosuppressant) BMSCs were transplanted into the striatum of a PD model rat, after intracerebral stem cell application Grafted dopamine neurons migrated and extended beyond the injected site, and approximately 30% of the cells remained in the striatum 10 weeks after transplantation. The grafted striatum showed the migration of transplanted cells that expressed neurofilament and TH. Brain slice culture experiments demonstrated the production of dopamine in the transplanted brains. MSCs induced increased neuronal plasticity (neurorescue) with increased modulation of cell survival (and an increased striatal dopamine level), enhanced neurogenesis (progenitor cells), and a decreased modulation of inflammation, gliosis and death signaling [73,74]. No tumor formation was observed in the brain, demonstrating that dopamine neurons induced from BMSCs do not have the ability to form tumors [51].
It could be concluded that Cell transplantation is a strategy with great potential for the treatment of Parkinson's disease, and many types of stem cells, including neural stem cells and embryonic stem cells, are considered candidates for transplantation therapy [75-79]. Mesenchymal stem cells are a great therapeutic cell source because they are easy accessible and can be expanded from patients or donor mesenchymal tissues without posing serious ethical and technical problems. They have trophic effects for protecting damaged tissues as well as differentiation ability to generate a broad spectrum of cells, including dopamine neurons, which contribute to the replenishment of lost cells in Parkinson's disease.
MSCs provide strong possibilities for clinical application, because they are easily accessible cells with few ethical problems and can be efficiently expanded in vitro to achieve therapeutic scale. Importantly, MSCs are already widely used clinically to treat osteoarthritis and myocardial infarction, so they have an established record in clinical applications. Furthermore, they are easily obtained from patients or marrow banks, autologous transplantation, or transplantation with the same HLA subtype from a healthy donor, which may minimize the risks of rejection.