Cryoglobulinemic vasculitis is a complex and destructive disease process that affects multiple organ systems. The pathophysiology includes formation of immune complex deposits that create an inflammatory response in various organs, yielding distinct presentations such as purpura, arthralgias, neuropathy, fever, and pulmonary vasculitis. Over 30% of cryoglobulinemic vasculitis cases present with glomerulonephritis, which carries a worse prognosis. The majority of mixed cryoglobulinemia cases are due to active infection with hepatitis C virus (HCV). Treatment of cryoglobulinemic vasculitis involves treatment of underlaying HCV, as well as high doses of immunosuppressive agents such as steroids and rituximab, often for multiple months, but with resolution of symptoms. It is rare for a patient who achieved sustained virologic response after HCV treatment to develop mixed cryoglobulinemia. Here, we present a rare case of cryoglobulinemic glomerulonephritis in a patient who was successfully treated for HCV infection.
Cryoglobulinemic vasculitis, Hepatitis C virus, Rituximab, Sustained virologic response
Cryoglobulinemic vasculitis is a complex and destructive inflammatory disease process that affects multiple organ systems including the skin, joints, peripheral nervous system, and kidneys [1]. In most cases, the disease is characterized by mixed cryoglobulinemiain which abnormal immune complexes, termed cryoglobulins, composed of IgM, IgA, and/or rheumatoid factor aggregate in the blood. Cryoglobulins can be found in chronic infections, lymphoproliferative diseases, and connective tissue diseases, such as Sjogrens and systemic lupus erythematosus (SLE) [2]. Cryoglobulins are proinflammatory and can cause several symptoms, including purpura, arthralgias, fever, hemoptysis, hypercoagulable states, and glomerulonephritis. Cryoglobulinemic vasculitis is often precipitated by infectious etiologies and most commonly by hepatitis C virus (HCV) infection [3]. Treatment of cryoglobulinemic vasculitis involves use of antiviral agents and high doses of immune suppressants, often for multiple months. Here, we present a rare case of cryoglobulinemic glomerulonephritis in a patient who had been successfully treated for HCV infection.
A 60-year-old man with a history of remote deep vein thromboses and HCV infection presented with dyspnea and myalgia that he had been experiencing for one week. His HCV infection nine years prior was treated with ledipasvir/sofosbuvir, resulting in sustained virologic response (SVR). At the time of admission, his vital signs were unremarkable except for a blood pressure of 162/76 mmHg. Physical examination demonstrated expiratory wheezing bilaterally, 1+ pitting edema, and bilateral petechial rash of the lower extremities (Figure 1). Urinalysis revealed proteinuria (100 mg/dL) and hematuria (more than 100 RBC/hpf on microscopy with dysmorphic appearance). The patient’s serum creatinine level (2.41 mg/dL; baseline 1.1 mg/dL) and rheumatoid factor level (207 IU/ml) were elevated, while the complement component 3 (C3) and component 4(C4) levels were decreased (71 mg/dL and < 5 mg/dL, respectively). Antineutrophil cytoplasmic antibody was negative, and HCV viral load was undetectable. Anti-glomerular basement membrane and antiphospholipid antibodies were also negative. Chest X-ray revealed appreciable bilateral pulmonary congestion, and transthoracic echocardiogram showed an ejection fraction of 54%. The patient developed hemoptysis for which a subsequent bronchoscopy confirmed diffuse alveolar hemorrhage. Serum cryoglobulins were detected, and he was treated with plasmapheresis for five days and a short course of pulse dose methylprednisolone (1 g/day) for three days. Renal biopsy confirmed cryoglobulinemic glomerulonephritis with a membranoproliferative pattern of injury (Figure 2 and Figure 3). The patient was subsequently managed with rituximab infusions at a dose of 375 mg/m2 per week over four weeks and a prolonged prednisone taper, which resulted in improved renal function and clinical status. Renal function normalized after a few months. Rheumatoid factor became undetectable two weeks after the first four-doses of rituximab. Both C3 and C4 returned to normal range 12 weeks after rituximab.
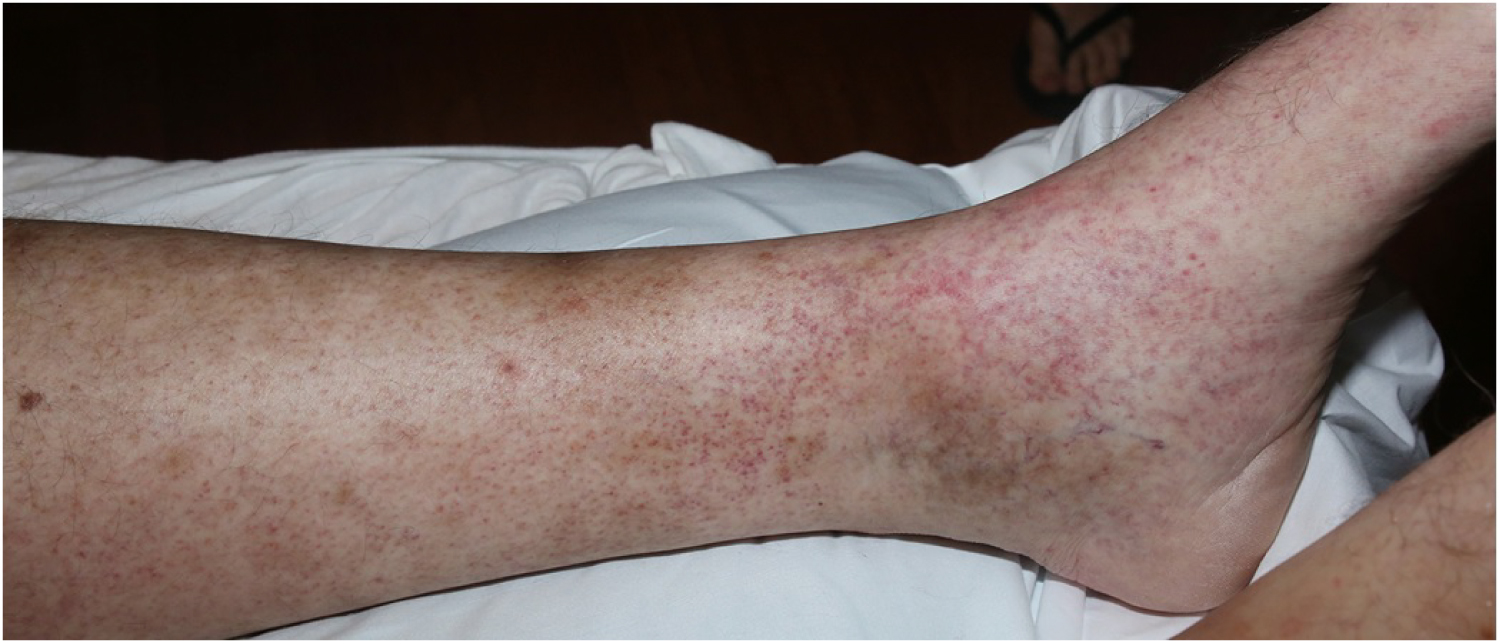 Figure 1: Petechial rash of the lower extremities in a patient with cryoglobulinemic vasculitis.
View Figure 1
Figure 1: Petechial rash of the lower extremities in a patient with cryoglobulinemic vasculitis.
View Figure 1
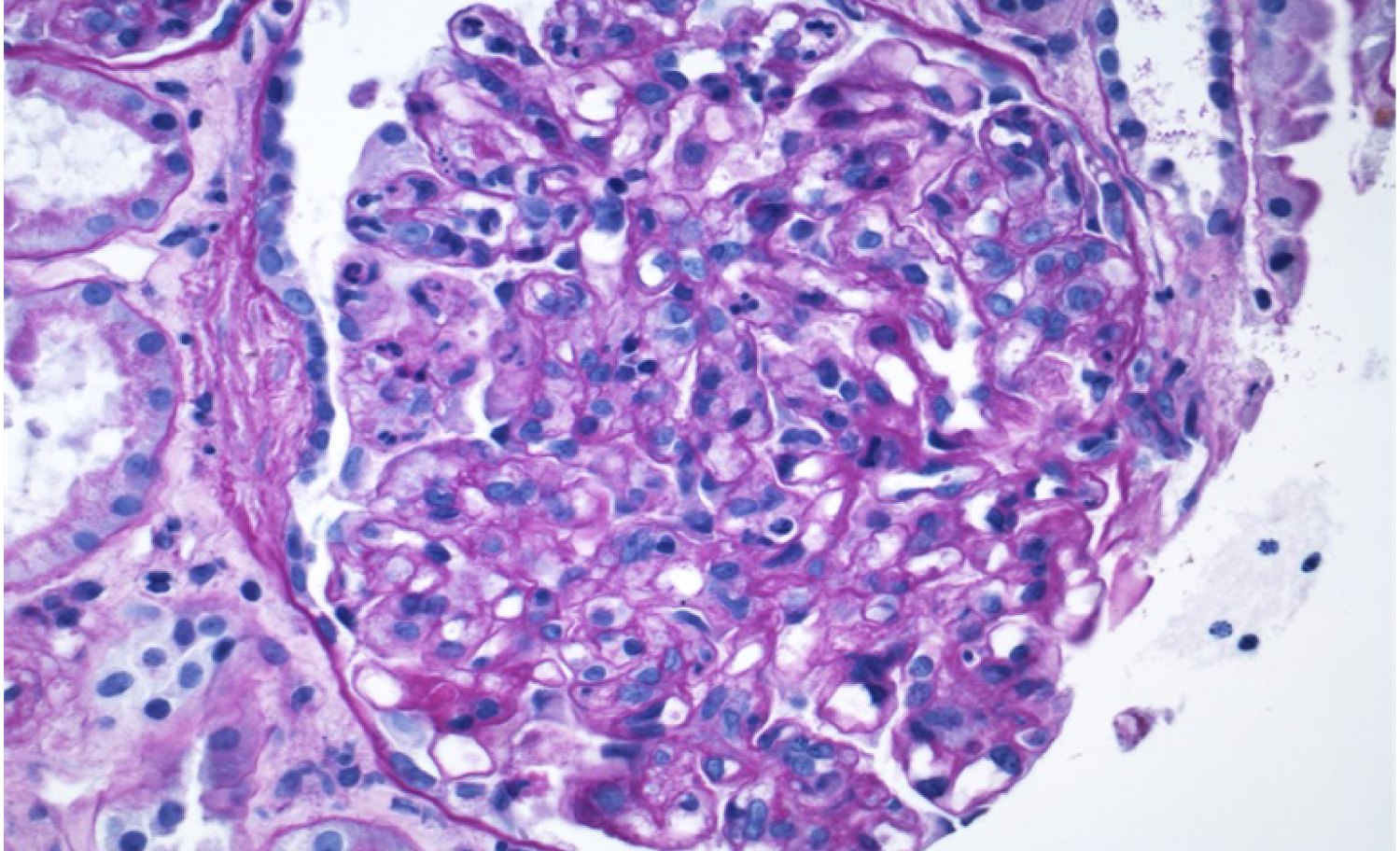 Figure 2: Kidney biopsy sections demonstrated cryoglobulinemic glomerulonephritis with hyaline pseudothrombi, and membranoproliferative glomerulonephritis injury pattern (x400, H&E stain).
View Figure 2
Figure 2: Kidney biopsy sections demonstrated cryoglobulinemic glomerulonephritis with hyaline pseudothrombi, and membranoproliferative glomerulonephritis injury pattern (x400, H&E stain).
View Figure 2
 Figure 3: Electron microscopy of renal biopsy showed severe podocyte effacement (> 80%) and endocapillary hypercellularity in patient with cryoglobulinemic glomerulonephritis.
View Figure 3
Figure 3: Electron microscopy of renal biopsy showed severe podocyte effacement (> 80%) and endocapillary hypercellularity in patient with cryoglobulinemic glomerulonephritis.
View Figure 3
Cryoglobulinemic vasculitis can present with non-specific signs and symptoms affecting multiple organ systems. Mixed cryoglobulinemia type II accounts for 65% of cases. In type II cryoglobulinemia, monoclonal IgM antibodies aberrantly complex with polyclonal IgG and the autoantibody rheumatoid factor to form immune complexes called cryoglobulins. Cryoglobulinemic vasculitis affects multiple organ systems due to an inflammatory cascade involving HCV antigen-stimulated B-cell polyclonal expansion followed by production of the cryoglobulins. These immune complexes precipitate at temperatures less than 37 ℃, and thus deposit in the vascular endothelium. Cryoglobulins bind the C1qR receptor on endothelial cells, causing release of vasoactive peptides, activation of the classical complement pathway, and production of low levels of C4 [4]. Release of C4 promotes recruitment of proinflammatory cytokines and neutrophils, which manifests clinically as the signs and symptoms of systemic vasculitis [5].
Cryoglobulinemic vasculitis patients often exhibit renal involvement, depressed serum C4 levels, elevated rheumatoid factor, and detectable HCV viral load [6]. Among patients, the main symptoms of vasculitis include purpura/petechia, arthralgias, weakness and fever. Such patients can also exhibit cardiac ischemia, neuropathy, and gastrointestinal bleeding. Over 30% of patients also develop membranoproliferative glomerulonephritis and in some cases pulmonary vasculitis, which results in a worse prognosis [2].
Treatment of cryoglobulinemic vasculitis involves antiviral agents if there is active HCV infection, with successful clinical responses seen in up to 78% of patients [7]. Other studies compared the use of pegylated interferon therapy with or without the anti-CD20 agent rituximab (375 mg/m2/week for four weeks) in patients with active HCV and cryoglobulinemia. Patients treated with the rituximab showed a significantly improved clinical success rate of 83% after three years compared to those that received the standard therapy (40%) [8,9]. Another study showed that over 90% of patients that used oral direct acting antiviral agents achieved SVR over 12 weeks of treatment [10]. Data are lacking in cases of active cryoglobulinemia and undetectable HCV viral load. Data from the French Autoimmunity and Rituximab Registry showed a significant clinical response with complete remission in patients with non-HCV mixed cryoglobulinemic vasculitis after six months of treatment with rituximab 375 mg/m2/week for four doses and low-dose prednisone tapering to 5 mg/day [8]. Other studies have shown that two doses of rituximab at 250 mg/m2 can be effective with a clinical response rate of 79% in patients with HCV and active cryoglobulinemia [11].
Literature reports a 10-year survival rate of 74% after HCV treatment [3]. However, the presence of glomerulonephritis portends a worse prognosis, with 15% of patients advancing to end stage renal disease and 40% of patients experiencing subsequent cardiac disease, liver failure, or infection [3]. Many patients with SVR return to their previous baseline after treatment with rituximab, but more data need to be collected to further quantify this.
Mixed cryoglobulinemia is associated with HCV in 70% to 90% of cases [2]. Less than 100 cases of mixed cryoglobulinemic vasculitis following SVR after HCV infection have been reported in the literature. Although not determined, the mechanism of the immune response in such cryoglobulinemic vasculitis may involve impaired cryoglobulin clearance from past infection or persistent inactive HCV antigens [4].
Cryoglobulinemic vasculitis remains a rare cause of systemic vasculitis. Although not well-reported, this disease process can occur after successful HCV treatment. Few cases of mixed cryoglobulinemic vasculitis following SVR from HCV have been described in the literature. Here, we report one such case with associated membranoproliferative glomerulonephritis that was successfully treated with steroids, rituximab, and plasmapheresis for coexisting diffuse alveolar hemorrhage. Due to the morbidity and mortality associated with cryoglobulinemic vasculitis, clinicians must be aware of this disease phenomenon, diagnostic modalities and associated treatments.