Background: Acute kidney injury (AKI) is associated with increased mortality in pediatric intensive care unit (PICU) patients. Early detection of AKI helps clinicians to manage promptly. Renal angina index (RAI) is established from changes in serum creatinine and condition of patients. Neutrophil-to-lymphocyte ratio (NLR) is calculated from a complete blood count (CBC). There are very few studies on the application of RAI and NLR in PICU. This study aims to evaluate the efficacy of RAI and NLR as AKI biomarker in PICU patients.
Methods: A retrospective observational study was performed in 173 critically ill children, aged 1-15 years-old, from August 2018 to November 2019. RAI and NLR were recorded on the day of PICU admission. The primary outcome was the diagnostic precision (sensitivity, specificity, negative predictive value (NPV) and summary receiver-operating characteristics (ROC) analysis) of RAI and NLR to predict subsequent AKI, AKI-requiring RRT and mortality within 28 days after admission to the PICU.
Results: From the analysis of the 173 patients, RAI with a cutoff of 8 was associated with an area under the curve (AUC) of 0.71 in the ROC analysis with NPV of 98% to predict the subsequent AKI. The incorporation of NLR with a cutoff of 5.5 improved the performance of RAI, especially in the nonsurgical group to predict the needs for RRT, with an AUC of 0.88.
Conclusion: RAI could be used as a reliable point-of-care biomarker for the early detection of subsequent AKI. NLR improved the performance of RAI, especially in the nonsurgical group to predict the need for RRT.
Acute kidney injury, Renal angina index, Neutrophil-to-lymphocyte ratio, Renal replacement therapy, Prediction
Acute kidney injury (AKI) is a common complex disorder manifested as a rapid progress loss in renal excretory function, resulted by heterogenous etiologies [1,2]. The incidence of AKI has raised up to 27% in critically ill children in the past decades, occurring in both high-income and low-income country [3,4]. AKI is associated with severe complications and a mortality risk. Mortality rates according to AKI have declined recently reflecting the important of the early recognition of this syndrome and improvement in patient care. Therefore, early stratification of patients at risk for AKI, identification of risk exposures or increased awareness of this condition is a foundation for AKI management [1,5]. Progression to AKI-requiring renal replacement therapy (RRT) significantly increases mortality rate [6]. Early recognition and prediction of patients at risk for RRT play essential roles in assisting the clinicians caring for these patients.
The renal angina index (RAI) is established by a composition of patient AKI risk conditions and signs of renal injury. RAI creates risk stratification for patients [7-13] and helps identify critically ill children who are the most at risk of developing subsequent AKI according to the definition of AKI definition in pediatric intensive care unit (PICU). There are various studies investigated to apply the risk score in sick children for the early detection of AKI but there are very few studies working on this issue in developing countries. Recent studies are interested in the biomarker of AKI from a complete blood count (CBC). To our knowledge, neutrophils are important compositions of innate immunity and impose the acute phase of inflammation, which advocated their role as an early biomarker of subsequent AKI. Lymphocytes produce proinflammatory cytokine that stimulate development of AKI. Therefore, neutrophil-to-lymphocyte ratio (NLR), an easily available parameter found in a CBC highly interested by clinicians these days to use as a tool to detect early detect patients at risk of developing AKI [14,15]. As it can provide information for early diagnosis and predict adverse outcomes associated with AKI. Although there are systematically reviews about AKI biomarkers from CBC in adult but a standardized cutoff value for NLR has not been established [15-18]. Moreover, there are very few studies about NLR using as AKI biomarker in critically ill children.
Thus, this study has aimed to validate the use of RAI and NLR in predicting subsequent AKI, AKI that requires RRT, and mortality in critically ill children admitted to the PICU in a tertiary care center in developing country.
This retrospective observational study enrolled all patients, 1 to 15-years-old, admitted to the PICU at King Chulalongkorn Memorial Hospital between August 2018 and November 2019. Patients who had AKI stage 3 according to KDIGO AKI definition were excluded from the study. After approval of ethic, suitable patients were enrolled. Data were collected on demographic variables; age, sex, comorbidities, baseline eGRF and serum creatinine (SCr), renal angina index (RAI), stage of AKI and CBC on the day of PICU admission. The outcomes that we collected were incidence of subsequent AKI, needs for RRT and mortality within 28 days after admission to the PICU, unless the patient was discharged or died before 28 days. Data were innominate upon data entry.
The estimated GFR was calculated, using the modified bedside Schwartz formula, from serum creatinine and height in centimeters [19]. Baseline serum creatinine defined by the lowest value available within 90 days prior to admission; if no data were available, the baseline eGFR was assumed as 120 ml/min/1.73 m2 [20]. RAI (range 1 to 40) as a component of the risk and injury scores of AKI [9] shown in Figure 1 and NLR calculated from CBC shown in Figure 2 were collected on the day of admission to the PICU.
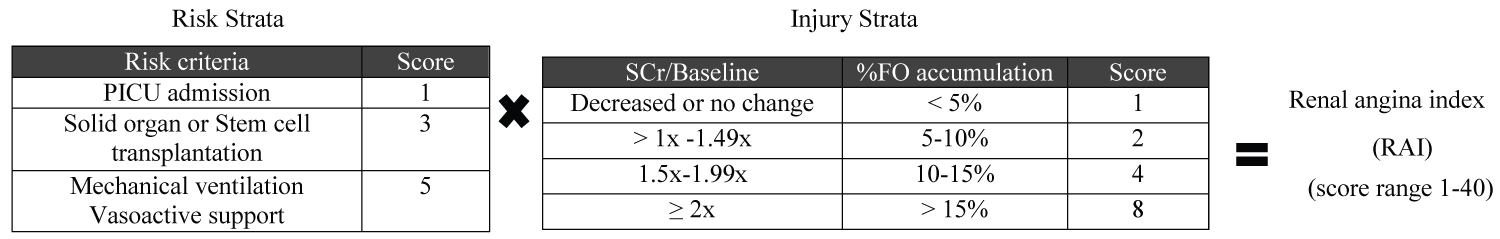 Figure 1: Renal angina index (RAI). The scheme was derived from Basu, et al. [2].
Figure 1: Renal angina index (RAI). The scheme was derived from Basu, et al. [2].
PICU: Pediatric Intensive Care Unit; SCr: Serum Creatinine; FO: Fluid Overload
View Figure 1
 Figure 2: Neutrophil-to-lymphocyte ratio.
View Figure 2
Figure 2: Neutrophil-to-lymphocyte ratio.
View Figure 2
The patients were described by their demographic, clinical and laboratory parameters. Continuous variables were expressed as median (interquartile range: IQR). Differences in continuous and categorical variables between two groups were assessed using a Wilcoxon rank sum test and Chi-square test or fisher exact test, respectively. Sensitivity, specificity, positive predictive value (PPV), and negative predictive value (NPV) were calculated using the receiver operating characteristic (ROC) curve. Empirical estimation of the cut point for a diagnostic test was measured using the Liu method. All p values reported are two-sided. Statistical significance was defined as a p value < 0.05. Cox proportional hazard regression was used to determine the factors associated with incidence of AKI. Multivariate models were developed by adjusting for covariates with p value < 0.1 in univariate models. The sample size was estimated to be 120 patients, based on the assumptions that the proportion of critically ill patients developing AKI is similar to those of the AWARE study [3]. Stata version 15.1 (Stata Corp., College Station, Texas) was used for the analysis.
The primary outcome was the diagnostic accuracy (sensitivity, specificity, PPV, NPV and summary ROC analyses) of RAI and NLR to predict the subsequent AKI, AKI-requiring RRT and mortality within 28 days after PICU admission.
Of the 294 patients admitted to the PICU during those periods of time, 173 patients were enrolled for this study (Figure 3). The baseline characteristics of these patients are shown in Table 1 and Table 2. There is no different in age, sex, weight, height, and body surface area (BSA) between groups by RAI with cutoff of 8. Majority of patients in this study have oncologic and/or infectious disease conditions. Infectious disease is significantly different between groups by RAI with cutoff of 8 (11/79 (13.9%) vs. 36/94 (38.3%); p < 0.001).
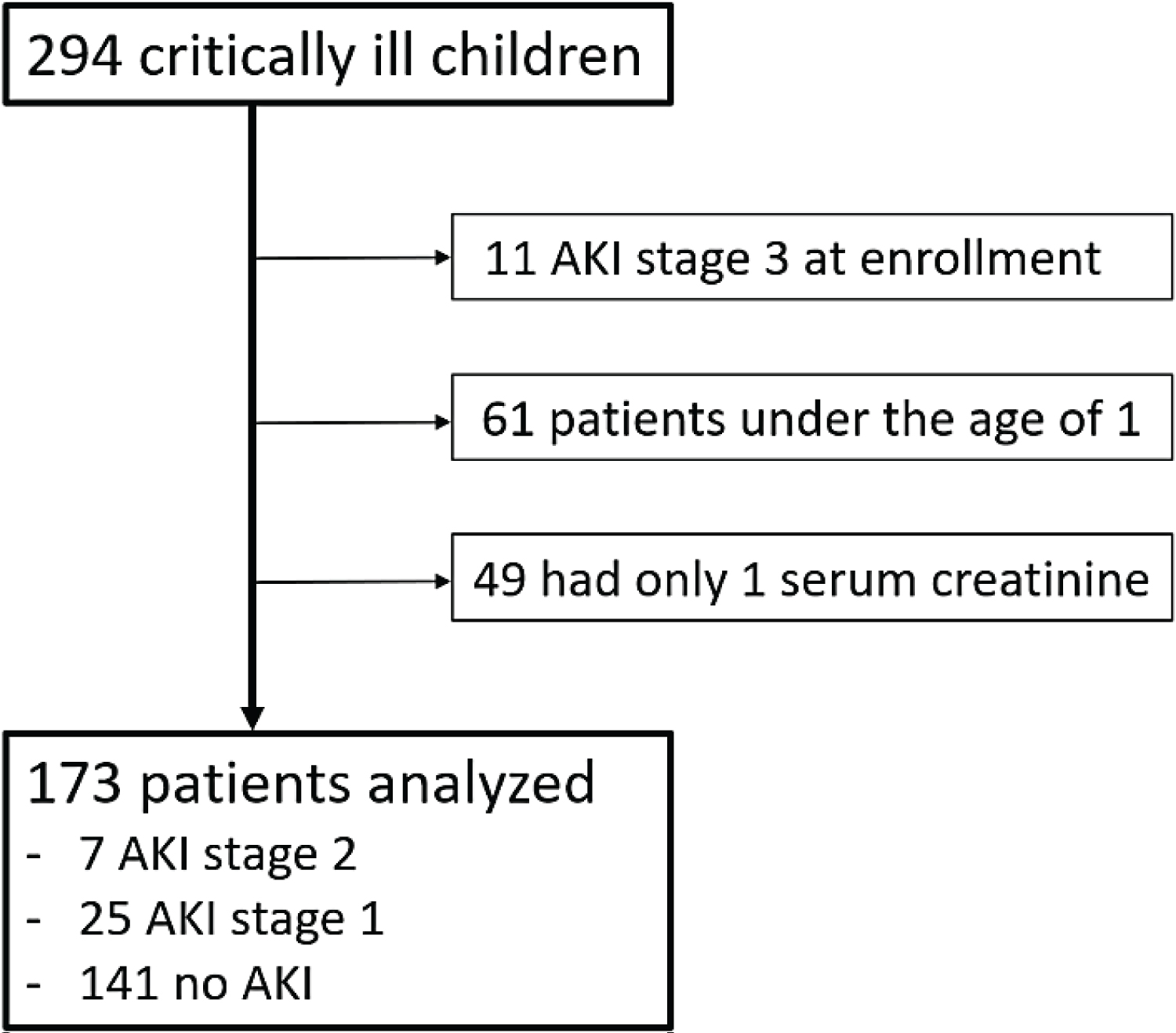 Figure 3: The flowchart of the enrolled patients.
Figure 3: The flowchart of the enrolled patients.
AKI: Acute kidney injury.
View Figure 3
Table 1: Baseline characteristics. View Table 1
Table 2: Laboratory values and biomarker score at enrollment. View Table 2
Primary diagnosis on the day of PICU admission were shock (22%), cardiovascular condition (5.2%), respiratory condition (24.3%), surgical/trauma condition (40.5%), central nervous system (7.5%) and pain management or sedation (0.5%). At enrollment, 141 (81.5%) patients had no AKI. Of the total of 173 patients enrolled, 22 patients (12.7%) developed subsequent AKI and 4 patients (2.3%) developed AKI requiring RRT within 28 days after admission to the PICU. Eighteen (81.8%) of the 20 patients developed subsequent AKI in the first seven days. Mortality rate was 6.9% in this study.
The incidence of subsequent AKI was significantly higher in patients with RAI ≥ 8 (20/94 (21.3%) vs. 2/79 (2.5%); p = 0.002) (Figure 4). Twenty (91%) of the 22 patients who developed AKI had RAI ≥ 8. The RAI ≥ 8 had potential to predict development of AKI with an AUC of 0.71 (95% confidence interval (CI) = 0.64-0.78). The RAI ≥ 8 had a high NPV of 98% (95% CI = 91-100%), with sensitivity and specificity of 91 and 51%, respectively, and PPV of 21% (95% CI = 14-31%) (Figure 5). After analyzed the factors associated with incidence of AKI, RAI ≥ 8 was significantly associated with an increased risk of subsequent AKI (HR 7.13 (1.67 - 30.57), p = 0.01 and adjusted HR 11.44 (1.15-114.07), p = 0.04) (Table 3).
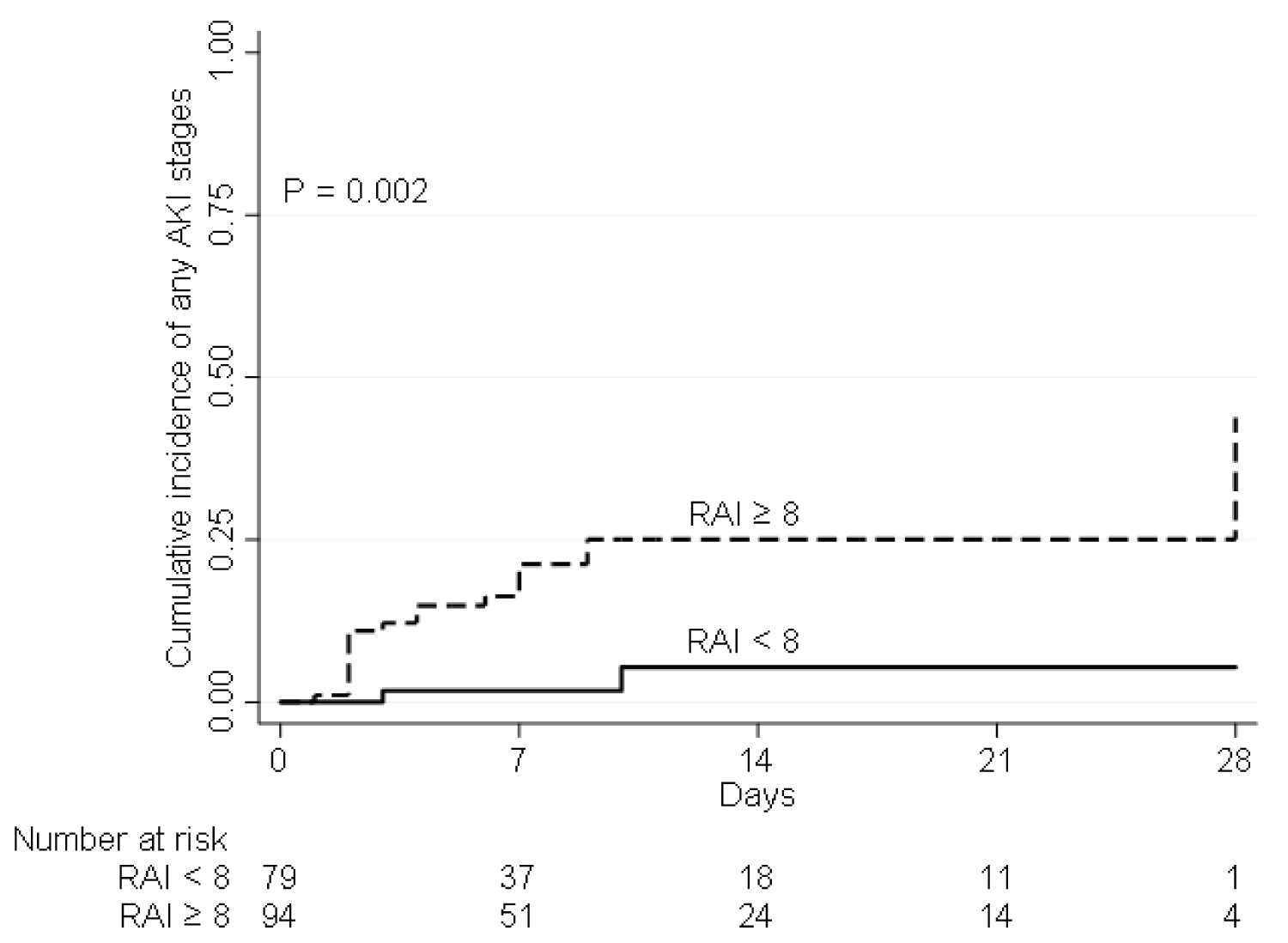 Figure 4: Comparison of a cumulative incidence of AKI within 28 days after PICU admission between the patients with RAI scores ≥ 8 and patients with RAI scores ≥ 8 at enrollment.
Figure 4: Comparison of a cumulative incidence of AKI within 28 days after PICU admission between the patients with RAI scores ≥ 8 and patients with RAI scores ≥ 8 at enrollment.
AKI: Acute Kidney Injury; RAI: Renal Angina Index
View Figure 4
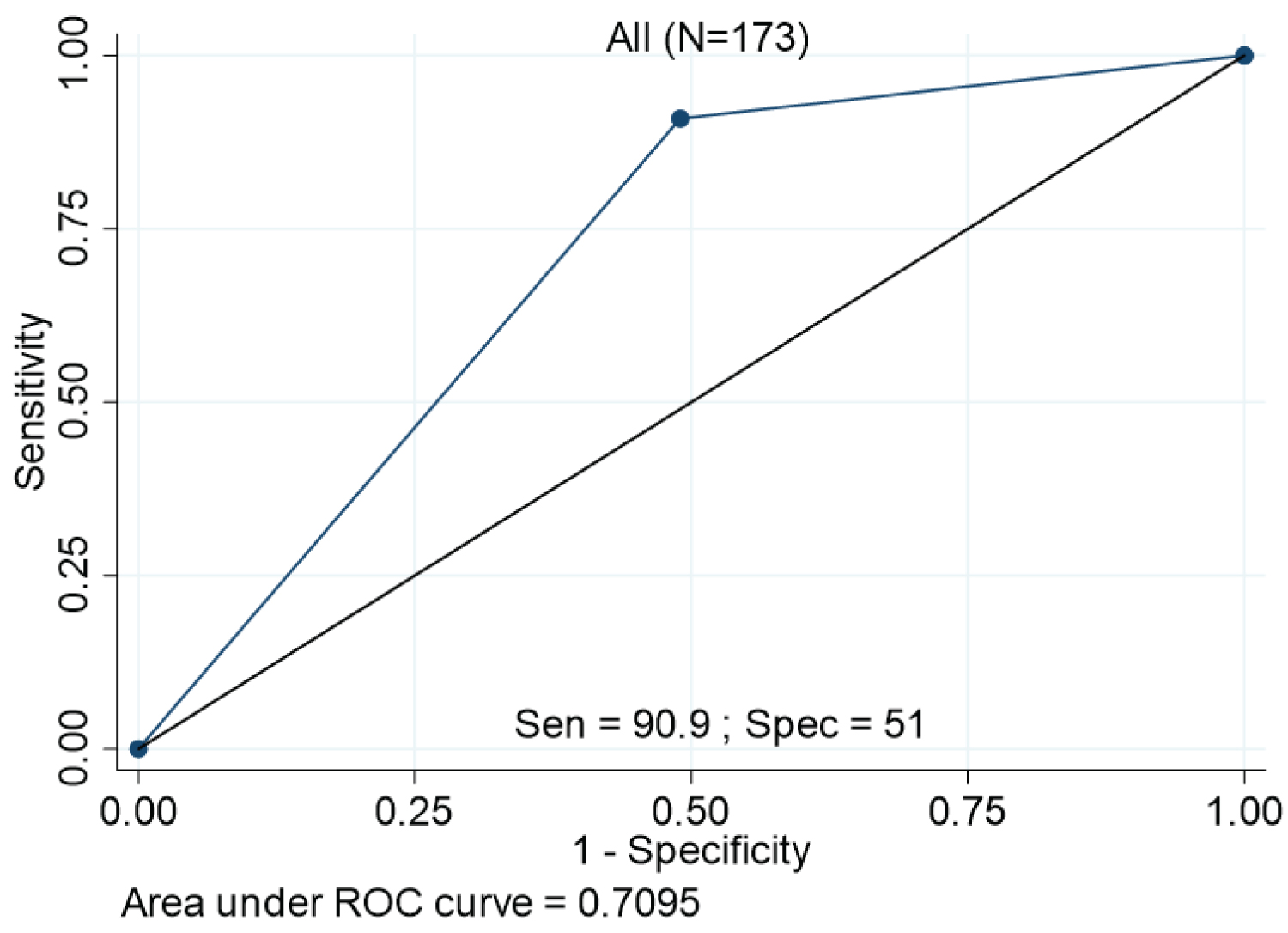 Figure 5: ROC curve. The plot represents ROC curve of RAI scores ≥ 8 for prediction for incidence of AKI within 28 days after PICU admission.
View Figure 5
Figure 5: ROC curve. The plot represents ROC curve of RAI scores ≥ 8 for prediction for incidence of AKI within 28 days after PICU admission.
View Figure 5
Table 3: Risk factors associated with incidence of subsequent AKI. View Table 3
The performance of NLR as AKI biomarker with a cutoff of 5.5 was not as good as the performance shown in adult [1]. NLR ≥ 5.5 had an AUC of 0.48 (95% CI = 0.37-0.59), NPV of 86.4% (95% CI = 78.2-92.4%), sensitivity and specificity of 36.4 and 58.9%, respectively, and PPV of 11.4% (95% CI = 5.1-21.3%) to predict subsequent AKI.
The performance in predicting subsequent AKI with NLR ≥ 5.5 in children is poor with only an AUC of 0.48 (95% CI = 0.37-0.59). However, after we performed a subgroup analysis, NLR showed a good performance for assessment of AKI-requiring RRT within 28 days after admission to the PICU in non-surgical group with an AUC of 0.83 (95% CI = 0.78-0.88) in the ROC analysis. And the NLR with this cutoff value also had a high NPV of 100% (95% CI = 94.5-100%), with sensitivity and specificity of 100% and 65.7%, respectively. After being combined with the RAI, with a cut-off of 8 and NLR, with a cut-off of 5.5, they contributed greatly to stratification of patients at risk for AKI-requiring RRT in the non-surgical group, with an AUC of 0.88 (95% CI = 0.84-0.92) (Table 4).
Table 4: Performance of the incorporation of the RAI and the NLR in subgroup analysis. View Table 4
Over the past decade, there have been great efforts to develop and standardize novel biomarkers for early detection of AKI due to their burden. In these days, RRT has bypassed from a therapy for patients with renal failure to a timely therapeutic intervention to support the patient with AKI by facilitating renal recovery and preventing worsening of outcomes [1,5]. We revealed that 81.8% of patients with AKI developed AKI in first 7 days after admission same as the data from the AWARE study that shown 75% of increasing incidence of AKI within 4 days after admission. So, we advocate clinicians to aware the subsequent AKI in this time frame. Therefore, the novel point-of care AKI biomarkers that assist clinicians for early diagnosis of AKI, especially in critically ill children, play the important role aimed to limit AKI progression.
Basu, et al. published the first study reporting that the fulfillment of renal angina index can be utilized to identify critically ill patients at the highest risk of developing severe AKI according to KDIGO AKI definition on day 3 after admission to the PICU with AUC values ranging from 0.54-0.78 [9]. The purpose of renal angina had been applied in many countries around the world due to its easy-to-use and good prognostic ability beyond serum creatinine and fluid accumulation. The result from our study showed good competitive performance of RAI for predicting subsequent AKI with an AUC of 0.71. Basu, et al. found that the RAI prediction of AKI by change in creatinine clearance from baseline provided superior result in AUC values to change in fluid overload. They concluded that the AUC values for RAI using for AKI prediction in critically ill children were not different for whichever the kidney injury outcome was used (change in creatinine clearance from baseline or change in fluid overload) [9]. From the AWARE study, the result ensured that serum creatinine alone missed AKI in the 67.2% of children who met the criteria of AKI of urine output [3]. Sethi, et al. assured that discrimination of RAI by fluid overload was superior to RAI by change in creatinine clearance. Therefore, one of the limitations of our study is that the fluid balance was not always been recorded in all children, and hence may be more children have AKI and missed due to unrecorded data. However, there is still the argument on this issue. And our study found that RAI by change in creatinine clearance alone still had a reliable prognostic accuracy and could be used as a biomarker to early detect subsequent AKI in PICU patients with high NPV.
NLR, easily obtained from CBC, showed that diagnostic accuracy not as good as the RAI. Until now, there is limited information about application of the NLR to predict pediatric AKI in advance. Based on our knowledge that AKI is a complex syndrome caused by numerous etiologies, we perform a subgroup analysis and found that NLR had a strong diagnostic capability to predict RRT needs in nonsurgical patients, especially when incorporated with the RAI. The sensitivity and specificity were up to 100% with a high AUC when we used RAI ≥ 8 and NLR ≥ 5.5 as the biomarkers for prediction of AKI-requiring RRT in non-surgical group. Most of patients in non-surgical group were primary diagnosed with sepsis or respiratory condition, hence the NLR, inflammation-related biomarker, could be explained how the NLR can enhance the performance of predicting subsequent AKI in these population. More studies are needed to investigate in critically ill children diagnosed with sepsis whether NLR can be used as a sepsis-associated AKI biomarker and what cutoff value should be used.
Another limitation of this study is that we had cardiac intensive care unit apart from PICU and we did not collect the data from our cardiac intensive care unit. Thereby applying the results of this study in cardiac patient should be careful.
For future direction, after we can stratify the patients at risk for subsequent AKI or AKI-requiring RRT, we can do further management and/or investigation such as furosemide stress test, urine neutrophil gelatinase-associated lipocalin (NGAL) or early RRT. As we know from previous studies that RRT was a timely therapeutic intervention to support the kidney and stopped further injury, knowing the patients at risk for RRT in advance is the key.
The RAI, with a cutoff of 8 was shown to be a reliable point-of-care biomarker for the early detection of subsequent AKI in critically ill children. Incorporation of NLR, with a cut-off of 5.5 improved the performance of RAI, especially in nonsurgical group to predict needs for RRT.
All the authors declared that there are no competing interests.
This study was approved by Institutional Review Board of King Chulalongkorn Memorial Hospital, Thailand. The requirement to obtain informed consent for this study was waived due to its observational nature. We would like to thank all the patients for invaluable data.