Bullous pemphigoid (BP) is a rare autoimmune subepithelial blistering disease affecting mostly older adults [1]. Studies have calculated an annual incidence ranging from 2.4% to 21.7% per million population. While triggers are found in less than 15% of cases [2,3], drugs are well-known trigger factors. Recently, dipeptidyl peptidase-4 inhibitors (DPP4i) have been associated to this condition [4]. Our study presents four cases of older adults with BP associated to the use of DPP4i. The relevance of this study is to emphasize the association between these drugs and BP in older adults where polypharmacy is frequent.
Geriatric dermatology, Polypharmacy, Diabetes, Autoimmune
BP: Bullous Pemphigoid; DPP4i: Dipeptidyl peptidase-4 inhibitors; Di-BP: Drug-Induced Bullous Pemphigoid; CDC: Centers for Disease Control and Prevention; ADA: American Diabetes Association; FDA: Food and Drug Administration; EASD: European Association for the Study of Diabetes; F: Female; M: Male; T2DM: Type 2 Diabetes Mellitus; CKD: Chronic Kidney Disease; SAH: Systemic Arterial Hypertension; HF: Heart Failure; COPD: Chronic Obstructive Pulmonary Disease; HCV: Hepatitis C Virus Infection; Mg: Milligrams
Bullous pemphigoid (BP) is an autoimmune subepithelial blistering disease [1] with an estimated annual incidence of PA between 2.4 to 21.7 per million people [3]. During the last decade, there has been an increase in its incidence [5-7]. In Mexico, a retrospective study reported an incidence of 15 cases per 100,000 people between 2006-2011 [1]. Although the pathogenesis of BP is still not completely understood, there is strong evidence of autoantibodies against the hemidesmosomal anchoring proteins BP180 and BP180, as well as an imbalance between regulatory T cells and autoreactive helper T cells [8]. Risk factors include advanced age, neurological diseases (eg, dementia, Parkinson's disease, or psychiatric disorders), malignancies, and medications [9].
Drug-induced bullous pemphigoid (Di-BP) is a variant of BP secondary to the topical or oral use of some drugs [10]. The clinical picture is similar to classic BP, starting with a nonbullous phase consisting of urticarial-like wheals along with scaly patches, followed by a bullous phase characterized by tense, pruritic blisters on normal skin or with an erythematous and edematous base primarily on the skin, axillary folds, lower abdomen, groin areas, and inner thighs [11,12]. The mucosa may be involved in 10% to 25% of cases, and healing with post inflammatory hyperpigmentation may occur with the risk of scarring. Milia is seen at sites of previous blisters or erosions [11-13].
Di-BP is diagnosed in less than 15% of patients [2] as it is difficult to differentiate it from classic BP. The most suggestive risk factor being polypharmacy, recent use of any new medication, presentation at early ages, significant mucosal condition, and histopathology with marked eosinophilic infiltrate and necrotic keratinocytes or intradermal vesicles [2]. There are multiple drugs reported as triggers of Di-BP; however, the level of evidence is variable (Table 1). Dipeptidyl peptidase-4 (DPP4i) inhibitors are among the drugs with the highest association level with BP [14,15].
Table 1: Medications implicated in drug-associated bullous pemphigoid. View Table 1
We describe four cases of DPP4i-induced BP in patients older than 70 years with a history of polypharmacy to emphasize the challenge during evaluation in these patients and the importance of diagnostic suspicion.
Clinical and histological medical reports of patients presenting with BP related to the use of DPP4i in two hospitals from Mexico between July 2018 and May 2022 were reviewed.
Results show data from two men (50%) and two women (50%) aged 70 to 87-years-old with a mean age of 80 years. Table 2 describes the clinical characteristics of the four patients. Three of the patients presented non-bullous and bullous phases, however, one presented just in the bullous phase. In three cases the use of linagliptin was documented, while the use of sitagliptin was documented in only one case. The latency between the use of DPP4i and the appearance of the lesions varied from one week to one year, with a mean of 17 weeks. All patients presented with a disseminated condition. Figure 1 describes the manifestation of some patients. Three patients underwent a biopsy and have histopathological confirmation; however, clinical diagnosis only was done in one patient. Figure 2 describes the biopsy of one patient. The DPP4i was withdrawn from all patients and treatment with prednisone 15 mg to 20 mg/day was started. Two patients also received antibiotic treatment initially (one patient doxycycline and the other lymecycline). One patient also received topical clobetasolas an adjuvant. Partial response to treatment was seen in three patients and one showed a complete response. Response time varied from one week to five months. Three patients presented a chronic evolution and one patient is still in follow-up. Only one patient did not require maintenance therapy as a complete response was seen after stopping the DPP4i. One patient presented milia during the resolution phase. One patient had to be hospitalized for a BP complication. All patients were taking more than one drug at diagnosis.
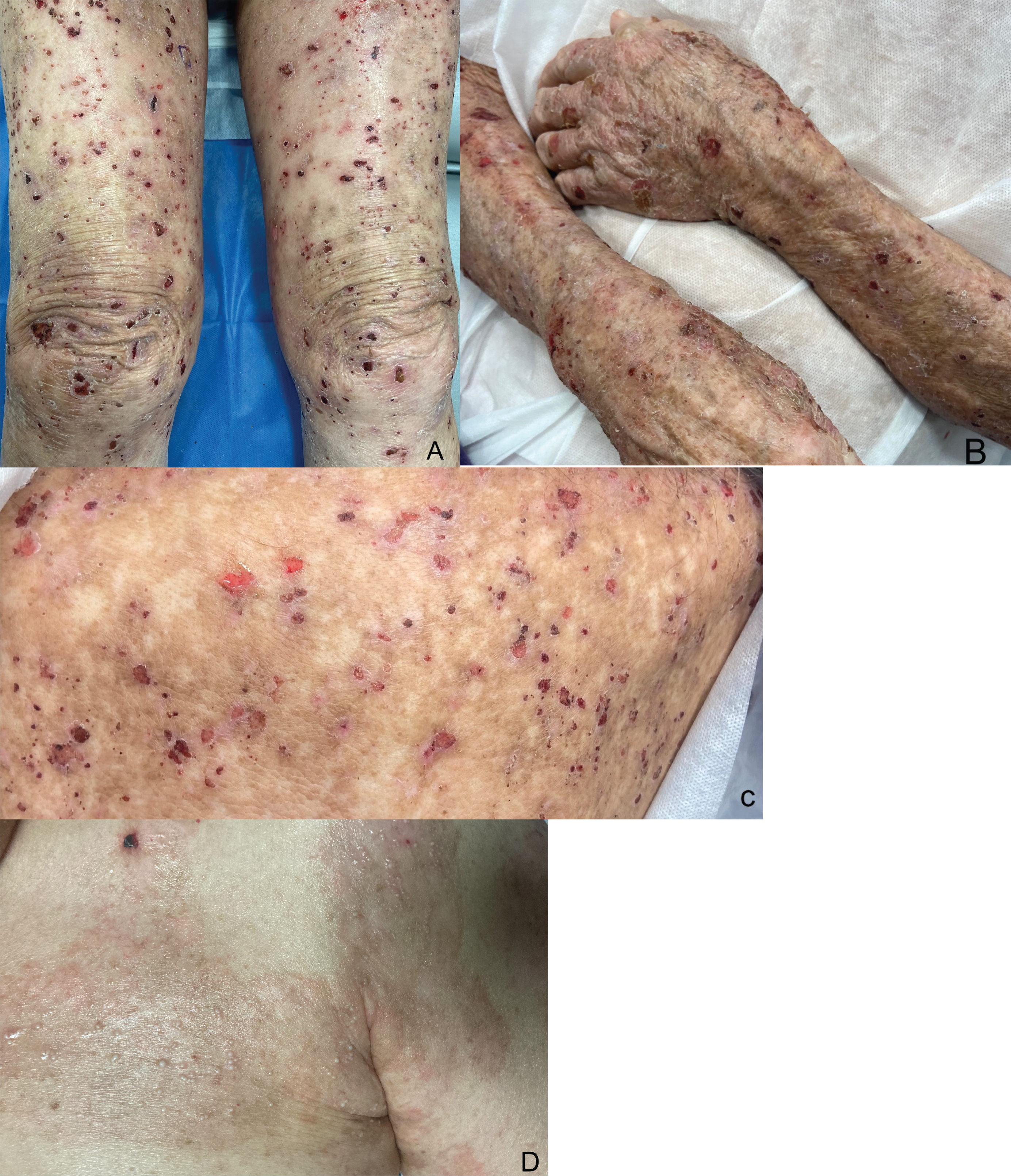 Figure 1: Clinical manifestations of iDPP-4-induced bullous pemphigoid. A and B) Tense blisters and multiple exulcerations, atrophic-looking scars and residual spots on the lower pelvic limbs and arms. B) In addition, multiple traces of scratching and xerosis are observed on the arms; C) Erosions and blood crusts in the posterior thorax; D) Atrophic scars and milia cysts in the anterior chest.
View Figure 1
Figure 1: Clinical manifestations of iDPP-4-induced bullous pemphigoid. A and B) Tense blisters and multiple exulcerations, atrophic-looking scars and residual spots on the lower pelvic limbs and arms. B) In addition, multiple traces of scratching and xerosis are observed on the arms; C) Erosions and blood crusts in the posterior thorax; D) Atrophic scars and milia cysts in the anterior chest.
View Figure 1
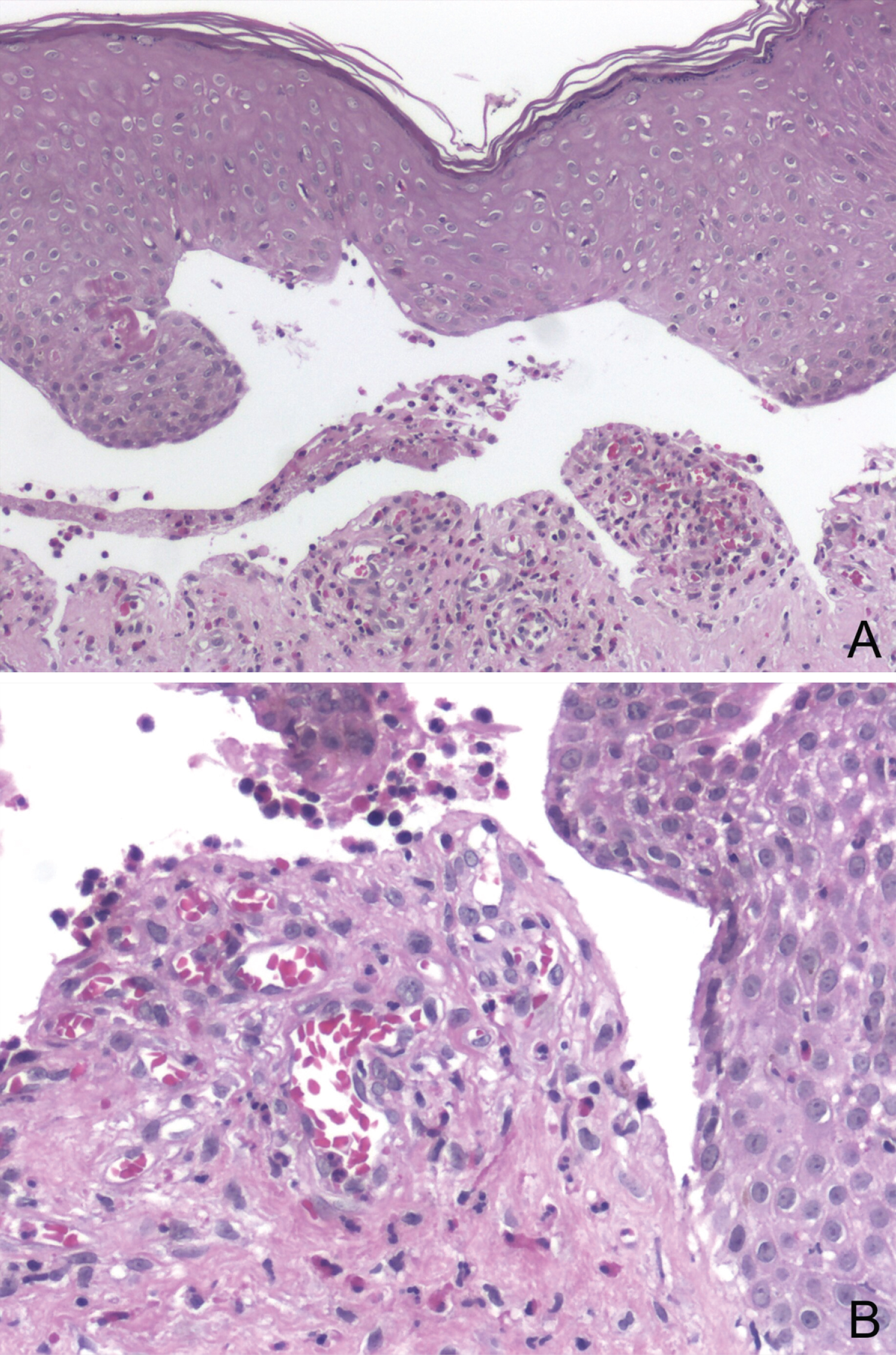 Figure 2: H&E stain of a biopsy of the skin edge of an established blister. A and B) Subepidermal blister with an edematous papillary dermis; B) Inflammatory infiltrate rich in eosinophils and fibrin.
View Figure 2
Figure 2: H&E stain of a biopsy of the skin edge of an established blister. A and B) Subepidermal blister with an edematous papillary dermis; B) Inflammatory infiltrate rich in eosinophils and fibrin.
View Figure 2
Table 2: Characteristics of four patients with Bullous Pemphigoid induced by DPP4i. View Table 2
DPP4is (alogliptin, anagliptin, linagliptin, sitagliptin, vildagliptin, and teneligliptin) are drugs used for the treatment of type 2 diabetes mellitus. They were Approved by the Food and Drug Administration (FDA) in 2006 and were added to the guidelines of the American Diabetes Association (ADA) and the European Association for the Study of Diabetes (EASD) in 2018 [16]. These drugs work by increasing the levels of incretin hormones, glucagon-like peptide-1, and glucose-dependent insulinotropic peptide resulting in improved b-cell responsiveness to glucose concentrations and suppression of glucagon secretion [17]. The minimal risk of hypoglycaemia, their cardio protective and nephroprotective effect, as well as the lack of weight gain are reasons for considering them a treatment of choice in older adults [18,19].
Commonly reported short-term side effects include upper respiratory tract infections, urinary tract infections, headaches, and gastrointestinal disturbances [20,21]. While their long-term safety has not been established, some of their serious side effects include acute pancreatitis, liver dysfunction, hypersensitivity reactions, and severe joint pain. Despite a lack of robust evidence, the occurrence of any of the above warrants an immediate withdrawal of the drug [22-24].
The first cases of BP associated with DPP4is were reported in 2011, since then evidence supporting their association has increased [3,4,14,15,25-42]. Although the pathophysiology is not fully yet understood, it is hypothesized that DPP4 is modify the immune response and/or alter the antigenic properties of the epidermal basement membrane resulting in bullous pemphigoid [43]. Currently, the incidence of DPP4i-induced BP is estimated to be 0.86/1000 patients [44].
Results from studies looking at the strength of association between DPP4i and BP differs. The systematic review and meta-analysis by Yang, et al. revealed an OR = 7.38 (CI = 7.38, 95% CI 2.00-27.2, I2 = 0, P = 0.003) [4]. It seems that the association is stronger with vildagliptin (OR = 10.7, 95% CI, 5.1-22.4) compared to linagliptin (OR = 6.7, CI, 2.2-19.7), with little or no significant association when looking at sitagliptin or saxagliptin [4,14,35,40]. Its association to alogliptin, anagliptin, and teneligliptin has been reported only in scarce cases and pharmacovigilance databases [28,31,34,43].
This case series exemplify the significant difficulty of diagnosing DPP4i-induced BP because of polypharmacy in older adults. Polypharmacy is defined as the use of multiple drugs (usually five or more) per day. Data from the CDC (Centers for Disease Control and Prevention) reveals that a third of adults between the ages of 60 and 70 have polypharmacy, increasing their risk of drug side effects [45]. Patients with polypharmacy may use several drugs associated to BP. Patients from this series used not only DPP4i but also angiotensin-converting enzyme inhibitors, proton pump inhibitors, diuretics, and other groups of drugs related to BP which could synergize the effects of the DPP4i.
A complete medical history is crucial to suspect and withdraw triggering drug(s) avoiding complications, including the use of high-dose steroids or intravenous immunoglobulin. Diagnosis is based on clinical symptoms and histological and immunopathological findings. Latency lasts an average of 7 months as reported by the FDA [28], however, it can vary from 8 days to 37 months [43]. Our cases presented a lower mean latency than what has been reported. While studies suggest a lower age mean for drug-induced BP when compared to classic BP, other studies similar to ours show that DPP4i occurs at older ages finding a relationship between the start of the DPP4i and the presentation of BP symptoms with an improvement upon withdrawal confirming the diagnosis by the criteria of the World Health Organization System-Uppsala Monitoring Center [42,46].
Typical findings of drug-induced BP that contrast with classic bP include: Intraepidermal vesicles, necrotic keratinocytes, thrombus formation, and marked eosinophilic infiltrate [10]. Anti-BP180/BP230 enzyme-linked immunosorbent assays can also differentiate DPP4i-associated BP from conventional BP [10,47]. In our setting, biopsy is not available for all because of financial and infrastructure limitations.
Patients with drug-induced BP have a good prognosis; most of our patients presented a chronic and persistent course requiring maintenance treatment, a complication that has been previously reported [10]. Remission is achieved by withdrawing triggers and topical and/or systemic steroids plus adjuvant treatment such as doxycycline, dapsone, methotrexate, azathioprine, mycophenolate mofetil, or immunoglobulin, depending on each patient [36]. Except for one patient, all patients were immediately withdrawn from DPP4i as patients who continue to take DPP-4i are reported to relapse more than those who immediately discontinued the drugs [36].
The increasing aging of the population has paralleled the rise in the prevalence of chronic conditions and polypharmacy. The escalated use of medications such as antidiabetic drugs including DDP4i could explain the increased incidence of BP. Physicians should be watchful when using DP4i and other BP-associated drugs in older adults in order to suspect BP early and withdraw the suspected drugs as drug-induced BP usually to withdrawal of the suspected drug with little or no recurrence [13]. Being up to date on drugs that trigger this condition is crucial to improve treatment and prognosis in this group of patients where polypharmacy is common.
All participants signed informed consent forms.
Data is available upon request to the corresponding author.
The authors report no conflicts of interest in this work.
This work received no funding.
Axel Daniel Velázquez Ramos and Sonia Toussaint Caire collected data.
We certify that all listed authors have substantially contributed to the development of the concept of the manuscript, in the generation of the cases presented, and/or in the writing and editing of the manuscript.