More than 350 million people are infected with Hepatitis B Virus (HBV) globally. The main aim of any chronic hepatitis B therapy is to prevent liver cirrhosis and its sequelae, including Hepatocellular Carcinoma (HCC). This is a cross sectional study that analysed medical records of 281 HBsAg seropositive patients in order to determine those factors that are associated with the reduction in the HBV viral load and the duration of HBV lamivudine monotherapy in order to attain a virological response. HBsAg patients in this study received the reverse transcriptase inhibitor drug lamivudine (100 mg/day). For this study, all HBsAg treatment naïve patients had baseline HBV levels between 200 and 104 copies/Ml. Age and duration of antiviral treatment were negatively correlated -0.11 (p = 0.07). Female patients, patients aged between 30-39 yrs and 40-49 yrs, delay in seeking HBsAg antiviral monotherapy following the appearance of initial symptoms, and all patients excluding those who are uneducated were significantly associated with the likelihood of having a mean HBV viral load in the plasma of 5log10 decline one-year after the initiation of lamivudine monotherapy irrespective of the presence of detectable HBV DNA. Findings from this study shows that HBsAg treatment naïve patients who started lamivudine monotherapy early following the initial appearance of HBV symptoms were associated with a viral load decline of 5log10 in plasma irrespective of the presence of detectable HBV DNA within one year after the commencement of lamivudine treatment compared to HBV treatment naïve patient who started treatment late.
More than 350 million people are infected with Hepatitis B Virus (HBV) globally [1]. Hepatitis B seroprevalence is defined as high, low or intermediate based on the seroprevalence of Hepatitis B antigen (HBsAg) carriers in the population [1]. Parenteral transmission among adults who engaged in unprotected sex remains the main cause for HBsAg transmission. Vertical transmission of HBsAg from mother to the unborn child is also a common mode of transmission of HBV especially in most parts of Africa. More than 20% of HBV carriers develop serious sequelae after initial infection [2]. Healthcare emergency personnel, commercial sex workers, individuals with multiple sex partners, illicit drug users, and homosexuals are at elevated risk of becoming infected with HBV [2]. HBsAg carriers are also at elevated risk for developing cirrhosis, hepatocellular carcinoma, and hepatic decomposition [3]. Until 2012 standard Interferon (IFN) and Lamivudine (LAM) were the only approved drugs for HBV treatment [4,5]. Despite the availability of other HBV antiviral drugs such as tenofovir, nevirapine, and stavudine in Sierra Leone, LAM still remains the popular choice because it is easily affordable. The main aim of any chronic hepatitis B therapy is to prevent liver cirrhosis and its sequelae, including Hepatocellular Carcinoma (HCC). Other objectives of an HBV therapy is for a spontaneous or treatment-induced clearance of HBsAg as well as HBsAg seroconversion to anti-HBsAg followed by a long-term period of low-level HBsAg DNA replication. There is paucity of research data relating to the predictors of positive treatment outcome for chronic hepatitis B infection due to the heterogeneity of HBV treatment options, variation of treatment efficacy definition and treatment endpoints. HBV treatment efficacy definition and treatment endpoints vary significantly between clinical trials and are usually uncorrelated. A complete HBV treatment success is referred to as Sustained Virologic Response (SVR). SVR is the virologic and biochemical response due to HBV therapy which produces an undetectable or suppressed HBV DNA as well as the normalization of aminotransferase levels. However, the occurrence of HBV relapses following HBV antiviral therapy termination makes the case definition of SVR for HBsAg patient difficulty. For both, chronic HBeAg-positive and negative HBsAg patients, HBsAg seroconversion to anti-HBsAg would be the best definition for SVR. However, in most HBV patients, SVR is rarely achieved whether spontaneously or by treatment-induced.
This is a cross sectional study that analysed medical records of 281 chronic HBV HBsAg seropositive patients in order to determine those factors that are associated with the reduction in the HBV viral load and the duration of HBV LAM monotherapy prior to attaining a virological response. All study participants prior to the starting of LAM monotherapy had normal Alanine Aminotransferase Level (ALA) ranging 33-56 U/L (Figure 1). Because only ALA test was done there was no diagnostic confirmed of liver damage due to HBV infection in these patients. Lamivudine was prescribed to these patients irrespective of their liver status because it is the most affordable drug used to commence HBV therapy in most resource-poor countries including Sierra Leone [6]. HBsAg patients whose treatment outcome data were used in this study visited Bo Government Hospital (BGH) in between September 2010 to November 2013. All study participants had no prior knowledge about their HBsAg seropositive status and had only visited BGH laboratory for HBsAg testing based on suspicion of being infected with HIV or HBV or both. Study participants came from all chiefdoms around Bo District and represents different ethnic and socioeconomic backgrounds. HBsAg patients who were diagnosed outside Bo District were excluded from this study. This study followed for one year chronic HBV treatment naive patients who went through HBsAg antiviral monotherapy with lamivudine in Bo District in Sierra Leone from September 2010 to November 2013.
 Figure 1: ALA levels for chronic HBV HBsAg seropositive patients prior to start of lamivudine monotherapy. From Figure 1 ALA levels of chronic HBV HBsAg seropositive patients whose medical records were analysed in this study range between 33-56 UL. View Figure 1
Figure 1: ALA levels for chronic HBV HBsAg seropositive patients prior to start of lamivudine monotherapy. From Figure 1 ALA levels of chronic HBV HBsAg seropositive patients whose medical records were analysed in this study range between 33-56 UL. View Figure 1
The BGH which provided data for this study is located in Bo District - 300 km from the capital city Freetown. Patient's medical records were retrieved from BGH after a written consent was obtained from the hospital authorities. This study involves the analysis of secondary data of HBV seropositive patients who were diagnosed at the BGH and received lamivudine monotherapy in between September 2010 to November 2013. ICD-9-CM 070.3 was used in this study for all HBsAg diagnosis and classification. Specifically, ICD-9-CM 070.3 was used to identified chronic viral HBV HBsAg seropositive patients in this study who shown no signs and symptoms of liver problems, don't have clinically diagnosed hepatic coma or did not report liver problems prior to the commencement of lamivudine monotherapy. HBV patients who were initially diagnosed as HBsAg seropositive were requested to do re-diagnosis after 6 weeks for confirmation of their HBV status. HBV patients who were HBsAg seropositive were later prescribed antiviral drugs for treatment which they bought from various pharmacies in the country. They were also asked to report every 4 weeks for further medical examinations and observations.
The BGH uses the Rapid Determine HBsAg immune chromatographic assay (Inverness Medical Japan Co; Ltd, Chiba, Japan) test for all HBsAg diagnosis in this study. Clinical sensitive and specificity of the Rapid Determine HBsAg immunoassay test are 95% and 99%, respectively [7]. Rapid Determine test's clinical sensitivity and specificity depends on the technology on which they are manufactured [8]. A Malawian study had previously raised concern about the specificity of the Rapid Determine HBsAg diagnostic test [9]. Rapid Determine HBsAg immunoassay test is commonly used in resource-constraint countries like Sierra Leone because it is affordable and is technically undemanding. Its dual purpose of HBV diagnosis and monitoring makes it a popular choice also. HBsAg diagnosis using Rapid Determine HBsAg immunoassay test involves collecting from a patient 50 ul blood sample using finger stick and an EDTA serological kit, transfer the blood to a sample pad and apply drops of chase buffer to the blood sample on the sample pad. The blood sample is allowed to wait for 10-15 minutes before results could be read. A Rapid Determine HBsAg immunoassay test is positive when a visible red bar appears in the patient's window that is darker or lighter than the control window. A red bar in the control window (window C) with no red bar in the patient window reads negative HBsAg result. An invalid HBsAg test result occurs when a red bar of equal strength appears in both the control and patient windows.
SAS 9.2 version [10] was used for both descriptive and exploratory statistical analysis of HBV patients' demographics and treatment-related factors in this study. HBV patients' demographic characteristics analysed in this study includes age (continuous and categorical), sex, occupation, and educational status. HBV patient's treatment-related data analysed in this study were duration of LAM monotherapy before experiencing a virological response, delay period in seeking LAM monotherapy following initial diagnosis, and the reduction in viral load (virus copies/Ml in plasma) one year after the initiation of LAM monotherapy. Duration of LAM monotherapy before experiencing a virological response and the reduction in mean viral load one year after the initiation LAM monotherapy are the dependent variables for analysis in this study. Duration of LAM monotherapy before experiencing a virological response and the delay in seeking LAM monotherapy were represented as median while mean HBV viral load one year after the initiation of LAM monotherapy was represented as mean. Spearman's correlation was used to determine whether age, duration of LAM monotherapy before experiencing a virological response, and the delay in seeking LAM monotherapy following initial diagnosis are correlated while Pearson's chi-squared test was used to compare sex, occupation, age (categorical), HBV viral load one year after the initiation of LAM monotherapy, and education status. Multivariate logistic regression was used to determine those HBV patient factors that determine the likelihood of an HBV patient having a reduced mean HBV viral load one year after the initiation of LAM monotherapy. Multivariate Poisson regression was used to determine those factors associated with the duration of LAM monotherapy before experiencing a virological response. This study considers a HBsAg patients to have attained a virological response when there is a decline in the mean HBV viral load in the plasma of 5log10 within one-year after the initiation of LAM monotherapy irrespective of the presence of detectable HBV DNA. All hypothesis test results in this study are two-tailed, with p = < 0.05 indicating a statistical significant association.
HBsAg patients in this study received the reverse transcriptase inhibitor drug lamivudine (100 mg/day). The effectiveness of LAM on HBV treatment varies with time. The seroconversion rate for HBV HBeAg patients on LAM therapy with viral load < 104 pg/Ml improves by 27% after 2 years, 40% after 3 years, and 47% after 4 years of treatment [11]. For HBsAg seropositive patients with chronic HBV disease, antiviral therapy is recommended when the HBV DNA level is > 20,000 IU/mL (105 copies/mL) or when the EASL level is > 2,000 IU/mL [12]. Antiviral therapy is also recommended for HBsAg seropositive patients with chronic HBV disease and HBV DNA level is > 20,000 IU/mL (105 copies/mL) when the serum Alanine Aminotransferase (ALT) is elevated for 3-6 months [13]. For this study a viral load reduction implies a decline in the mean HBV viral load in the plasma of 5log10 one-year after the initiation of LAM monotherapy irrespective of the presence of detectable HBV DNA. Because of the high cost of the HBV antiviral drugs in resource-poor countries HBV monotherapy is common. There has been conflicting views regarding what the baseline HBV viral load should be before the initiation of antiviral therapy. For this study, all HBsAg treatment naïve patients had baseline HBV levels between 200 and 104 copies/Ml.
Ethical clearance for this study was obtained from the Njala University Ethics Committee which reviewed the study protocol. This study used anonymized patient medical records and was declared to have met the criteria for exemption from obtaining informed consent from the study participants.
Out of 348 patients who met the case definition of HBsAg seropositive after diagnosis at the KGH Laboratory in Bo in between September 2010 to November 2013, we analysed the medical record of 281 (80.75%) patients: 142 (50.53%) male and 139 (49.47%) females. The mean viral load one year after the initiation of LAM monotherapy and the duration of LAM monotherapy before experiencing a virological response were not normally distribute (Figure 2). HBsAg patients median age was 28 (IQR = 30), median delay period to seek HBsAg treatment following initial diagnosis of HBV was 6.00 months (IQR = 6.00 months) while the median duration for HBsAg LAM monotherapy was 9.0 months (IQR = 10.00 months). The mean viral load of HBsAg patients one year after the initiation of LAM monotherapy was -3.56log10 virus copies/Ml in plasma. One hundred and seventy (60.50%) of the HBsAg patients who sought HBsAg LAM monotherapy had diminished viral load and survived one year after the initiation of HBsAg LAM monotherapy. One hundred and eleven (39.50%) HBsAg patients died one year after the initiation of LAM monotherapy. Fifty-four (19.22%) females and 57 (20.28%) males died during HBsAg LAM monotherapy. Equal number 85 (30.25%) of males and females recorded reduction of mean viral load one year after the initiation of LAM monotherapy. HBsAg patients with primary education recorded the highest (27.05%) treatment failure (died) during HBsAg LAM monotherapy while unemployed HBsAg patients recorded the lowest number (1.07%) of cases to survive during one year of HBV antiviral treatment. The correlation between age, delay in seeking HBsAg LAM monotherapy and duration of LAM monotherapy were not significant. The Pearson correlation coefficient for age and the delay in seeking HBsAg LAM monotherapy following initial diagnosis of HBV was 0.11 (p = 0.08). Age and duration of LAM monotherapy were negatively correlated -0.11 (p = 0.07).
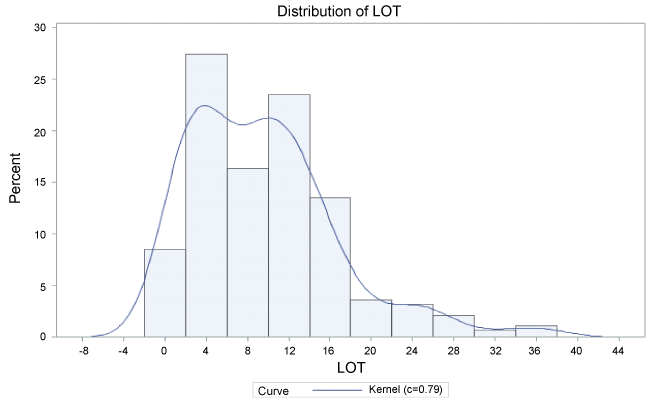 Figure 2: Distribution of duration of HBV antiviral monotherapy for HBsAg naive patients follow up for one year. From Figure 2 the duration of lamivudine monotherapy for HBsAg treatment naive patients is not normally distributed: majority of the patients spent 4-16 months on lamivudine monotherapy. View Figure 2
Figure 2: Distribution of duration of HBV antiviral monotherapy for HBsAg naive patients follow up for one year. From Figure 2 the duration of lamivudine monotherapy for HBsAg treatment naive patients is not normally distributed: majority of the patients spent 4-16 months on lamivudine monotherapy. View Figure 2
We used Cox regression model to investigate how age, sex, occupation, and delay in commencing LAM monotherapy following initial diagnosis of HBV influenced the survival of HBsAg seropositive patients within one year of therapy and the result is presented in Table 1. The finding shows that delay in commencing LAM monotherapy following initial diagnosis of HBV was significantly associated with the hazard rate. Specifically,there was an increase risk of death within one year among HBsAg patients who commenced HBsAg LAM monotherapy late after initial diagnosis of HBV (AHR = 0.97, 95% CI [0.96,1.43]). This study considers failure to initiate HBsAg LAM monotherapy within 6 months following the initial diagnosis of HBV as late.
Table 1: Cox regression analysis of HBsAg naive patients characteristics. View Table 1
We used Nelson-Aalen non-parametric estimates to determine the cumulative hazard rate following one year of LAM monotherapy among HBsAg treatment naive patients (Figure 3). We observed that beyond 10 months after commencing LAM monotherapy, 50% of the HBsAg treatment naive patient population failed their treatment. Also, the duration which the first 25% of HBsAg treatment naive patient population failed LAM monotherapy was longer (11 months) than the duration it took the second 25% of the HBsAg treatment naive patient population. It took 2 months for the second 25% of HBsAg treatment naive patients to fail LAM monotherapy. This shows that the hazard of LAM monotherapy failure progressively increases at least 10 months after the commencement of treatment.
 Figure 3: Hazard of lamivudine monotherapy failure among HBsAg treatment naive patients and length of antiviral monotherapy. Figure 3 Survival curve shows that 50% of HBsAg treatment naive patients failed their treatment 10 months after commencing HBV lamivudine monotherapy. View Figure 3
Figure 3: Hazard of lamivudine monotherapy failure among HBsAg treatment naive patients and length of antiviral monotherapy. Figure 3 Survival curve shows that 50% of HBsAg treatment naive patients failed their treatment 10 months after commencing HBV lamivudine monotherapy. View Figure 3
Multivariate logistic regression model was used to determine those factors that are associated with the likelihood of a HBV patient having a mean HBV viral load in the plasma of 5log10 decline one-year after the initiation of LAM monotherapy irrespective of the presence of detectable HBV DNA. Female patients, patients aged between 30-39 yrs and 40-49 yrs, delay in seeking HBsAg LAM monotherapy following initial diagnosis of HBV, and all patients excluding those who are uneducated were significantly associated with the likelihood of having a mean HBV viral load in the plasma of 5log10 decline one-year after the initiation of LAM monotherapy irrespective of the presence of detectable HBV DNA. Specifically, holding other covariates in the model constant, female HBsAg patients undergoing HBV LAM monotherapy were 1.09 times (p = 0.04) more likely to have a mean HBV viral load in the plasma of 5log10 decline one-year after the initiation of LAM monotherapy irrespective of the presence of detectable HBV DNA compared to men. Adjusting for other covariates in the model HBsAg treatment naive patients aged 30-39 yrs are 0.52 times (p = < 0.01) more likely to have a mean HBV viral load in the plasma of 5log10 decline one-year after the initiation of LAM monotherapy irrespective of the presence of detectable HBV DNA compared to HBsAg naive patients > 50 years. Also, HBsAg treatment naive patients aged 40-49 yrs are 0.38 times (p = 0.04) more likely to have a mean HBV viral load in the plasma of 5log10 decline one-year after the initiation of LAM monotherapy irrespective of the presence of detectable HBV DNA compared to HBsAg treatment naive patients > 50 years. HBsAg treatment naive patients with basic, secondary and medical education were significantly associated with having a mean HBV viral load in the plasma of 5log10 decline one-year after the initiation of LAM monotherapy irrespective of the presence of detectable HBV DNA compared to HBsAg naive patients who were uneducated.
Multivariate Poisson regression model was used to determine those factors associated with the duration of LAM monotherapy prior to experiencing a mean viral load decline of 5log10 in plasma irrespective of the presence of detectable HBV DNA within one year of LAM monotherapy among HBV treatment naive patients. The median duration for LAM monotherapy prior to experiencing a mean viral load decline of 5log10 in plasma irrespective of the presence of detectable HBV DNA within one year of LAM monotherapy among HBV treatment naive patients in this study was 9.0 months (IQR = 10.00 months). There was a gender difference in the duration ofLAM monotherapy prior to experiencing a mean viral load decline of 5log10 in plasma irrespective of the presence of detectable HBV DNA within one year of lamivudine monotherapy among HBV treatment naive patients. Men had longer duration for LAM monotherapy prior to experiencing a mean viral load decline of 5log10 in plasma irrespective of the presence of detectable HBV DNA within one year among HBV treatment naive patients than women (P < 0.01). In multivariable analysis, adjusting for other variables in the model, men (IR = 0.76, 95% CI [0.43,0.71]) were associated with longer duration of LAM monotherapy prior to experiencing a mean viral load decline of 5log10 in plasma irrespective of the presence of detectable HBV DNA within one year among HBV treatment naive patients than women. The goodness-of-fit chi-squared test for the Poisson regression was 453.690 (df = 498, P = 0.03) which indicates that the regression model fits the study data well. Also HBV treatment naive patients in this study who delayed in starting LAM monotherapy following initial diagnosis of HBV were associated with shorter duration of experiencing LAM monotherapy failure within one-year after commencing treatment compared to HBV treatment naive patients who seek LAM monotherapy early following initial diagnosis of HBV. In multivariable analysis, adjusting for other variables in the model, HBV treatment naive patients who delay in seeking LAM monotherapy following initial diagnosis of HBV were associated with shorter duration of experiencing LAM monotherapy failure within one-year after commencing treatment compared HBV treatment naive patients who seek LAM monotherapy early following the appearance of initial symptoms (IR = 0.85, 95% CI [0.31,0.42]).
Annually, there are approximately 620,000 HBV related deaths each year [14]. Three quarter of the global population are infected with HBV, six percent are chronic carriers and over 600,000 people die annually from acute HBV disease or chronic sequelae secondary to HBV infection [14]. HBV transmission continues to increase worldwide despite the upscaling of HBV management programmes. HBV, HCV, syphilis and genital herpes Type 2 and HIV share common transmission routes. Sierra Leone has a growing HBV epidemic. Some studies have estimated the national HBsAg seroprevalence rate between 2.6%-5.1% [15] although few independent investigations have documented alarming seroprevalence rates for the infection for different population groups in the country [16]. Findings from this study shows that HBsAg treatment naïve patients who started lamivudine monotherapy early following the initial appearance of HBV symptoms were associated with a viral load decline of 5log10 in plasma irrespective of the presence of detectable HBV DNA within one year after the commencement of lamivudine treatment compared to HBV treatment naïve patient who started treatment late. This may be attributed to the low viral load existing prior to the initiation of lamivudine monotherapy. HBV viral population in early infection is lower than the viral population during late infection since less viral particles may have been produced. The total amount of plasma virus production is 1011 HBV particles [17]. Also it may have been that HBsAg treatment naïve patients in our study who started lamivudine monotherapy early and may have encountered a lower viral population upon commencement of lamivudine monotherapy most of the serum viral load were cleared off during an initial exponential viral load decline phase thereby leading to a viral load decline of 5log10 in plasma irrespective of the presence of detectable HBV DNA within one year of treatment compared to those who started treatment late. For HBsAg treatment naïve patients who started LAM monotherapy late and encountered high existing viral load within the serum from which a considerable amount of viral load will still exist even after an exponential viral load decline phase. Late HBsAg treatment naïve patients may have experienced less viral load decline because of LAM resistance which may also be due to a high viral load and which may is also be associated to the late lamivudine treatment. Lamivudine has been reported to have resistance rates ranging from 6% to 32% after 1 year of therapy [18].
This study shows that HBsAg treatment naïve patients who initiated the lamivudine monotherapy at the initial appearance of HBV symptoms showed a viral load decline of 5log10 in plasma. The treatment with lamivudine was more effective in those patients who started early compared to the naïve patients who started late treatment.