Kidney is considered as the secondary target organ of nanoparticle (NP) toxicity. Since it is the primary organ of excretion, NPs are expected to adversely affect the renal system. Therefore, a comprehensive review of recent knowledge on renal toxicity of engineered nanoparticles (ENPs) was made. Mechanistic paradigms of their toxicity have also been discussed.
In vitro and in vivo studies indicated that carbon nanotubes (CNT) caused cytotoxicity, glomerular degeneration and proximal tubular necrosis. Salient feature of their toxicity was the accumulation of hyaline like substances in the renal tissue. Fullerenes caused mitophagy, cytoskeletal changes and cell death, however, their pro-oxidant nature had not been established.
Amongst metal oxide NPs, silver nanoparticles (AgNPs) could induce mitochondrial damage, loss of brush border membranes and inflammation of podocytes. These effects were attributed to "neprotic syndrome and "minimal change disease". Gold (AuNPs) and platinum nanoparticles (PtNPs) also affected renal function. Vacuolar degeneration, cloudy swelling and hyaline deposits were recorded in the cortex of AgNPs treated rats. Cadmium sulphide nanoparticles (CdSNPs) have been considered as potent renal toxins. However, their differential effects were observed in specific cell lines and animal models. Coating of CdSNPs also affected their renal toxicity. Zinc oxide nanoparticles (ZnONPs) induced oxidative damage and genotoxicity. Polytoxic events contributed to renal toxicity of copper nanoparticles (CuNPs). Massive necrobiosis was also observed. Coating of iron oxide nanoparticles (IONPs) also influenced their toxicity. Glomerular amyloidosis was witnessed in silica nanoparticle (SiNP) treated rats. Titanium oxide nanoparticles caused glomerualar, interstitial and tubular changes in the kidney. These changes could be reversed after the treatment with antioxidants i.e. lycopene and quercetin.
In general, these reports indicated that ENPs manifested toxicity through membrane damage; oxidative stress; mitochondrial injury; cytoskeletal changes, apoptosis and necrosis. Smaller particles caused greater toxicity than their larger counterparts. Species differences in their renal effects were also recorded. However, further studies on different cell types and mechanisms like autophagy, ER stress and reductive stress have been suggested prior to their selective pharmaco-therapeutic applications.
Engineered nanoparticles, Renal toxicity, Carbon nanotubes, Fullerenes, Metallic nanoparticles, Oxidative stress and apoptosis
Recent discoveries in nanotechnology have introduced nanomaterials/nanoparticles (NMs and NPs) to agriculture, industry and commercial sectors. These particles/structures/devices possess a size or shape restricted to nanometer level [1]. The increased use of NPs in agriculture, industry, electronics, cosmetics, medicine and drug delivery etc. has raised concerns on their environmental and health effects [2]. Therefore, an assessment of their toxicity becomes mandatory from public health point of view. Skin, pulmonary and reticulo-endothelial system (RES) including liver and spleen have been identified as the main target organs. Beside RES, secondary target organs viz. kidney, heart, bone marrow and central nervous system are also considered vulnerable to their toxic effects [3,4]. NPs can translocate across the plasma membrane through endocytosis and get deposited in primary or secondary organs [5]. Physicochemical properties such as shape; size; composition; surface charge; surface chemistry; solubility; roughness, rigidity and elasticity are known to influence their uptake and bioaccumulation in different organs [6-8]. Nonetheless, much is not know on their specific cellular and subcellular effects in kidney.
Kidney in mammalian species performs various functions. Not only does it excrete metabolic end products but regulates the synthesis and release of hormones i.e. renin and erythropoietin. Functional unit of kidney is a nephron that maintains fluid homeostasis, osmoregulation and waste filtration. A single nephron consists of two functional units i.e. the glomerulus and a hairpin shaped tubule composed of a proximal tubule; the loop of Henle, a distal tubule and collecting duct. Each unit of nephron comprises specific cells. The glomerulus is lined with glomerular endothelial cells (GEC); a glomerular basement membrane (GBM); podocytes, mesengial cells and parietal epithelial cells [9]. Effects of different NPs on these cellular components of nephron need to be established (Figure 1).
 Figure 1: Cellular components of glomerulus.
View Figure 1
Figure 1: Cellular components of glomerulus.
View Figure 1
The proximal tubule consists of three segments, the S1 (pars convoluta), S2 (transition between pars convoluta and pars recta) and the last S3 (pars recta). It reabsorbs virtually all the filtered low molecular weight proteins by specific endocytic mechanisms. Two cellular carriers are involved in this process: The capillary endothelial cells at the basolateral side of the proximal tubular cells and the tubulo-interstitium between the capillaries and the tubular cells [10]. Renal peritubular capillaries have fenestrations (60-70 nm wide) covered by a diaphragm (3-5 nm thick). In order to cross the tubulointerstitium, nano-particles need to be smaller than the size of the diaphragm (< 5 nm). They ought to be positively charged as the fenestrae are negatively charged due to the presence of heparin sulphate. Moreover, the tubulointerstitium does contain fibroblasts and dendritic cells within extracellular matrix consisting of proteoglycans, glycoproteins, fibrils and interstitial fluid. Tubular epithelium being highly susceptible to NPs may face chemical threats leading to cell injury, finally manifesting into cell death [11].
Although, sufficient data are available on the effects of NPs on primary target organs i.e. RES, mechanistic paradigms viz. cytototoxicity; oxidative stress; mitochondrial dysfunction, cytoskeletal effects and DNA damage in renal tissue are poorly understood. Present review discusses recent information available on the toxicity of different NPs i.e. carbon nanotubes, fullerenes, metal oxide nanoparticles and quantum dots on renal system in different cell and animal models.
Carbon nanotubes were first described by Iijima [12]. They are further classified as single walled (SWCNT), double walled (DWCNT) and multiwalled (MWCNT) CNTs. They possess unique properties like strength; hardness; thermal conductivity; micro-wave absorption, electrical and catalytic properties [13]. They can be manipulated for specific applications in electronic devices, waste water treatment and drug delivery systems [14,15].
In vitro studies: Limited information is available on the renal toxicity of CNTs. A few workers have studied their effects using different cell lines. Cell mortality in NRK-52E proximal tubular cells was reported after exposure to increased concentration (0.25-100 µg) of MWCNTs [16]. Results on lactate dehydrogenase and MTT assay indicated membrane damage and mitochondrial injury. Genotoxic effects of CNTs were also confirmed by the same group of workers. Cell viability and cytotoxicity in human embryonic kidney cell lines (HEK-293) treated for 48 h with different sized MWCNTs and at concentration ranging from 3-300 µg/ml was reported by Reddy, et al. [17]. They attributed these effects to cell membrane damage, inflammation and oxidative stress. Interestingly, experiments made on another cell line - mouse kidney cortical collecting duct clone 4 (mpkCCDcl4) at a concentration of 0.001-100 µg/ml of SWCNTs and MWCNT could not affect cell viability. It was also hypothesized that lower concentration of CNTs expressed severe biological effects. CNTs at higher concentration form aggregates that act like microparticles while at low concentration they behave like nanoparticles [18]. Similar observations have been made in HK-2 cells at a concentration of 0.5-256 µg/ml and MWCNT were found to be non toxic. CNTs were also found to inhibit cell growth. Solubilised SWCNT (0.125-10 µg/ml) dispersed in water with SDS arrested cell growth at G0/G1 phase of normal rat kidney epithelial cells (NRK-52E). DNA damage was attributed to p53 dependent signalling pathway [19].
In vivo studies: Bio-distribution of CNTs in kidney of experimental animals is also poorly known. Pristine and functionalized CNTs could remain in the lungs for a month or even a year [20]. This study showed that if cleared, they move to gastrointestinal tract through mucociliary escalator. Thereafter, a fraction can translocate to other organs viz. liver, kidney and spleen. Injected CNTs have a short biological half life of minutes to hours. Attempts were made to delineate differences if any between the toxicity of pristine and functionalized NPs in the kidney. In a study, MWCNT functionalized with carboxylic group even at a low dose (2.5 mg/kg) after 20 days of injection (< 8 nm) caused significant changes in the kidney of Wistar rat. The lesions included inflammation in cortex and medulla, accumulation of hyaline like substances, glomerular degeneration and proximal tubular necrosis in a dose dependent manner [21]. Intra-tracheally installed pristine MWCNT (1 mg/kg for 30 days) induced nephrotoxicity in rat whereas those treated with MWCNT and PEG showed no toxic effects. Renal tissue damage was exhibited as collapsed glomeruli, packed mesengial and endothelial cells and apoptosis [22]. Thus it could be concluded that vectors might influence the toxic endpoints of CNTs.
Mechanistic paradigm: On the basis of available information, it could be concluded that CNTs even at low concentration are potent renal toxins. In vitro studies made on different human and animal cell lines indicated cytotoxicity, cell membrane damage, reduced cell growth, inflammation, oxidative stress and DNA damage. In vivo studies made in rat kidney showed glomerular and tubular injury. Mechanisms of toxicity involved ROS generation, protein expression and genotoxicity. Further studies on their effects on renal cells i.e. podocytes, mesengial cells, basement membrane and parietal cells are needed (Table 1).
Table 1: Summary of mechanisms involved in renal toxicity of nanoparticles. View Table 1
Fullerenes are the molecules with 60 or 70 atoms of carbon denoted as C60 or C70. They were first discovered by Kroto, et al. [23]. In structure, they resemble a soccer ball and possess a three dimensional structure with unique physical and chemical properties [24]. They are lipophilic in nature and can cross membrane and blood brain barrier [25].
In vitro studies: In vitro study on fullerenol, a hydroxylated derivative of fullerene on procine kidney LLC-PK1 epithelial cells exposed for 24 and 48h (0.32-95, 808 µg/ml) showed cytotoxicity. It was mainly attributed to cytoskeletal effects, mitophagy, ATP depletion and cell death [26]. Another study was made by Blazer-Yost, et al. [18] who observed a significant decrease in trans epithelial resistance (TEER) in mpkCCDcl14 cell line. Their pro-oxidant nature is yet to be established.
In vivo studies on these NPs are not available.
Further studies are needed to understand the cellular and molecular mechanisms of fullerene toxicity. However, cytoskeltal changes and mitophagy may play a crucial role in their renal toxicity.
Silver nanoparticles are extensively used in cosmetics, textiles, surgical prostheses and antimicrobial activities. Approximately 450 consumer products are known to contain AgNPs. Therefore, it becomes imperative to study their adverse health effects, if any using suitable test models.
In vitro studies: In recent years, experimental evidence has gathered on their uptake and bioaccumulation in soft tissues. In vitro study showed that internalization of AgNPs in procine kidney (PK15 cells) was facilitated by endocytosis. Silver concentration in cells exposed to 50 mg/ml of AgNP and 50 mg/ml of Ag varied in 106 cells. Although viability of cells was affected by both the particles, bioconcentration was higher for nanoparticles than microparticles [27]. It was also demonstrated by Recordati, et al. [28] that smaller particles (10 nm) induced greater toxicity than the larger particles (40-100 nm) in mice.
In vivo studies: Though several reports are available on renal toxicity of AgNPs in mice and rat, adverse effects in the kidney of an African cat fish, Clarius gariepinus have also been reported. Histopathological observations made in the kidney of fish treated with 10 -100 µg/L (20-40 nm) AgNP showed tubular as well as glomerular changes. These included glomerular hypertrophy, hydropic degeneration, dissociation of renal tubules and necrosis. Most of these changes were restored after a recovery period of 15 days [29]. Acute effects in the kidney of rat injected with 2000 mg/kg AgNPs were recorded by Hussein [30]. Ultrastructural changes in the kidney viz. inflammation of renal epithelium, thickening of the basement membrane and cytoplasmic vacuolation were observed. Podocytes showed elongation of pedicels. Kidney stem cells (mKSCs) were also used as a model to examine the effects of AgNPs in rat. It affected podocyte differentiation. Proximal tubular cell markers showed differentiation of a specific lineage. It was concluded that AgNPs stimulate mKSCs differentiation [31].
Mitochondrial damage, loss of brush border membranes, inflammed podocytes and degeneration of their pedicels were observed in the kidney of Wistar rat after exposure to 50 ppm and 200 ppm of AgNPs for 60 days [32]. These changes were correlated to Neprotic syndrome and "Minimal Change Disease". Prolonged treatments led to cell proliferation and disturbances in normal apoptotic pathways. Finally, it has been concluded that AgNPs are capable of inducing multi-organ toxicity in mice [33].
Mechanistic paradigm: These results suggest that AgNPs are potent inducers of renal cellular damage. They may disrupt mitochondrial function, damage brush border membranes and cause apoptosis in renal cells.
Gold particles are now being increasingly used in drug delivery, cellular labelling, imaging and diagnosis of diseases like cancer, diabetes and Alzheimer. However, its adverse health effects are poorly known. Important applications of platinum particles include catalysis, cosmetics and dietary supplements.
In vitro studies: These NPs were able to penetrate cells when incubated with renal cell sediment [34]. The uptake of GNPs by renal proximal tubular cell line-TH1 was studied by Samkova, et al. [35] through TEM. Bioaccumulation was determined by ICP. GNP nanourchins could cause molecular changes in He La and proximal tubular cells [36]. These workers recorded endoplasmic reticulum stress and reductive stress and changes in RNA and protein homeostasis.
In vivo studies: A detailed study on GNP toxicity in Kyoto Wistar rats was made by Abdelhalim and co-authors [37]. These workers noted several lesions in rat kidney viz. vacuolar degeneration, cloudy swelling, hyaline droplets, and casts in the proximal tubular cells. Proximal tubules were more affected than distal tubules. Glomerular changes included hyper cellularity, mesengial cell proliferation and basement membrane thickening. These effects were attributed to ROS and diminished antioxidant mechanisms. An injection of 18 nm GNP to golden hamsters also caused renal tubular necrosis [38]. Like other nanoparticles, shape, size and labelling of GNPs affected the toxicity in renal cells. GNPs varying in size (13 nm to 60 nm), possessing different shapes (spheres and stars) and labelled with 11-mercaptoundecanoic acid (MUA) or with sodium citrate affected human renal HK-2 cells [39]. These workers incubated HK-2 cells with GNPs and recorded cytotoxicity, loss of lysosomal integrity, changes in mitochondrial membrane potential, lipid peroxidation, GSH and ATP status. It was observed that smaller NPs were more toxic. 60 nm nanospheres and nanostars were less toxic. Similar observations have been made for platinum nanoparticles. SnPt of less than 1 nm could induce nephrotoxicity and disrupted renal function. Intriguingly, these effects were not observed in mice treated with 8 nm NPs [40].
Mechanistic paradigm: Internalization and excretion dynamics of GNPs have been studied in various cancer cell lines [41]. Renal toxicity is caused by endoplasmic reticulum stress, changes in RNA and protein homeostasis, oxido-reductive stress and mitochondrial changes. However, such studies on renal cancer cells are still awaited. Limited information is available on platinum nanoparticles.
Cadmium sulphide nanoparticles are frequently used to produce hybrid solar cell, semiconductors, Ni-Cd batteries, metal alloys and coatings, fluoresecent imaging and probe stabilizers.
In vitro studies: Cadmium ions possess special affinity with renal tissue. Cadmium based quantum dots (QDs) have attracted attention of a large number of workers. Human kidney cells were found to be susceptible to CdNPs. Its cytotoxicity in IP15 (mesengial) and HK-2 (proximal epithelial) cell lines was studied by Pujalte and colleagues [42]. Cell mortality was noted in a dose dependent manner. These effects were attributed to ROS and oxidative stress. Coating of QDs was also found to affect their toxicity. Cd QDs coated with zinc sulphide (5.1 nm) induced toxicity in LLC-PK1 cells [43]. Intriguingly, mouse renal adenocarcinoma (RAG) cell line treated with QDs containing CdSe core shell of ZnS (1.5 µg/ml) for 2, 6, 24 h enhanced the production of ROS [44]. A comparative study on the effects of QDs with core shell CdSe@ZnS coated with polymerized histidine formaldehyde (pHF) made in procine and human renal proximal tubule cells showed differences in their bioaccumulation. It was suggested that human proximal cells were more susceptible than procine cells [45].
In vivo studies: Exposure of female ICR mice to CdONP for 6 weeks by inhalation produced multiorgan effects including renal fibrosis [46]. Experiments performed in our own laboratory by feeding rats (10 mg/kg b.w.) on CdSNPs (5-9 nm) on each alternate day for 45 days revealed extensive damage in proximal tubular cells. Ultrastructural observations showed nuclear, mitochondrial and ER lesions [47].
Mechanistic paradigm: Nanoparticles of cadmium induced renal toxicity through ROS and oxidative stress. Consequently, genotoxicity and ER stress have also been observed.
Zinc oxide nanoparticles are also used in various consumer products. These include antimicrobial and antifungal agents, paints, textiles, plastics, glass, ceramics, rubber, cosmetics and food products. However, their potential effects on renal tissue remain largely unknown.
Nanoparticles of zinc can accumulate renal tissue and affect its structure and function. Metabolic profile studied in rat kidney treated with different doses of ZnONPs viz. 100, 300 and 1000 mg/kg for 14 days demonstrated significant increase in the values of taurine, lactate, acetate, creatinine, phosphocholine and hippuric acid in the urine samples [48]. These workers concluded that ZnONPs could disturb the energy balance and cause mitochondrial and membrane changes in rat kidney.
In vitro studies on its cytotoxicity/genotoxicity have also been made. Uzar and co-workers [49] exposed rat kidney epithelial cells (NRK -52E) to ZnONPs of size ranging from 10-15 nm and different concentrations (25-100 µg/ml). Significant damage to DNA was recorded through comet assay. Another study made on podocytes of rat kidney showed a decrease in cell viability after exposure to 10, 50, 100 µg/ml ZnONPs. Observations made using flow cytometry indicated an increase in intracellular accumulation and consequent generation of ROS leading to apotosis [50]. Pretreatment of podocytes with a ROS inhibitor N-mercaptopropionyl glycine inhibited apoptosis caused by ZnONPs. These workers attributed these changes to oxidative stress. Role of oxidative DNA damage triggered by Zn2+ ions was also discussed by Scherzad, et al. [51]. Human embryonic kidney cells were affected in a dose dependent manner. TC50 determined through trypan blue exclusion method confirmed its effect on growth of these cells.
In vivo studies: Comparatively very few in vivo studies have been made to determine ZnONP induced nephrotoxicity in animal models. A study made on three concentrations (5, 50, 300 µg/kg) in Wistar rat yielded interesting results. Low dose caused more severe histopathological changes than a higher dose. Similar results were obtained on apoptotic index also [52]. Several workers agree that lower doses of NPs are more toxic than higher doses.
Mechanistic paradigm: Based on available information, it could be concluded that ZnONPs are potent nephrotoxins. However, cellular toxicity has been attributed to membrane damage, mitochondrial injury, oxidative stress and apoptosis.
Copper nanoparticles are frequently used in antibiotics, antimicrobial and antifungal agents. They are also employed in conductive inks and pastes and as substitute to metals in electronic displays and transmissive thin film applications.
In vivo studies: Though copper toxicity is very well known, health safety issues related to copper nanoparticles have been poorly addressed. It affects renal structure and function in a dose dependent manner. Glomerulonephritis and degeneration of tubular epithelium were observed in ICR mice exposed to 232, 341, and 1080 mg/kg CuNPs. Highest dose caused massive necrobiosis [53]. Contrariliy, a low dose (70 mg/kg) also caused glomerulonephritis and renal tubular necrosis in the same animal model [54]. This study suggested greater renal toxicity of CuNPs at a lower dose than higher dose. Similar conclusion has been drawn by other workers also for different NPs. Metabolomic studies made earlier attributed CuNP induced renal toxicity to mitochondrial dysfunction, enhanced ketogenesis, fatty acid β-oxidation and glycolysis [55]. Histopathological studies made in Swiss mice after treatment with 200, 413 and 600 mg/kg b.w. of CuNPs showed glomerular and epithelial damage [56]. These effects were attributed to oxidative and nitrosative stress responsive signalling pathways. Gene expression changes specially those involved in metabolism, oxidative phosphorylation, cell cycle, mitogen activated protein kinases (MAPK) signalling and glutathione metabolism have also been studied in Wistar rat fed for 5 consecutive days on 100 and 200 mg/kg CuNPs [57]. Higher dose caused severe proximal tubular necrosis that was wanting in rats treated with a low dose.
A few studies on its toxicity using alternate animal models have also been made. Renal tubular necrosis was recorded in rainbow trout (Onchorhynchus mykiss) after exposure to 20 or 100 mg/L CuNPs [58]. An in vitro study was made on renal epithelial (A6) cells of frog (Xenopus laevis) to study mechanisms of CuNP induced renal toxicity [59]. They attributed the effects to polytoxic events viz. cellular uptake, endocytosis, ROS generation, DNA damage and apoptosis.
Micro and nanoparticles of copper are known to accumulate in renal tissue. However, differences in their bioconcentration were investigated in a study made in rats fed on equivalent dose of 500 mg/kg of copper micro and nanoparticles. Kinetics revealed higher concentration of copper in the kidney of CuNPs treated rats than bulk copper treated rats [60]. Renal toxicity caused by different forms of nanoparticles viz. copper nanoparticles and copper carbonate nanoparticles has been studied in male rats [61]. Histopathological observations showed severe organ damage by copper carbonate NPs than CuNPs.
In vitro studies: A few in vitro studies to analyze the toxicity of CuNPs on renal cells have also been made. CuONPs (34.9 nm) caused significant cytotoxicity (IC50, 11.6-16.4 µg/ml), genotoxicity (1-9-8.4 fold), oxidative stress and apoptosis in the range of 2.5-80 µg/ml [62]. Coating o CuNPs also influenced its retention and clearance in renal tissue. A novel study was made by Yang and co-workers [63] who showed that clearance and degeneration of glutathione coated CuNPs was faster than the glutathione sulphide complexes.
Mechanistic paradigm: In nut shell, the mechanisms that account for CuNP induced renal toxicity include ROS, RNS, oxidative stress, DNA damage and apoptosis. Its lower dose is more toxic than higher dose. Further, bulk particles are less toxic than nanoparticles. Specific effects on mesengial cells and podocytes remain to be studied.
Magnetic iron oxide nanoparticles (IONPs) are now increasingly used in drug delivery, magnetic resonance imaging (MRI), thermal ablation therapy, in vivo cell tracking and separation of cells and molecules.
Assessment of toxicity, if any implicated by these NPs thus becomes an important subject from health risk assessment point of view. Available literature shows that very few in vitro and in vivo studies have been undertaken to describe their health effects. Alike other NPs, IONPs are adsorbed by the blood through opsonisation, followed by their uptake and elimination. These processes are influenced by the size of the NPs. A few reports indicate that IONPs > 100 nm are rapidly trapped by liver and spleen whereas those < 10 nm are subjected to renal clearance.
In vitro studies: In vitro study on cellular uptake and cytotoxicity was made in macrophages and cancer cells using commercially available IONPs of different sizes and coatings. An in vitro study made in BALB/C mice by the same group of workers discovered that polyethylenimine (PEI) coated IONPs exhibited higher uptake and cytotoxicity than PEGylated NPs. These effects were attributed to ROS and apoptosis [64].
Damage caused by IONPs to renal tissue has been studied by a few laboratories. IONPs (5-11 nm) administerd to mice for 7, 15, 30 days affected renal function and induced histopathological changes. A multi-organ study made in male mice also showed renal tissue damage after the treatment with 25 and 50 mg/kg for four days [65].
In vitro studies made in MDCK and LLC-PK epithelial cells showed depletion of thiols caused by uncoated and oleic acid coated IONPs. The study further concluded that expression of transferring receptor/CD 71 also decreased in these cells [66]. It has been realized by several workers that coating of IONPs can be an important factor in its toxic manifestations. Super paramagnetic polyacrylic coated IONPs infused in BALB/C mice could accumulate in proximal tubular epithelium, podocytes and mesengial cells. These observations were confirmed using magnetic resonance imaging and transmission electron microscopy. However, no renal function impairment was noticed [67]. Similar study made on silica coated IONPs in human HK-2 epithelial cells showed cell growth inhibition in a dose dependent manner [68].
In vivo studies: It was considered worthy to find out a biologically safe range of IONPs for medical use. With this objective Ma, et al. [69] exposed Kumming mice to IONPS at dose range of 0, 5, 10, 20 and 40 mg/kg. Biomarkers of toxicity viz. ROS, GSH, MDA, protein cross links and 8-OHdG were analysed. These workers have recommended a safest dose of 5 mg/kg body weight. However, further studies are needed to validate renal toxicity of IONPs.
Mechanistic paradigm: Iron overload is known to cause several diseases in man. IONPs manifest their toxicity through ROS, oxidative stress and DNA damage. Comparative studies between IONPs and iron ions need to be made. Moreover, its effects on the structure and function of renal cells need to be explored further.
Various consumer products viz. drugs, cosmetics, printer toners, varnishes and food items are known to contain silica nanoparticles (SNP). Diagnostic biosensors for glucose, lactate, L-glutamate and hypoxanthine also use SNPs [70]. Processes like leukemia, cell differentiation [71], cancer therapy [72], drug delivery [73] and enzyme immobilization [74] are also studied through SNP based biomarkers.
Two excellent reviews on their biological and harmful effects are available in literature. Napierska, et al. [75] for the first time discussed in vitro and in vivo toxicity of crystalline and amorphous silica and described their uptake, size and dose dependent toxicity, potential to generate ROS and inflammatory changes. Researches made on SNPs after 2010 have also been discussed [76]. These workers drew general conclusions that their acute toxicity was severe than chronic toxicity. Physico-chemical properties determined their toxicokinetic behaviour.
In vitro studies: Renal toxicity of SNPs seems to be least understood. However, the results of available studies are presented in this article. Cyto-toxicological information available on human embryonic kidney (HEK-293) cells exposed to SNPs (20-50 nm) reveals that it manifests its toxicity through oxidative stress [77]. Another in vitro study on human HK-2 cells and procine LLC-PK1 cells confirmed NP internalization and consequent localization in vesicles. Size and duration of exposure to SNPs influenced their toxicity [78].
In vivo studies: Histopathological studies in rodent models have also shown renal effects of SNPs. A peculiar condition of glomerular amyloidosis along with renal tubular necrosis was recorded in mice after chronic oral exposure (200 nm) to amorphous silica [79]. Renal interstitial fibrosis was another lesion observed in BALB/C mice intraperitoneally injected mesosporous silica nanoparticles (MSN) at a concentration of 150, 300 and 600 mg/kg for 2 and 12 days. Value of fibrosis markers i.e. NFkB p65 also increased in renal homogenates treated with MSN. These results were supported by in vitro studies made in NRK-52 cells exposed to MSN [80]. Only one study is available on the renal toxicity of cadmium doped SNPs. Coccini, et al. [81] observed condensed/collapsed glomerulii, packed mesengial and endothelial cells after 7 and 30 days of exposure to SNPs. Toxicogenomic studies, however, did not support these histopathological changes. Dose dependent morphological changes in kidney of rat were observed after exposure to 25, 50, 100 and 200 mg/kg MSN for 30 days [82]. In this novel study, role of ROS and TLR4/MYD88/NRFkB, JAK2/STAT3/ PPARy and Nrf2/ ARE/HO-1 signalling pathways in mSN induced nephrotoxicity was investigated. Briefly, these observations are expected to be helpful in planning safe use of SNPs.
Mechanistic paradigm: The above paragraphs clearly show that renal toxicity of SNPs is poorly understood. Glomerular amyloidosis was a peculiar lesion induced by SNPs. Inflammation, oxidative stress and changes in intracellular signalling pathways are involved in renal toxicity of SNPs. Further studies are needed on its effects on renal cells.
These particles are ubiquitous now. They are used in the production of several consumer products viz. paints, paper, cosmetics, toothpastes and pharmaceutical agents. Medical applications include antimicrobial drugs, skin care and photodynamic therapy.
Interesting research has been carried out on its pharmacological effects. A few workers from Poland reviewed its photosensitizing potential [83]. It was demonstrated that in the presence of UV light TiO2NPS produce ROS. These species contribute to cell death and thus offer protection against psoriasis and cancer. Thus TiO2NPs in combination with other molecules may work as photosensitizing agents in photodynamic therapy.
In vitro studies: Renal toxicity of TiO2NPs in different cell and animal models has been studied by a few workers. It expressed specific effects on different cell lines. Cytotoxic profile was found to be higher in LLC-PK1 cells than IP15 cells. ROS level was enhanced in both the cells, however, internalization was controlled by their size. TEM results confirmed their localization in vesicles [84]. Increase in ROS was recorded in other cell line NRK-52E also after exposing them to 20 µg/ml TiO2NPs for 24, 48, 72 and 96h [85]. The relative number of mitoses decreased while an increase in apoptotic cells was observed.
In vivo studies: In vivo studies on its renal toxicity have been made in rat, mice as well as fish. Kidney of male rats intra-peritoneally injected with 30, 50, 70 mg/kg TiO2NPs showed several lesions viz. deposition of hyaline like material, inflammation of Bowmans' capsule and tubular degeneration [86]. Another study from Al-Doaiss, et al. [87] also showed histopathological changes i.e. glomerular, tubular and interstitial lesions, hyaline casts and fibrosis in rats treated with different doses of TiO2NPs (126,252,378 mg/kg b.w.) for 24 and 48 h. Dose dependent effects of NPs were observed. A metabonomic study made in rats exposed to different doses of TiO2NPs for 4 days, 1 month and 2 months indicated variations in morphological and physiological parameters in renal tissue of rat [88]. Functional changes were more prominent at higher doses but metabonomic changes were conspicuous even at the lowest dose.
Therapeutic reversal of these effects has also been studied by a few workers. Effects of lycopene and quercetin were monitored in rats pre-treated with TiO2NPs. Altayeb, et al. [89] reported that lycopene (10 mg/kg) administered through gastric tube to rats treated with 150 mg/kg TiO2NPs, ameliorated its renal toxicity. Not only the tubular degeneration was wanting, immunohistochemical studies on desmin, anti-proliferating cell nuclear antigen (PCNA) and caspase-3 also indicated protective effects. Quercetin also protected rats against renal toxicity of TiO2NPs. Observations made on renal proximal tubules showed diminished values for malondialdehyde, catalase, super oxide dismutase and reduced apoptosis [90,91].
Mechanistic paradigm: Mechanisms responsible for TiO2NPs induced renal toxicity remain unknown at present. However, protective effects expressed by certain antioxidants viz. quercetin and lycopene suggest involvement of oxidative stress related processes in its toxicity.
It has been established now that kidney is a major secondary target organ for NP toxicity. In vitro and in vivo studies reviewed in this article confirmed their cytotoxicity in various cell types. Briefly, NPs are adsorbed, internalized, circulated and distributed in renal system depending upon their physicochemical properties [92]. Distinct blood proteins can adsorb to NP surface forming a protein corona. Protein corona can facilitate their elimination or allow their sustained presence in systemic circulation. Thus a specific and new biological identity is bestowed upon NPs [93,94].
A few studies included in this review describe effects of NPs on glomerular endothelial cells (GECs), glomerular basement membrane (GBM), podocytes and mesengial cells. For example, MWCNT can cause glomerular degeneration [21], while AgNPs induce glomerular atrophy in fish [29]. Elongation of podocytes occurs in rat [30]. Experimental evidence indicates mesengial cell proliferation and basement thickening in rats treated with AgNPs [36]. QDs affected mesengium in IP15 cells [45]. All these workers have equivocally attributed these effects to increased generation of ROS and consequently to oxidative stress and apoptosis [95]. These changes can be treated as warning signals for different renal diseases. Impairment of GEC can lead to albuminuria, glomerulosclerosis and vascular disease [96]. Injury to podocytes represents glomerular dysfunction [97]. Mesengial cell dysfunction is a key event in nephropathy [98]. Intriguingly, targeted NP-mediated drug delivery to mesengial cells and podocytes has been found useful to treat various renal diseases [99]. Designing of NPs plausibily with no effects on cellular components of glomerulus should be considered as an important objective of nanomedicine.
Another set of investigations discussed in present review deal with their effects on renal tubular epithelial cells. Tubular epithelium being highly susceptible to NPs showed apoptosis, necrosis and degenerative changes. These changes have been summarized in Figure 2. Specific lesions like amyloidosis, fibrosis and necrosis were observed in mice treated with SiNPs and rats treated with TiO2NPs. Photosensitizing potential of TiO2NPs opens new avenues in nanomedicine research. These observations need to be considered while formulating targeted drug delivery strategies to prevent renal diseases.
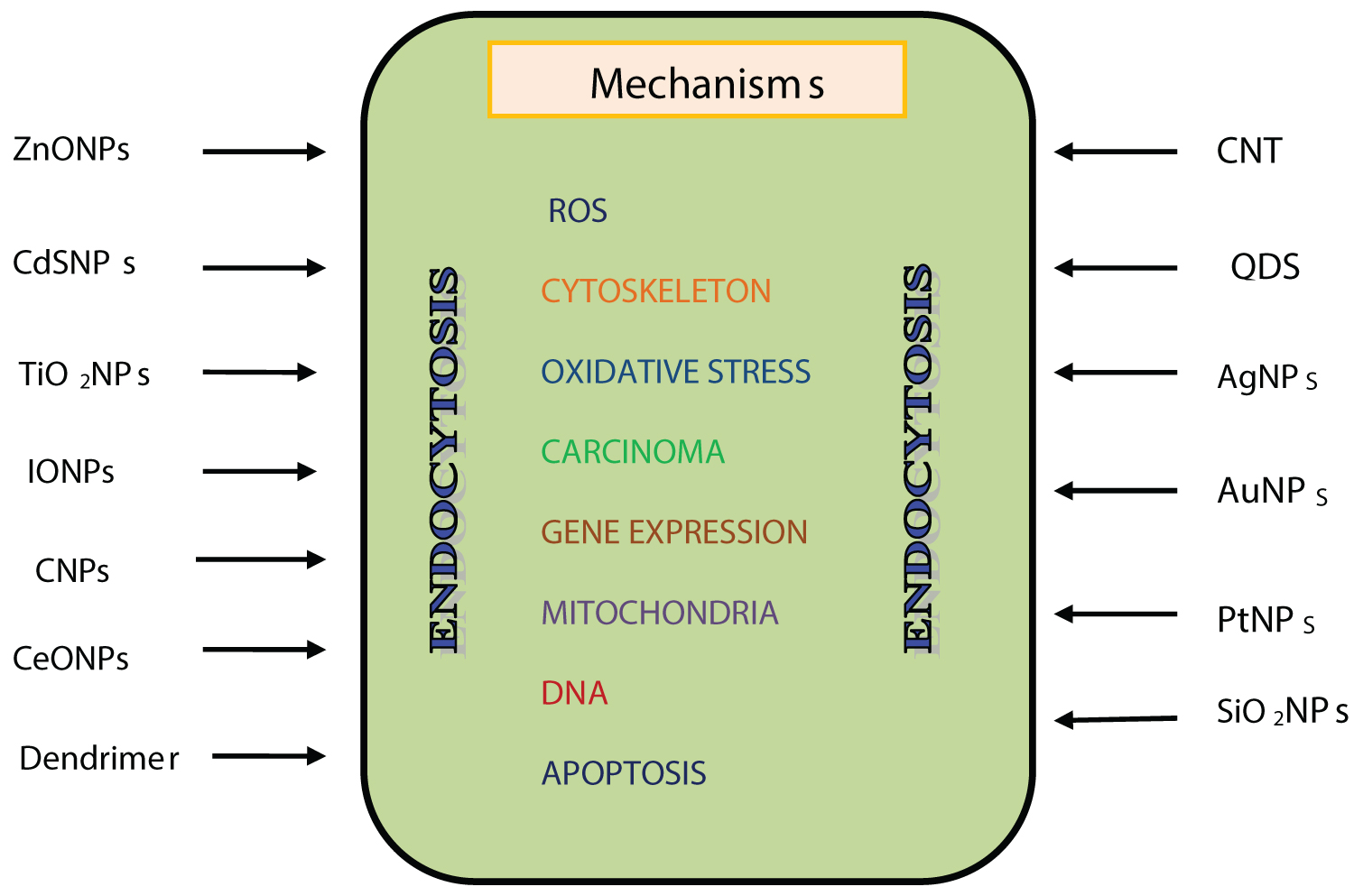 Figure 2: Mechanisms of renal cell death caused by ENPs.
View Figure 2
Figure 2: Mechanisms of renal cell death caused by ENPs.
View Figure 2
Although significant advances in NP research have been made during last few years, i) Acute and chronic effects of pristine and functionalized NPs; ii) Coated and uncoated NPs and iii) Comparative studies between NPs and their bulk counterparts are still needed. Precise understanding of potential molecular/biochemical mechanisms involved in renal toxicity of NPs should form the basis of selective therapeutic targeting of NPs.
The author declares no conflict of interest in the preparation of this article.
Financial assistance from Indian Science Congress Association, Kolkata received during the preparation of this article is gratefully acknowledged.