Although mandibular advancement devices (MADs) are utilized for the management of mild to moderate OSA, there are concerns about temporo-mandibular joint (TMJ) issues. Biomimetic oral appliance therapy (BOAT) differs from MADs as it aims to achieve midfacial redevelopment in combination with mandibular repositioning. In this study, we tested the hypothesis that mild to moderate cases of OSA can be addressed with combined maxillo-mandibular correction using BOAT.
Nineteen adults diagnosed with mild to moderate OSA, following an overnight home sleep test (HST) that had been interpreted by a physician, participated in this study. Each subject was treated by a dentist with advanced training in dental sleep medicine. At each monthly follow-up, the devices were adjusted to optimize their efficacy. The mean apnea-hypopnea index (AHI), respiratory disturbance index (RDI) and oxygen desaturation index (ODI) of the study sample was calculated prior to and after BOAT. The findings were subjected to statistical tests.
Prior to treatment the mean AHI of the study subjects was 12.8hr-1 ± 5; the mean RDI was 18.6hr-1 ± 8.2, and the ODI was 6.3% ± 3.5. Follow-up HSTs after approximately 9 mos. indicated the mean AHI decreased significantly to 6.2hr-1 ± 2.9 after BOAT (p < 0.001), which represents a fall in the mean AHI by 51.5% for the study sample. The mean RDI fell to 12.3hr-1 ± 6.9 (p < 0.001), and the ODI was improved to 2.6% ± 1.7 (p < 0.001).
This study suggests that combined maxillo-mandibular correction may be a useful method of managing mild to moderate cases of OSA in adults, and represents an alternative to MADs and continuous positive airway pressure therapy. However, further studies are needed to determine the additional advantages of combined maxillo-mandibular correction.
Biomimetic, Oral appliance therapy, Palatal expansion, Mandibular advancement
MAD: Mandibular Advancement Device; OSA: Obstructive Sleep Apnea; TMJ: Temporo-Mandibular Joint; BOAT: Biomimetic Oral Appliance Therapy; HST: Home Sleep Test; AHI: Apnea-Hypopnea Index; RDI: Respiratory Disturbance Index; ODI: Oxygen Desaturation Index; CPAP: Continuous Positive Airway Pressure
Obstructive sleep apnea (OSA) is a common sleep disorder with serious medical implications. Although several therapeutic options are available for this condition, patient compliance may result in inadequate treatment. For example, continuous positive airway pressure (CPAP) therapy is not well-tolerated due to unwanted side effects, and patient compliance can be as low as 50% [1]. Thus, other treatment options for OSA include: weight loss, positional therapy, oral appliance therapy, hypoglossal nerve stimulation, and surgery. Indeed, in a pilot study, the surgical removal of extensive, bilateral torus mandibularis decreased the apnea-hypopnea index (AHI) by 15% in a 47-year-old male with moderate OSA [2]. However, instead of using various treatments alone, clinical outcomes might be improved by combining different treatment modalities [2]. For instance, surgical treatment is used in cases of OSA where CPAP or mandibular advancement device (MAD) failures may have occurred. In this respect, the role of surgery is to facilitate long-term device tolerance or use [3]. For example Başal, et al. described a severe case of OSA that was treated with combined surgical methods [4]. Post-operatively, the AHI was radically-reduced while using CPAP and, post-surgically, the CPAP intolerance was eliminated. Thus, a combination of surgical methods might increase CPAP compliance.
Indeed, other treatments such as weight loss, nasal surgery and positional devices may be combined with surgical procedures of the upper airway. Azbay, et al. studied multilevel upper airway surgery on CPAP tolerance [5].
Procedures such as septoplasty and uvulo-pharyngo-palatoplasty (with or without modified tongue base suspension) were deployed. Post-operatively, about 50% of the study sample reported that their symptoms were resolved, and about 50% tolerated CPAP without problems. On the other hand, Hui, et al. investigated the therapeutic effects of a MAD combined with either nasal or pharyngeal surgery [6]. The AHI of the combined surgical group was significantly lower post-operatively, and the authors concluded that surgical enlargement of the nasal and pharyngeal airways combined with a MAD is an effective treatment in patients with severe OSA. Similarly, Wang, et al. evaluated the effect of CPAP combined with a MAD in the treatment of severe OSA [7]. After 3 months of treatment, the AHI in the CPAP-combined with MAD group and the CPAP-alone group were significantly lower than that in the MAD-group alone; but there was no difference in AHI between the CPAP-combined with MAD group and the CPAP-alone group. However, the oxygen desaturation index (ODI) in the CPAP-combined with MAD group was significantly lower than that in the CPAP-alone group. Therefore, the authors concluded that CPAP combined with a MAD was better than CPAP or MADs alone in the treatment of severe OSA.
While MADs have long been successfully deployed for the management of OSA, either in isolation or in combination, there is some concern about temporo-mandibular joint (TMJ) symptoms, changes in the occlusion and unwanted tooth movements [8]. Biomimetic oral appliance therapy (BOAT) differs from conventional MADs as it aims to correct the nasal airway [9] through midfacial redevelopment [10] in combination with mandibular repositioning, which aims to improve the oropharyngeal airway in adults [11]. But, there are very few studies in the literature that have investigated a combination of midfacial development in conjunction with mandibular repositioning in the treatment of OSA. Therefore, in this study we tested the hypothesis that mild to moderate cases of OSA can be addressed using a combined maxillo-mandibular protocol in adults.
After obtaining informed consent, we included 19 consecutive adults that had been diagnosed with mild to moderate OSA following an overnight home sleep test (HST). The rights of the subjects were protected by following the Declaration of Helsinki. Inclusion criteria were: subjects aged > 21 yrs; diagnosed without severe OSA following an overnight sleep study that had been interpreted by a physician; good oral appliance compliance; no history of hospitalization for craniofacial trauma or surgery; no congenital craniofacial anomalies, and dentate upper and lower arches. The exclusion criteria included: age < 21 yrs; lack of oral appliance compliance; active periodontal disease; excessive tooth loss; poor oral hygiene, and systemic bisphosphonate therapy.
Following a confirmed diagnosis of mild to moderate OSA by a medical physician, careful history-taking and craniofacial examination was undertaken for each subject by a dentist (SEC) with advanced training in dental sleep medicine, and each subject was treated using BOAT. BOAT differs from the conventional MADs as it aims to remodel the upper airway through combined maxillo-mandibular correction with midfacial development and mandibular repositioning. Therefore, a bite registration was obtained in the upright-sitting position with the jaw posture corrected in the antero-posterior and vertical axes specific for each subject, using a sibilant phoneme registration protocol or 'phonetic bite' [12]. Upper and lower polyvinyl siloxane impressions were also obtained. The upper model was then mounted on an articulator and the lower model was mounted relative to the upper model, using the bite registration captured. Blinded to one investigator (GDS), a biomimetic oral appliance with mandibular repositioning capabilities (mandibular-Repositioning Nighttime Appliance: mRNA appliance®; Figure 1) was prescribed for the study population. The mRNA appliance® is FDA-cleared as a Class II medical device for mild to moderate cases of OSA. The BOAT protocol has previously been deployed to address maxillo-mandibular and upper airway deficiencies in both children and adults [13-18]. All appliances used in this study had: a labial bow; 6 anterior 3-D axial springs™; a midline screw; bilateral occlusal coverage; retentive slots (Figure 1); a beaded, pharyngeal stent (Figure 2), and a nighttime mandibular repositioning component (Figure 3) with a screw-fin mechanism to titrate the mandibular advancement in 1mm increments. All subjects were instructed to wear the appliances during the evening and at nighttime (for approx. 12-16hrs. in total), but not during the day time and not while eating, partly in line with the circadian rhythm of tooth eruption [19], although this only occurs in children. Proffit [20] notes that an appliance needs to be worn for at least 8hrs in the mouth to have a clinical effect. Written and verbal instructions were given to all subjects.
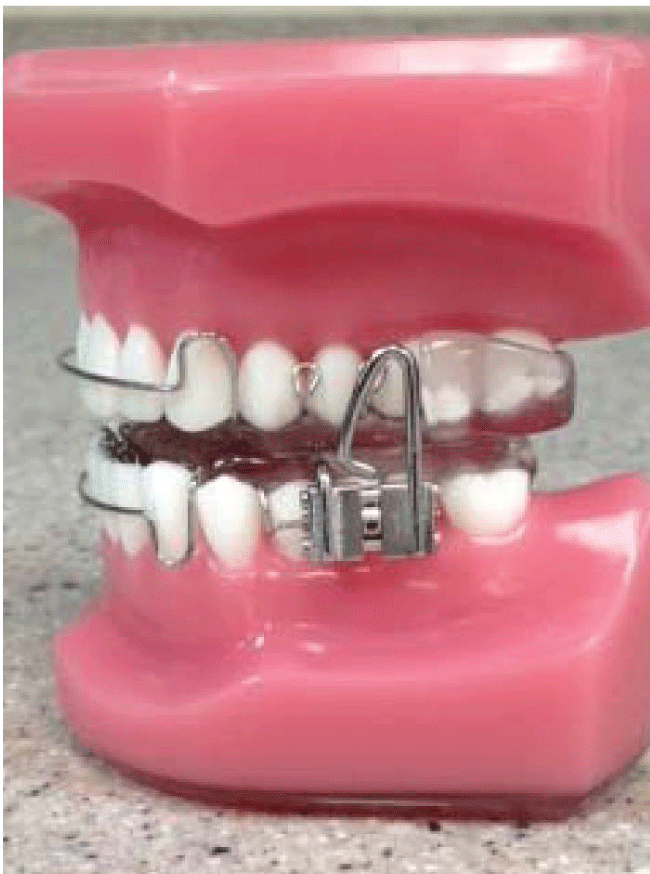 Figure 1: The biomimetic oral appliance (mandibular-Repositioning Nighttime Appliance: mRNA appliance®) used in this study. The mRNA appliance® is FDA-cleared as a Class II medical device for mild to moderate cases of obstructive sleep apnea. Note that the lower component has a screw-fin mechanism to titrate the mandibular repositioning in 1mm increments.
View Figure 1
Figure 1: The biomimetic oral appliance (mandibular-Repositioning Nighttime Appliance: mRNA appliance®) used in this study. The mRNA appliance® is FDA-cleared as a Class II medical device for mild to moderate cases of obstructive sleep apnea. Note that the lower component has a screw-fin mechanism to titrate the mandibular repositioning in 1mm increments.
View Figure 1
 Figure 2: The upper component of the biomimetic oral appliance used in this study with: 6 anterior 3-D axial springs™, a midline screw, bilateral occlusal coverage, retentive slots, and a labial bow.
View Figure 2
Figure 2: The upper component of the biomimetic oral appliance used in this study with: 6 anterior 3-D axial springs™, a midline screw, bilateral occlusal coverage, retentive slots, and a labial bow.
View Figure 2
 Figure 3: The upper component of the biomimetic oral appliance used in this study showing the beaded, pharyngeal stent, which was adjusted to prevent the tongue and uvula form obstructing the upper airway.
View Figure 3
Figure 3: The upper component of the biomimetic oral appliance used in this study showing the beaded, pharyngeal stent, which was adjusted to prevent the tongue and uvula form obstructing the upper airway.
View Figure 3
BOAT requires professional adjustments approximately every 4 weeks, and all subjects reported for review each month. At each monthly follow-up, examination for the progress of craniofacial correction was recorded. Adjustments of the devices were performed to optimize their efficacy. The subjects were encouraged to maintain their treatment protocol as outlined at the beginning of treatment. Specifically, the tension/retention of the device was maintained by advancing the expansion screw in the upper component (Figure 1) by approx. 0.25mm weekly. The labial bow was adjusted so that it did not contact the labial surfaces of the anterior teeth. Gradually, the fitting surface acrylic/posterior border of the device was reduced to permit the hard palate to remodel inferiorly. The occlusal acrylic was also selectively equilibrated and gradually removed, to permit the lower teeth to track the changes in the occlusion. The beaded, pharyngeal stent (Figure 2) was adjusted to prevent the tongue and uvula from obstructing the upper airway. Finally, the 3D axial springs (Figure 1) were adjusted so that only gentle pressures were transmitted to the teeth and surrounding tissues. The functionality of the devices was checked with the subject activating a mild force on biting. Advancement of the mandible and development of the lower arch was implemented using the separate lower component (Figure 3). To permit arch re-coordination, the midline expansion screw of the lower component was activated. In addition, the screw-fin mechanism (Figure 3) was adjusted in 1mm increments to reposition the mandible in the position required to maintain an adequate upper airway at nighttime for sleep. The post-treatment HSTs were done approx. 9mos later and were interpreted by a medical physician. The mean AHI, RDI and ODI of the study sample were calculated prior to and after BOAT, and the findings were subjected to statistical analysis, using paired t-tests.
Figure 4 is an example that shows the changes in facial profile and head posture in a subject that participated in this study. Figure 5 is an example that shows study models indicating the intra-oral, maxillo-mandibular changes in a different subject who underwent the same procedure. Figure 6 shows the upper airway changes in another case treated using the same combined maxillo-mandibular protocol.
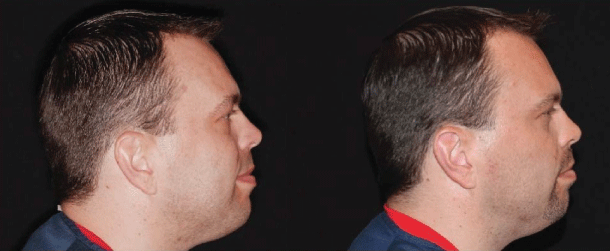 Figure 4: The changes in facial profile and head posture in a subject that participated in this study. The figure on the left shows the subject prior to treatment. The figure on the right shows the same subject after approx. 9 mths using the combined maxillo-mandibular protocol.
View Figure 4
Figure 4: The changes in facial profile and head posture in a subject that participated in this study. The figure on the left shows the subject prior to treatment. The figure on the right shows the same subject after approx. 9 mths using the combined maxillo-mandibular protocol.
View Figure 4
 Figure 5: Study models showing the intra-oral changes in a subject who underwent the combined maxillo-mandibular protocol. Note that the minimum inter-molar bone width of the maxilla has increased from 35.5mm to 37.5mm and the arch has become more symmetric and less crowded. Similarly, the mandibular arch shows improved alignment and width simultaneously.
View Figure 4
Figure 5: Study models showing the intra-oral changes in a subject who underwent the combined maxillo-mandibular protocol. Note that the minimum inter-molar bone width of the maxilla has increased from 35.5mm to 37.5mm and the arch has become more symmetric and less crowded. Similarly, the mandibular arch shows improved alignment and width simultaneously.
View Figure 4
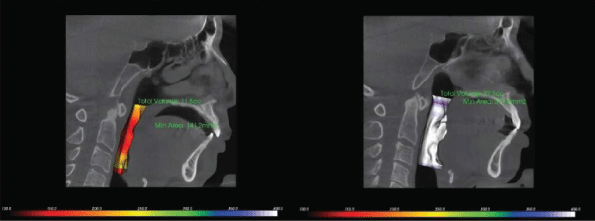 Figure 6: Mid-sagittal slices from a 3D cone-beam computerize axial scan, showing the upper airway changes using the same combined maxillo-mandibular protocol. Note that the upper volume has increased from 11.8cm3 pre-treatment to 27.5cm3 post-treatment, while the minimum cross-sectional area has increased from 141.2cm2 to 379.8cm2 post-treatment, using the combined maxillo-mandibular protocol.
View Figure 6
Figure 6: Mid-sagittal slices from a 3D cone-beam computerize axial scan, showing the upper airway changes using the same combined maxillo-mandibular protocol. Note that the upper volume has increased from 11.8cm3 pre-treatment to 27.5cm3 post-treatment, while the minimum cross-sectional area has increased from 141.2cm2 to 379.8cm2 post-treatment, using the combined maxillo-mandibular protocol.
View Figure 6
Prior to treatment the mean AHI of the study subjects was 12.8hr-1 ± 5; the mean RDI was 18.6hr-1 ± 8.2, and the ODI was 6.3% ± 3.5. The follow-up HSTs done after approx. 9 mos. showed that the AHI decreased significantly (p < 0.001) to a mean value of 6.2hr-1 ± 2.9 after BOAT, which represents a fall in the mean AHI by 51.5% for the study sample. The mean RDI fell to 12.3hr-1 ± 6.9 (p < 0.001), and the ODI was improved to 2.6% ± 1.7 (p < 0.001). Table 1 summarizes the overall findings. Therefore, it appears that upper airway parameters can be improved in adults with mild to moderate OSA by targeting midfacial correction in combination with mandibular advancement using BOAT.
Table 1: Summary of sleep parameters assessed in this study. View Table 1
Obstructive sleep apnea (OSA) is associated with increased cardiovascular diseases, such as high blood pressure and heart failure, as well as metabolic morbidities. In addition, the risk of OSA-related motor vehicle collisions is increased. Treatment of OSA includes changes in diet and lifestyle, CPAP, various oral devices, and upper airway surgical treatments [21]. In this study, we investigated the novel concept that combined maxillo-mandibular oral appliance therapy may be a useful method of managing mild to moderate cases of OSA in adults. This procedure represents an alternative to MAD therapy, where there may be TMJ concerns, or to CPAP therapy, where there might be compliance issues, although we did not directly compare MADs with combined maxillo-mandibular correction in this study. In fact, there are very few studies on simultaneous maxillo-mandibular correction. Dieltjens, et al. used a combination of positional therapy with a MAD for OSA [22]. While, both MAD and positional therapy were individually effective in reducing the AHI, the combination further reduced the overall AHI compared with either method alone. Therefore, there may be advantages to combined therapies in contrast to using one intervention alone, but there may be confounding factors.
Obesity is a major risk factor in the development of OSA. Indeed, Chirinos, et al. assigned patients with obesity and moderate-to-severe OSA to receive treatment with either CPAP, weight-loss intervention, or a combination of the two [23]. The combined interventions showed reductions in insulin resistance and serum triglyceride levels, but, while these changes were observed in the weight-loss group they were not noted in the group receiving CPAP alone. However, weight loss has a variable effect on the severity of OSA, and differences in the effect of weight loss on OSA may be due to underlying craniofacial characteristics. In this respect, Naughton, et al. studied mandibular length in association with changes in AHI following weight loss [24]. Their analysis indicated that the change in AHI correlated with a shorter mandibular body length. Thus, weight loss for severe OSA in obese patients may be useful in those with a shorter mandibular body length. As we did not measure mandibular lengths in this present study, additional studies are needed using 3D imaging techniques to quantify the craniofacial changes induced with combined BOAT. Nevertheless, the mandibular repositioning device we deployed in combination with midfacial redevelopment decreased the mean AHI by over 50% and to < 10hr-1 in a sample of non-obese, adult patients, which represents a successful outcome in a medical context.
Zhang, et al. explored soft palatal surgery combined with uvulo-pharyngoplasty and inferior turbinate radiofrequency for the treatment of OSA [25]. The AHI in their study sample was 16hr-1 at 6mos but relapsed to about 21hr-1 at 12mos post-operatively. Therefore, not all combination protocols yield enhanced results. In contrast, following a study that demonstrated beneficial changes after surgical correction of skeletal Class III cases [26], double jaw surgery is currently considered advantageous because it produces an increase in the pharyngeal airway dimensions. In this present study, non-surgical, double jaw correction also appears to enhance the upper airway (Figure 6) and this was confirmed since the mean ODI improved to 2.6% in the sample we tested after treatment. In addition, it is likely that the sleep architecture may have also improved since the mean RDI fell to 12.3hr-1 in our present study. Therefore, although long-term follow up using a larger sample size is needed to make more definitive conclusions on these initial findings, this present study suggests that combined, maxillo-mandibular, biomimetic oral appliance therapy with midfacial development and mandibular advancement, may be a useful method of managing non-severe cases of OSA in adults, and may represent a viable alternative to CPAP and MAD therapy alone. However, further studies are needed using 3D data to quantify the dentofacial advantages [27] of combined maxillo-mandibular oral appliance therapy.
This work was presented as a poster presentation at the American Academy of Dental Sleep Medicine in June, 2015. We would like to thank Dr Kim Cress, MD for her contributions to this work.
This study has been performed in accordance with the ethical standards laid down in the 1964 Declaration of Helsinki and its later amendments. All persons gave their informed consent prior to their inclusion in this study, including additional informed consent from all individual participants for whom identifying information is included in this article.
Professor G. Dave Singh DMD PhD DDSc is the former CEO of BioModeling Solutions, Inc., a wholly-owned subsidiary of Vivos BioTechnologies, Inc.