Physical exercises are highly effective in slowing muscle wasting and loss of function. However, whether vitamin D deficiency can interfere with this process in elderly individuals remains unclear. The objective of this study was to investigate the effect of 25(OH)D supplementation along with physical exercise in elderly women in an intervention study design.
We analyzed data from elderly women aged ≥ 60 years (mean age, 67 ± 5 years) who exercised regularly for at least one year at the Social Service of Commerce Santana, Community Center. We included 146 elderly women in the aquatic training program (AT), 99 elderly women in the multifunctional fitness program (MF), and 100 community-dwelling elderly women who had not been exercising for the last year as the control group (CT). In each group, we administered cholecalciferol to those who had 25(OH)D levels below the median levels. The individuals in the aquatic training supplemented group (ATSG), multifunctional fitness supplemented group (MFSG), and control supplemented group (CTSG) received one bottle of cholecalciferol each. They were instructed to consume 21,000 IU/week (4 drops/week) of cholecalciferol for 12 months. The timed up-and-go (TUG) test, 2-min step (2MST), 30-s chair stand (CS), functional reach (FRT), unipedal balance test with visual control (UB), and a portable dynamometer for the strength of hip flexors (HS) were conducted, and the serum 25(OH)D and intact parathyroid hormone (PTH) levels were measured at the beginning and end of the 12 months. The treatments were compared using a general linear model for repeated measures, with p < 0.005.
There was a significant increase in 25(OH)D levels in ATSG (p < 0.001), MFSG (p < 0.001), and CTSG (p < 0.001). The relationship between 25(OH)D levels and physical exercise was significant in TUG (p = 0.005), UB (p = 0.03), HS (p < 0.001), CS (p = 0.04), and 2MST (p = 0.02).
We found an interaction of 25(OH)D levels with physical exercise in elderly women for maintaining independence in daily activities and performance.
Functional capacity, Vitamin D, Performance, Aging, Physical exercises
Global awareness on vitamin D deficiency and its consequences has been increasing. Furthermore, there is substantial agreement among all guidelines that serum 25(OH)D concentrations < 25-30 nmol/L should be avoided in all age groups [1]. In addition, hypovitaminosis D is a world health problem, and Brazil has an elevated prevalence of hypovitaminosis D among its population [2]. In general, a 25(OH)D level of less than 20 ng/mL has been associated with neuromuscular performance issues in elderly people including poor physical performance, low muscular strength, and muscle weakness, thus suggesting that vitamin D supplementation may be beneficial [3,4].
In individuals with a vitamin D deficiency (≤ 20 ng/mL), proximal myopathy is observed by the atrophy of type II muscle fibers (fast twitch), which can have a negative effect on neuromuscular performance [5,6], and can contribute to osteopenia and osteoporosis by increasing parathyroid hormone (PTH) concentrations and bone turnover [7]. Based on this observation, many epidemiological studies have analyzed lower extremity function by testing performance during long walks among older adults who have low vitamin D levels [8].
Furthermore, vitamin D receptor (VDR) is found in most body tissues, including skeletal tissue, and active vitamin D has biological functions. Low vitamin D levels have been found to be associated with several conditions that can be common among elderly people, such as, sarcopenia, which is characterized by low muscle mass, falls, and fractures [9].
Vitamin D3 promotes muscular protein synthesis and activates calcium uptake in the sarcoplasmic reticulum, thus maintaining contractile efficiency in muscles [10]. A combination of nutritional intervention and resistance exercises seems to be the therapy of choice, especially for elderly people with sarcopenia [11]. In addition, Morley, et al. [11] reported that vitamin D supplementation was beneficial for elderly people who needed to increase muscle protein synthesis, muscle strength, muscle power, balance, and ability to maintain functional independence in the activities of daily living.
Physical exercises are highly effective in slowing down muscle wasting and loss of function; however, at times, elderly individuals do not exercise enough to prevent these changes [12]. Several studies in elderly people report a positive relationship between physical performance and vitamin D status [13].
While aquatic training programs are popular among elderly individuals, the resulting improvements in the performance and physical abilities of elderly individuals remain unclear. In contrast, the multifunctional fitness program has proved to be a good tool for improving physical capacities such as muscular strength, balance, agility, and aerobic resistance in elderly women [14]. In addition, this exercise program can also contribute to the prevention of physical function decline that results from aging [15].
No previous studies have analyzed the relationship between 25(OH)D supplementation, different modalities of physical exercise, and its neuromuscular effects. The aim of this study was to investigate the effect of 25(OH)D supplementation with physical exercises on the functional capacity and performance in elderly women.
The study was performed in accordance with the Declaration of Helsinki, and it was approved by the Ethics Committee of the Federal University of the Sao Paulo/UNIFESP. All subjects voluntarily signed a written consent.
This study included 458 elderly women aged ≥ 60 years, but only 345 elderly women completed the intervention after 12 months. Of the total, 146 individuals participated in the aquatic training program (AT) and 99 participated in the multifunctional fitness program (MF) at the Social Service of Commerce (SESC), São Paulo, Brazil. This group had been performing physical activity for at least one year. Furthermore, 100 community-dwelling elderly women from community centers providing services to elderly individuals living in the same geographic area, who had not been performing any regular physical exercise for at least one year, were also included in the study as the control group (CT). The control group did not receive any information about vitamin D supplementation and physical exercises. The participants were excluded if they were receiving hormonal treatment, were on medication for weight loss, were receiving vitamin D supplementation of > 600 IU/day, and/or had a creatinine level of > 1.5 mg/dL, a total calcium level of > 10.5 mg/dL, a psychiatric disorder, and/or an acute illness that would prevent them from exercising or following the protocol. The use of antihypertensive, antidiabetic, and antilipid medications was continued during the protocol, according to the medical prescriptions.
To confirm the physical activity levels of the participants, a short version of the International Physical Activity Questionnaire (IPAQ) was used [16]. The aquatic training supplemented group (ATSG) and multifunctional fitness supplemented group (MFSG) were classified by IPAQ as irregularly active, i.e., meeting at least one of the criteria of recommendation regarding the frequency or duration of physical activity: 5 days/week or 150 min/week. The control group was classified as sedentary (SSG) [17].
In the study, a maximum of three absences per month were allowed, beyond which the participant was excluded from the programs. The participants were guided by the same physical education instructor, twice a week. The activities were performed indoors, for one year, and in the same place.
The aquatic training and multifunctional fitness sessions lasted 60 min, starting with 10-min warm-up exercises, followed by cardiorespiratory training, coordination exercises, and strength and resistance exercises. The sessions ended with 10 min of stretching and balance exercises.
Originally, this study included 458 elderly women who were recruited and invited for laboratory and physical tests; however, 113 (24.6%) participants were unable to participate until the end of evaluations for various reasons. This included 88 (19.2%) participants who did not participate in the second phase of the study, 9 (1.9%) who only participated in the blood collection phase, 1 (0.2%) who died, 3 (0.6%) who met the exclusion criteria, 2 (0.4%) who relocated, and 10 (2.1%) who did not use vitamin D supplementation appropriately.
We followed an intervention study design. After data collection at baseline and the lack of consensus in the literature on vitamin D level cutoffs for performance evaluation during the study period (2010-2011), the median serum 25(OH)D levels of the participants were considered to define the supplemented group (SG) versus the non-supplemented control group (NG). Thus, the participants in each group (AT, MF, and CT) with serum levels of 25(OH)D below the median, 17.6 ng/mL for ATSG, 18.50 ng/mL for MFSG, and 14.60 ng/mL for SSG received cholecalciferol supplementation.
According to the Endocrine Society's practice guidelines on vitamin D, vitamin D deficiency, insufficiency, and sufficiency were defined as 25(OH)D levels of < 20 ng/mL, 21-29 ng/mL, and ≥ 30 ng/mL, respectively [18].
Two months before the initiation of the supplementation protocol, elderly women were instructed to discontinue the use of any vitamin D supplements. The supplemented group received 21,000 IU/week of vitamin D3 for 12 months. All participants were advised to take the supplement in the prescribed dose once a week (4 drops/week). To improve adherence to treatment, participants received a calendar in which the recommended dates for taking the supplement had been highlighted. On that calendar, there was also a designated area wherein the participants could mark the day of taking the supplement.
The vitamin D supplements were obtained at Magister® Pharmacy of Manipulation LTDA., São Paulo, SP, Brazil. The content of the cholecalciferol drops (5246 ± 20 IU/drop) was checked at the AFIP Laboratory for quality assurance in 17.5% of the sample.
The UNIFESP geriatrics outpatient service routinely prescribed at least 500 mg of calcium carbonate during the follow-up period, but calcium supplementation was not a part of our protocol. Participants were instructed to continue their usual diets, from which they ingested approximately 387 (294-513) mg/day of calcium [19].
Blood samples were collected after 12 h of fasting. All participants had their first blood sample collected in August 2010 and their second blood sample collected in October and November 2011. The serum concentration of 25(OH)D was determined by chemiluminescence using the LIAISON® 25 OH Vitamin D Total Assay (Product code: 310600; DiaSorin Inc., USA), with intra assay and inter-assay coefficients of 1.6% and 5.6%, respectively.
The intact PTH was determined by a chemiluminescence method (Elecsys® 2010, Roche Diagnostics, USA), with intra-assay and inter-assay coefficients of 3.0% and 3.5%, respectively. Total calcium was analyzed by an enzymatic colorimetric method (II Selectra Merck).
Anthropometric measurements included body weight (kg) and height (cm). Body mass index (BMI) was calculated as kg/m2, and individuals were categorized as underweight (< 20.0 kg/m2), normal weight (20.0-24.9 kg/m2), overweight (25.0-29.9 kg/m2), or obese (≥ 30.0 kg/m2) [20].
A portable dynamometer was used to measure muscle strength peak (kg) during isometric muscle contraction of hip flexors. The participant sat in a chair with the hips and knees flexed at 90°. The dynamometer was positioned perpendicularly on the patient's thigh, 5 cm above the upper edge of the patella. The participant had to raise the limb being tested against the dynamometer using maximum isometric muscle force for 5 s.
The participants were timed as they got up from a chair, walked 10 feet, without running, turned back, returned, and sat down again. The performances of two tests was measured at the baseline, and the final result was the mean of the two trials [21].
In a standing position, the subject had to flex one of her legs in a 90° for 30 s. Three attempts were made, and the average value was recorded [22].
The maximum displacement in horizontal distance that the subjects could reach ahead with the arms and trunk was measured. The test result was represented as the mean difference between the two trials [23].
The total number of repeated actions that the subjects could perform when they sat and stood up from a chair was recorded [24].
The number of full steps completed in 2 min while raising a knee to midway between the patella and iliac crest was measured. The result was presented as the number of times the right knee reached the target [25].
Statistical analysis was performed using the SPSS statistics software (version 19.0 for Windows). According to the G*Power software and based on expected observed power of 80% and an effect size of 49% (based in our previous results in a pilot study), in the TUG test score, the sample size included 432 individuals (72 individuals in each group). For characterization of the groups, we performed a descriptive analysis (mean ± standard deviation). The methodology was based on the median value for the 50% for vitamin D. The data in the tables are presented as mean and standard deviation. To verify the normality of the variables, the Kolmogorov-Smirnov test was used. The Z score was used to standardize data that did not follow the normality curve. The sphericity of the variables was corrected by Greenhouse-Geisser correction, when necessary. For the classification between the groups, the Bonferroni post-hoc test was used. The general linear model for repeated measures procedure was used for the analysis of physical performance measures; this method was used to evaluate the effect of time and the interaction for an association between physical exercises and supplementation. All data were adjusted for age and BMI. A value of α < 0.05 was considered significant.
Figure 1 and Table 1 shows the anthropometric measurements and clinical data of the groups before and after supplementation. A 25(OH)D deficiency was observed in the supplemented group (12.00 ± 3.00 ng/mL), and the increase in 25(OH)D level after treatment was significant (38.17 ± 12.41 ng/mL, p < 0.001). The non-supplemented group also showed increases in vitamin D levels pre- and post-treatment: 25.00 ± 8.40 ng/mL versus 28.05 ± 12.00 ng/mL (p < 0.001). In a comparison among the groups, there was a significant increase in vitamin D levels: ATSG, p < 0.001; MFSG, p < 0.001; and CTSG, p < 0.001. Body mass index and calcium levels were not statistically different between groups. Conversely, the PTH levels increased significantly in the CT group, which did not receive the supplements. The average age of the participants was 67 ± 5 years.
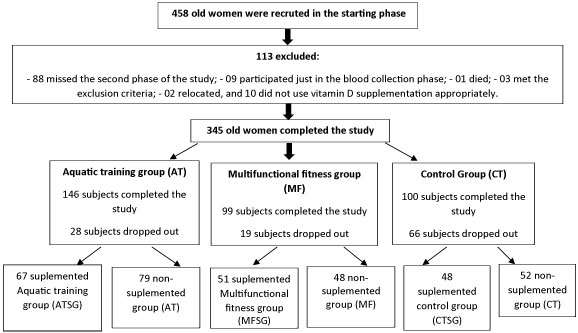 Figure 1: Study design. View Figure 1
Figure 1: Study design. View Figure 1
Table 1: Anthropometric measurements and clinical data, pre- and post-treatment in all groups. View Table 1
The data presented in Table 2 shows the mean ± SD values obtained on the physical tests in both groups. The comparison was made using the general linear model test (GLM) repeated measures: *, p ≤ 0.05; **, the effect of time with vitamin supplementation (comparison baseline and post-treatment intragroup) and interaction (effect of time and groups of physical tests with 25[OH]D); ¤¤indicates the superior treatment between ATSG vs. CTSG, p < 0.05; ¤indicates superior treatment between MFSG vs. CTSG; § indicates the superior treatment between MFSG vs. ATSG.
Table 2: Follow-up of physical tests in treatment and non-treatment groups. View Table 2
In the CTSG group, there was a significant interaction with vitamin D in the TUG test after supplementation (TUG B). Furthermore, the TUG test showed a significant superiority in the AT group versus the CT group. In the UB test, the MF group was superior in relation to the AT and CT groups. Conversely, the MFSG showed superiority to both the ATSG and CTSG. In the CS test, there was statistical significance in both the MFSG versus CTSG and the ATSG versus the CTSG, and in the non-supplemented groups, the significance was the same. In the 2MST test, MFST was statistically significant than CTSG and ATSG. The latter group was also superior in relation to CTSG. The MT and AT groups had significantly better results than the CT group.
Functional reach test (FRT); hip strength test (HS); timed up-and-go test (TUG); unipedal balance test with visual control (UB); 30-s chair stand test (CS); 2-min step test (2MST); _A, pretreatment; _B, post-treatment.
Data presented as the mean ± SD, the comparison made using the general linear model test (GLM) repeated measures; *, p ≤ 0.05; **, effect of time: comparison baseline and post-treatment intragroup; interaction: effect of time, and physical tests in groups with 25(OH)D supplementation.
Figure 2 shows that the MFSG values were superior in comparison with ATSG and CTSG in the following tests: HS (MFSG vs. CTSG, p = 0.003; MFSG vs. ATSG, p = 0.02; ATSG vs. CTSG; p = 0.001); 2MST (MFSG vs. CTSG, p = 0.01; MFSG vs. ATSG, p = 0.02), CS (MFSG vs. CTSG, p = 0.001); and UB (MFSG vs. CTSG, p = 0.002).
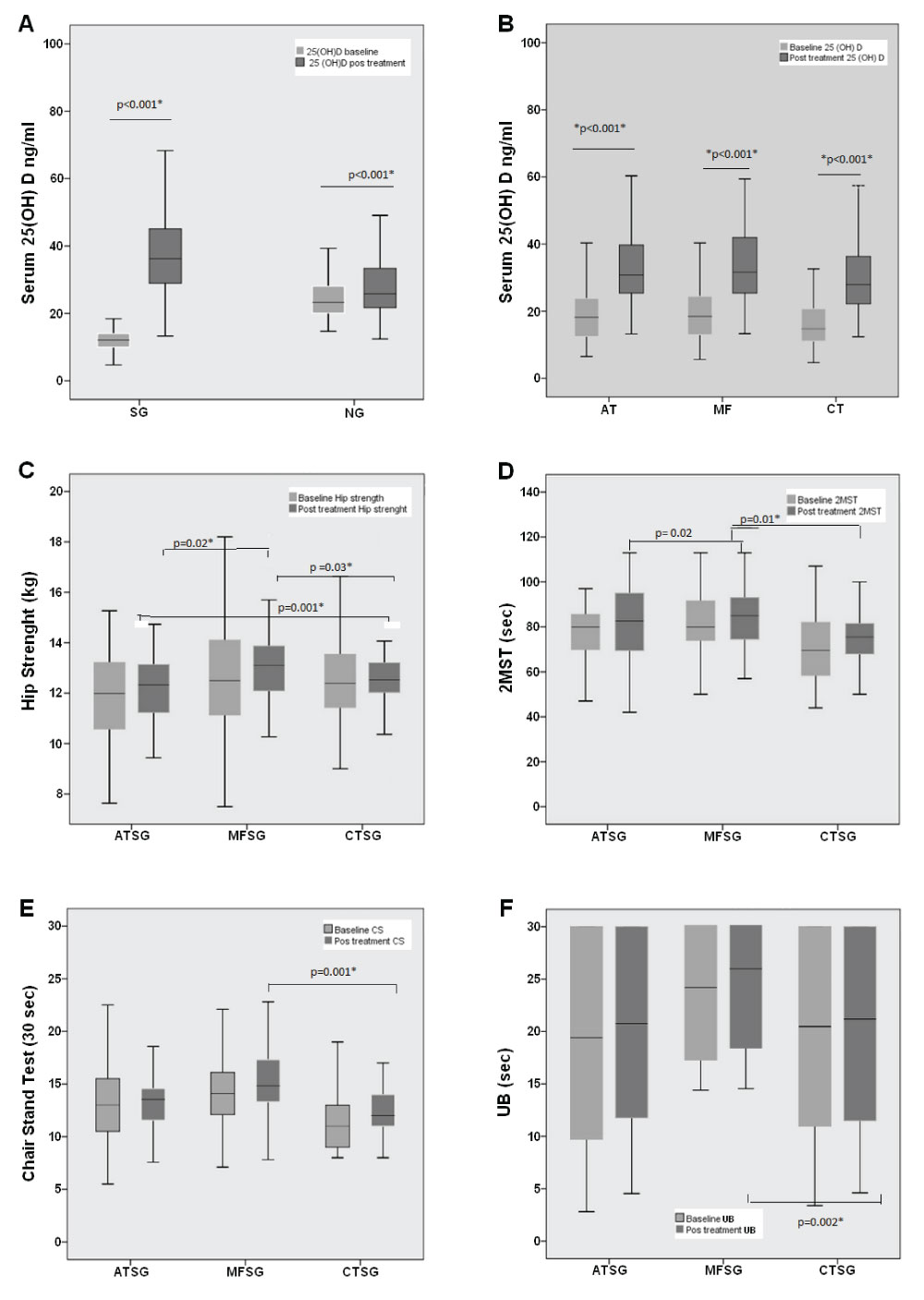 Figure 2: Supplementation of 25(OH)D among groups and significant superiority of the multifunctional fitness group (MFSG) when compared to aquatic training group (ATSG) and supplemented control group (CTSG) in the physical tests. (A) Serum vitamin D concentration at baseline and post-treatment; (B) serum vitamin D concentration at baseline and post-treatment among the aquatic training (AT), multifunctional fitness (MF), and control treatment (CT) groups; (C) hip strength test results among ATSG, MFSG, and CTSG at baseline and post-treatment; (D) 2-min step test results among ATSG, MFSG, and CTSG at baseline and post-treatment; (E) chair stand test results among ATSG, MFSG, and CTSG in baseline and post-treatment; (F) Unipedal balance test with visual control results among ATSG, MFSG, and CTSG at baseline and post-treatment. View Figure 2
Figure 2: Supplementation of 25(OH)D among groups and significant superiority of the multifunctional fitness group (MFSG) when compared to aquatic training group (ATSG) and supplemented control group (CTSG) in the physical tests. (A) Serum vitamin D concentration at baseline and post-treatment; (B) serum vitamin D concentration at baseline and post-treatment among the aquatic training (AT), multifunctional fitness (MF), and control treatment (CT) groups; (C) hip strength test results among ATSG, MFSG, and CTSG at baseline and post-treatment; (D) 2-min step test results among ATSG, MFSG, and CTSG at baseline and post-treatment; (E) chair stand test results among ATSG, MFSG, and CTSG in baseline and post-treatment; (F) Unipedal balance test with visual control results among ATSG, MFSG, and CTSG at baseline and post-treatment. View Figure 2
This study is the first to show that 25(OH)D supplementation in active elderly women who have been exercising improves functional test results. In a systematic review, Annweiler, et al. [26], provided important evidence suggesting that the 25(OH)D concentration was positively associated with walking velocity among elderly individuals [26]. They emphasized the necessity to conduct other studies, and we confirmed that 25(OH)D influences the TUG test results in elderly women.
By contrast, in our study, the TUG test result improved in CTSG, indicating that the 25(OH)D supplementation was effective in enhancing the agility, dynamic balance, and mobility in elderly women.
In a review by Daly (2010), the term "interaction" explained the combined effects of two or more variables as being more than the amount of their individual effects [27]. Thus, an "interaction" effect of 25(OH)D with multifunctional fitness programs implies multiplicative results in elderly women. In that review, regular progressive resistance with power training and balance exercises, such as those in multifunctional fitness programs, plus 25(OH)D supplementation (700-1,000 IU/day) were important for maintaining and optimizing strength and muscle function in elderly women.
There is evidence that 25(OH)D supplementation can improve muscle function and functional independence in the elderly [28,29]. In contrast, on the basis of meta-analyses [30], 25(OH)D supplementation has a small but positive impact on global muscle strength, more specifically of the lower limb, in elderly people. In addition, vitamin D levels depending on serum 25(OH)D concentrations in the elderly were positively correlated with improved functional performance and strength, as well as suppressed rates of decline in performance, which suggests a relationship between vitamin D and neuromuscular function [6].
In a recent publication, a larger meta-analysis consisting of 29 vitamin D supplementation trials has also confirmed a small but positive effect on muscle strength. The mechanisms by which vitamin D affects muscle strength and function may be can mediated by VDR. The number of VDRs present in human muscle tissues has been shown to decline with age. Thus, an important recent finding is that VDR expression can be changed by vitamin D supplementation [8].
The study by Ceglia, et al. [31] showed that vitamin D3 supplementation (4000 IU/day over four months) increased muscle mass or muscle-specific VDRs [31]. They concluded that the supplementation increased muscle fiber size by 10% and increased intramyonuclear VDR concentration by 30% in elderly women with insufficient vitamin D. This evidence was supportive of the clinical effects of vitamin D supplementation on muscle function [8].
In addition to determining a suitable dosage of 25(OH)D supplementation for elderly people to improve their physical performance, it is also important to evaluate their exercise routine. Unfortunately, this reluctance may be attributed to a lack of knowledge concerning appropriate exercises for elderly people with regard to potential risks and benefits of different modalities [32].
In our study, NG also showed a significant increase in the level of 25(OH)D. Even though this improvement was uncontrolled, it collaborated to improve the physical performance of the elderly people. Other studies that corroborate with these results include the trial of Moreira Pfrimer, et al. [33] involving postmenopausal women who practiced aquatic exercise, with the control group receiving 1000 IU of cholecalciferol supplementation for 24 weeks. The authors showed that both groups had significantly higher serum levels of 25(OH)D [34]. Similarly, after supplementation of 25(OH)D, even without regular practice of physical exercises, other study found an increase in muscular strength [33].
This study has limitations. There was no randomization, because the intervention was implemented in groups that were already involved in the exercise modalities for at least one year. We did not evaluate the factors that influenced serum vitamin D levels during the study such as lifestyle, nutritional data, and sunlight exposure. We evaluated the supplementation of vitamin D in individuals following different modalities of physical exercises to test their physical performance, and therefore, this was not a blind study. The sample was confined to only one geographical area. The results are relevant to a community of physically independent elderly women.
This study revealed that vitamin D levels were important for better physical exercise performance among elderly women. Before starting an exercise program, elderly people should be aware of their vitamin D levels.
In conclusion, the interaction of serum 25(OH)D levels with physical exercises can contribute to the maintenance of functional independence in activities of daily living.
The authors declare no potential conflict of interest with respect to the authorship and/or publication of this manuscript.
This study was supported by FAPESP (Fundação de Amparo à Pesquisa do Estado de São Paulo; Grant no. 09/53658-8). Thanks to the Social Service of Commerce for the opportunity to perform most of the study in its facilities.