Hypertension (HTN) and obstructive sleep apnea (OSA) are associated with increased risk for adverse cardiovascular events. Comorbid HTN with OSA is often resistant to pharmacologic treatment. Chronotherapy, the timing of treatment based on circadian rhythms, may prove to be a valuable, cost-effective tool for improving blood pressure (BP) control among those with OSA. This Systematic Review appraises the existing literature regarding morning (a.m.) vs. evening (p.m.) administration of antihypertensives among this population.
Ovid, PubMed, Embase, Scopus, and Cochrane were searched for studies examining nighttime administration or nighttime versus daytime administration of antihypertensive medications.
A variety of outcomes were examined. Of studies that examined average daytime and average nighttime BP, all except one found a significant decrease from baseline in both variables regardless of medication used or treatment time. Of four studies that examined BP dipping pattern, two found a significant increase in the percentage of participants with a normal dipping pattern, one reported a non-significant increase, and one reported no increase. One study and the case study report the effect on sleep BP surge secondary to hypoxemia and the natural early morning BP surge. The study found that the β-blocker carvedilol was more effective at lowering the hypoxia-induced sleep BP surge, while the calcium channel blocker nifedipine was more effective in lowering the natural morning BP surge. The case study found that the -blocker doxazosin was effective in lowering both measurements in one patient.
Findings suggest that nighttime versus daytime administration of antihypertensive drugs may have a greater impact on improving blood pressure metrics in adults with coexisting OSA and HTN. The current literature is insufficient to draw robust conclusions regarding the efficacy of chronotherapy in the targeted population. The success observed in some studies supports the examination of this intervention in future research.
Blood pressure, Cardiovascular disease, Chronotherapy, Cost-effectiveness, Etiology, High blood pressure, Hypertension, Obstructive sleep apnea
ACC: American College of Cardiology; AHA: American Heart Association; ACEI: Angiotensin Converting Enzyme Inhibitor; ARB: Angiotensin Receptor Blocker; BB: α-blocker; BP: Blood Pressure; CCB: Calcium Channel Blocker; CV: Cardiovascular; CPAP: Continuous Positive Airway Pressure; HCTZ: Hydrochlorothiazide (thiazide diuretic); HTN: Hypertension; MI: Myocardial Infarction; OSA: Obstructive Sleep Apnea; RAAS: Renin-Angiotensin-Aldosterone System; RH: Resistant Hypertension
Obstructive sleep apnea (OSA) and hypertension (HTN) affect more than 100 million people in the United States (U.S.) and abroad [1-3]. Both conditions are associated with poor health outcomes including end-stage renal failure, stroke, arrhythmias and ischemic heart disease [4,5]. Although both disorders are likely underdiagnosed, patients with one are frequently found to have the other [6,7]. The conditions share many of the same risk factors, including obesity, increased age, alcohol ingestion, and tobacco use [8,9]. They are often comorbid, with an estimated 30% of patients with HTN also having OSA, and about 50% of patients with OSA also having HTN [6,10]. HTN is one of the most important risk factors for death due to cardiovascular events (CV) and stroke. In 2015 the age-adjusted death rate for HTN related conditions was 21.0 deaths per 100,000 people, [2] and HTN was the 13th leading cause of death in 2016 [11]. The incidence of OSA is likely severely underestimated due to its vague clinical presentation (snoring, frequent awakening, and fatigue) [3,12] and diagnostic methods requiring polysomnographic measurements [13].
A growing body of research is beginning to elucidate a complicated bidirectional relationship between the two conditions [14,15]. Additionally, HTN in the setting of OSA is often resistant to pharmacologic treatment due to the augmentative effect of hypoxia occurring in OSA on the impaired extracellular fluid regulation of resistant hypertension (RH) [16,17]. Given the prevalence of these conditions, their known association with risk for cardiovascular (CV) events, and the many barriers to successful treatment, it is necessary to explore novel treatment regimens.
One intriguing avenue of study is determining the best time of day for dosing conventional medications so that they perform to their highest potential. Chronotherapy is the intentional timing of medications based on the administration of the drug in the morning (a.m.) or as the evening dose at bedtime (p.m.) based on knowledge of biologic patterns and 24-h variations in drug metabolism [18]. Previous research has found that sleep-time blood pressure (BP) is a strong indicator of risk for adverse CV-related events, and that administering at least one antihypertensive medication at night can reduce overall CV risk among hypertensive patients [19]. However, there is significantly less evidence available on the use of chronotherapy for hypertensive patients with OSA. The purpose of this systematic review is to synthesize the published evidence related to the efficacy of nighttime versus daytime administration of antihypertensive medications in the ambulatory adult population with OSA and HTN.
OSA is believed to lead to HTN as the result of increased stimulation of the sympathetic nervous system in response to intermittent hypoxemia, further augmenting the activation of pulmonary stretch receptors during apneic periods [14,20-22]. Hypoxemia generates vasoconstriction, oxidative stress, and systemic inflammation that promote the pathological vascular remodeling found in HTN [6,8]. Overstimulation of the renal sympathetic fibers and the activation of the renin-angiotensin-aldosterone system (RAAS) are also among the potential mechanisms by which OSA is conducive to the development of HTN. These result in electrolyte-mediated fluid retention and more collagenous vascular remodeling [22].
Conversely, HTN may also advance the physiologic changes that occur in OSA. This reciprocal relationship is primarily believed to be induced by the actions of aldosterone, as it has been proposed that hyperaldosteronism-related HTN also increases the likelihood and severity of OSA [22]. Several specific mechanisms have been proposed: 1) Interstitial volume accumulation as the result of increased RAAS activity produces pharyngeal edema leading to airway obstruction [14,23] 2) Increased vascular permeability induced by hyperaldosteronism also exacerbate the pharyngeal edema. 3) Central mineralocorticoid receptors interrupt the function of the respiratory center when activated by aldosterone [14]. Supported by these theories, recent clinical trials have demonstrated that renal denervation in patients with OSA and HTN decreased the systolic BP by 22 ± 23 mmHg (P < 0.001) at 3 months post-procedure [24].
A noteworthy characteristic of HTN coexistent with OSA is that it is often resistant to traditional pharmacologic treatment [16,25]. AHA describes RH as HTN that continues to persist despite the use of three classes of antihypertensive medication including a diuretic, or controlled HTN that requires the use of four or more medications [26]. Several studies have associated OSA with RH. A recent meta-analysis found that the odds of having RH were approximately three times greater for patients who had OSA than for patients who did not, (OR, 2.84; 95% CI 1.703 - 3.980) [16]. Additionally, OSA elevates the CV risk by 47% in patients with RH compared to those without the condition (OR, 1.47, 95% CI 1.33 - 1.62, median follow up 3.8 years) [27] and facilitates the concentric ventricular hypertrophy of the cardiac muscle and heart failure [14,28].
The 24-hour variation in BP is well-established in the literature. BP fluctuates in response to the release of several vasoactive substances release (e.g. catecholamines, endothelial nitric oxide synthase, and RAAS components), that also follow a circadian pattern [18]. BP tends to be lowest during sleep, followed by an early morning, pre-awakening surge continuing to rise throughout the morning and decreasing during the afternoon [29]. Many people experience a 10-20% drop in BP during sleep, which is commonly referred to as a "dipping" pattern and is mediated by the autonomic nervous system [22]. This pattern is associated with lower risk of adverse CV events occurring during this period, [29-31] while the natural increase in BP in the early morning (a.m. surge) facilitates pathological mechanisms that cause myocardial infarction (MI) and stroke [29,32].
Deviation from the traditional dipping pattern incurs risk for various types of negative health outcomes. Nocturnal BP that does not fall at least 10% ("non-dipping") is found in approximately 25-35% of hypertensive patients, but can also manifest in persons who are clinically normotensive [33]. Nocturnal BP can sometimes increase from daytime levels, which is referred to as "rising" [29]. Both non-dipping and rising BP patterns may be induced by the presence and severity of OSA, elevating CV and renal risk [29,31,34]. Conversely, "extreme dippers", or those who experience a fall in nocturnal BP greater than 20% of daytime levels, are more likely to have lacunar stroke and silent MI [29].
Chronotherapy is timed-administration of drug therapy founded on the knowledge of the circadian physiologic changes, and can be used to enhance the efficacy and safety of pharmacologic therapies [18,35]. A plethora of research studies performed in the 1980's and 1990's support the use of chronotherapy among hypertensive patients without OSA. Data gathered from these trials supports the theory that taking the full daily dose of at least one antihypertensive medication at bedtime can effectively lower nocturnal BP, thereby reducing the risk of adverse CV events [30,36,37]. Two separate arguments for the use of chronotherapy in this setting relate to the pathophysiology that arises when the conditions are comorbid, and the effect of circadian rhythms on pharmacokinetics.
The circadian fluctuation of the RAAS and the autonomic nervous system are key components which influence BP [12,33]. Normal physiology produces a decrease in sympathetic tone and an increase in vagal tone during nighttime hours, thus lowering the BP during sleep [18,33]. However, the apneic episodes in OSA create a hypoxemic state, inducing sympathetic stimulation, [22] and stimulating RAAS, [8] consequently contributing to the "non-dipping" pattern seen in many patients with OSA [31]. Additional CV risk is incurred when the natural a.m. surge occurs in this setting, especially when superimposed over nocturnal HTN [32].
The absorption, metabolism and the excretion of drugs vary rhythmically and are dependent on the physical characteristics of the chemical compound. A medication's efficacy is altered by the distribution and concentration of plasma proteins, sensitivity of the target receptors, and the drug's plasma binding affinity that takes place when drugs are administrated in a time-dependent manner [38]. For example, the β-blocker propranolol is highly hydrophobic and is well-absorbed in the morning when the gastric pH and intestinal perfusion are physiologically increased. In contrast, hydrophilic drugs such as atenolol have no diurnal variation in absorption [12,39]. A drug's lipophilicity, its protein binding capacity, and its concentration affect the volume distribution. One publication notes that plasma levels of albumin and another carrier protein, α1-glycoprotein, reach the lowest concentration at night [40]. This has implications for drugs with high protein-binding capacity, especially those with a narrow-therapeutic index, as the risk for toxicity may be increased with evening dosing. Conversely, this also suggests that chronotherapy could have the advantage of allowing lower doses with similar therapeutic effects [40,41].
Published evidence regarding drug metabolism indicates that the activity of cytochrome P450 enzymes also follows a circadian pattern, with the actions of different proteins exhibiting different peak metabolism times [41]. The enzymatic activity of hepatocytes is influenced by the hepatic blood flow and drug concentration in the portal vein [42]. Diurnal variations are seen in drug excretion and are related to the release of bile, renal blood flow, glomerular filtration, tubular reabsorption, and urinary pH, although reports vary [18,41,43].
The authors performed a search of PubMed, Cochrane, Scopus, Ovid, and EMBASE using the search terms listed below. The terms were searched in Titles and Abstracts, as well as topics (i.e. MeSH categories in PubMed). Date-related exclusion criteria were not used for this study. As such, no start time limitation was applied and the end time was the day the searches were performed, July 12, 2018 and January 19, 2019. Inclusion and exclusion criteria for the articles used for this review are listed in Table 1. No systematic or literature reviews were published regarding chronotherapy in patients with HTN and OSA at the time of the searches.
Table 1: Inclusion and exclusion criteria. View Table 1
The first and second authors read the abstracts and flagged the relevant studies using a standardized worksheet. The full text was obtained and read for studies with ambiguous relevance. Each author independently read and extracted data from all studies.
The combination of search terms used is as follows: (Chronotherapy OR Chronopharmacology OR "medication timing" OR "medication administration timing" OR "medication scheduling" OR "Evening dosing" OR "Evening versus morning dosing" OR bedtime) AND ("antihypertensive medication" OR "blood pressure drug" OR "Blood pressure medication" OR hypertension) AND ("obstructive sleep apnea" OR "sleep apnoea" OR "sleep disordered breathing" OR OSA).
Blood pressure and OSA diagnostic cutoff points were established according to the to the 2017 guidelines provided by the American College of Cardiology [ACC] and the AHA at 130/80 mmHg for primary HTN [44] and Apnea-Hypopnea Index (AHI) ≥ 5 apneic events/hour for mild OSA, AHI ≥ 15 events/hour for moderate OSA and severe OSA with AHI ≥ 30 events/hour [44] as defined by The American Academy of Sleep medicine (AASM).
Three randomized crossover trials [46-48], two non-randomized trials [49,50], and one case study [51] were included in this systematic review. Final sample sizes ranged from 11 [46] to 78 subjects [45]. One study was performed in the U.S. [46] two in Japan, [46,51] one in Australia, [47] one in Italy [49] and the other in Greece [50]. Information related to race/ethnicity was provided only in Ziegler, et al [48].
In all studies, the researchers assessed antihypertensive therapy administered at night, although the study design and therapy agents used varied. Medications used included the calcium channel blocker (CCB) barnidipine as an add-on therapy, [49] the angiotensin converting enzyme inhibitor (ACEI) perindopril, [47] and the selective α-blocker, doxazosin [51]. Three studies examined multiple medications, including the angiotensin receptor blocker (ARB) valsartan or a valsartan/amlodipine (CCB) combination drug, [50] nifedipine (CCB) and carvedilol, a nonselective β-blocker (BB)/α1-blocker, [46] nebivolol (BB) and hydrochlorothiazide (thiazide diuretic, HCTZ) [48] Subjects who were taking drugs that could alter BP were excluded, [48,50] or those already taking sympatholytics or other antihypertensives at bedtime [46]. Three studies and the case study did not have a morning dosing group, [46,48,49,51] while the researchers in two of the trials compared morning versus evening dosing [47,50]. Additional information about each study is listed in Table 2. Continuous Positive Airway Pressure (CPAP) was used as an adjuvant therapy, [47,50] but these results are not evaluated here as this is not the focus of this systematic review. It is imperative to mention that the chronotherapeutic effect of antihypertension medications was not the primary focus of the Ziegler, et al. [48] study. Nevertheless, the treatment drug was given at bedtime and the findings proved to be relevant to this systematic review.
Table 2: Study characteristics. View Table 2
Several outcome variables are reported across the studies. However, only the most common are compared here. These include the average daytime BP, [47-50] average nighttime BP, [46-50] and nocturnal dipping pattern, [47-50] (Table 2 and Table 3). Kario, et al. [47] and Yoshida, et al. [51] report hypoxia-induced sleep BP surge and early morning average BP, which are important targets of chronotherapy for HTN among patients with OSA. As such, these variables are also examined in this systematic review (Table 2 and Table 3). Ambulatory BP monitoring devices were used to assess the variables listed above. These automatically measure BP every 15-30 minutes for a minimum of 24-hour period. Kario, et al. [46] and Yoshida, et al. [51] used a specialized device with both an automatic set interval setting and a hypoxemia-triggered setting, allowing them to investigate the hypoxemia-induced sleep BP surge. One study used finger photoplethysmography to measure the baroreflex sensitivity by calculating the relationship between the SBP and heart rate of the study participants [48]. Additionally, polysomnography offered pertinent data regarding central BP hemodynamic patterns analyzed using hypoxia-triggered metrics [46] and 24-hour BP averages, [47,48,50] but these metrics are not discussed in this systematic review.
Table 3: Results, selected variables. View Table 3
Serinel, et al. [47], Ziegler, et al. [48], Crippa, et al. [49] and Kasiakogias, et al. [50] assessed mean daytime BP (average daytime BP), (Tables 2, Table 3 and Figure 1). Kasiakogias, et al. [50] found no statistically significant difference between the a.m. and p.m. dosing treatment groups (r = 0.26, P = 0.094 for a.m. dosing and r = 0.39, P = 0.012 for p.m. dosing), while Serinel, et al. [47] found that a.m. administration lowered mean daytime BP statistically more than evening dosing (Table 3 and Figure 1). This difference is unlikely to be of clinical significance, since the difference in wake SBP between groups was 1.8 mmHg (95% CI, 1.1 to 2.5, P < 0.001). Ziegler, et al. [48] and Crippa, et al. [49] did not have a morning treatment group. Crippa, et al. [49] observed a statistically significant decrease in daytime BP when patients took barnidipine at night (the difference in daytime SBP/DBP between groups was 9.3/8.5 mmHg, P < 0.05), while Ziegler, et al. [48] observed a significantly greater decrease in wake BP with p.m. nebivolol (-6.3/-5.3 mmHg) versus HCTZ (-3.7/-1.3 mmHg, P < 0.05) (Table 3 and Figure 1). Of additional interest, Ziegler, et al. [48] also report an increase in heart rate with HCTZ during the wake period (5.1 ± 1.35 bpm) and a decrease with nebivolol (-7.7 ± 1.9 bpm), (Table 3 and Figure 1).
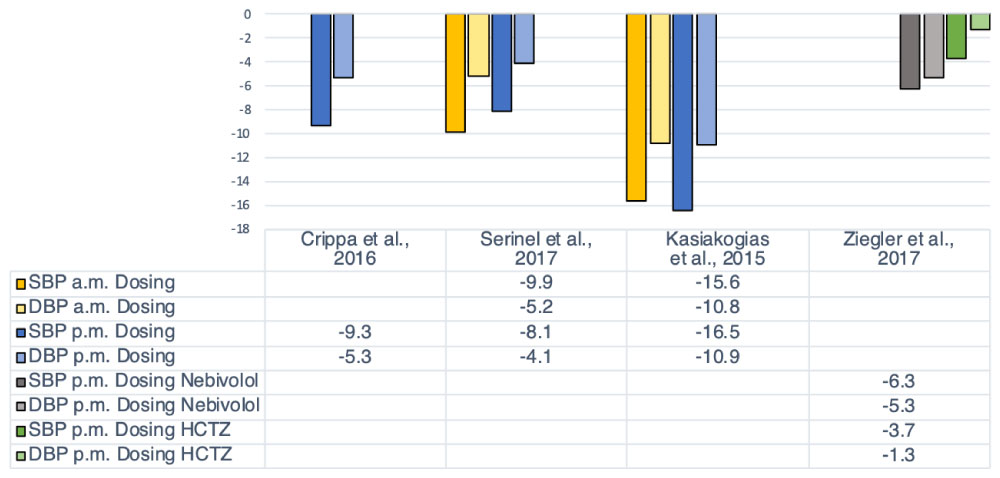 Figure 1: Changes from baseline in average daytime BP between the treatment groups.
All values reported in mmHg. SBP: Systolic Blood Pressure; DBP: Diastolic Blood Pressure; HTN: Hypertension; HCTZ: Hydrochlorothiazide.
Crippa, et al. [49] - p.m. dosing only with Barnidipine 10 mg;
Serinel, et al. [47] - a.m. and p.m. dosing with Perindopril 10 mg;
Kasiakogias, et al. [50] - a.m. and p.m. dosing with Valsartan 160 mg for stage 1 HTN, Amlodipine/Valsartan 5/160 mg, 10/160
mg, or 10/320 mg for stage 2 HTN;
Ziegler, et al. [48] - p.m. dosing only with Nebivolol 5-10 mg, HCTZ 12.5-25 mg.
View Figure 1
Figure 1: Changes from baseline in average daytime BP between the treatment groups.
All values reported in mmHg. SBP: Systolic Blood Pressure; DBP: Diastolic Blood Pressure; HTN: Hypertension; HCTZ: Hydrochlorothiazide.
Crippa, et al. [49] - p.m. dosing only with Barnidipine 10 mg;
Serinel, et al. [47] - a.m. and p.m. dosing with Perindopril 10 mg;
Kasiakogias, et al. [50] - a.m. and p.m. dosing with Valsartan 160 mg for stage 1 HTN, Amlodipine/Valsartan 5/160 mg, 10/160
mg, or 10/320 mg for stage 2 HTN;
Ziegler, et al. [48] - p.m. dosing only with Nebivolol 5-10 mg, HCTZ 12.5-25 mg.
View Figure 1
Authors of the same four studies also examined BP dipping pattern [47-50]. Serinel, et al. [47], Crippa, et al. [49] and Kasiakogias, et al. [50] found that more subjects converted to a dipping pattern with p.m. dosing, (78% conversion rate from 0 dippers at baseline to 32 dippers, P < 0.05 [49] and 24% dippers at baseline increased to 34% dippers with a.m. dosing versus 61% participants with p.m. dosing, P < 0.05, [50] (Table 3). Additionally, in the Greek study it was observed that a.m. dosing did increase the proportion of dippers, but this finding was not significant [50]. The dipping pattern was not altered with the evening administration of nebivolol or HCTZ, [48] (Table 3).
Mean nighttime BP was measured in five of the studies included in the review, [46-50] (Table 3). Again, all authors except Ziegler, et al. [48] reported a significant decrease from baseline, regardless of treatment group (Table 3 and Figure 2). However, for this variable Kasiakogias, et al. [50] observed that p.m. administration lowered mean nighttime BP more than a.m. administration. This difference was statistically significant (BP = 135.5/81.8 mmHg at baseline decreased to 112.6/68.4 mmHg for p.m. dosing; 117/71.3 mmHg for a.m. dosing, overall P < 0.05), (Table 2 and Table 3). On the other hand, Serinel, et al. [47] did not observe a significant difference between a.m. versus p.m. dosing. The authors describe higher nighttime mean BP with p.m. dosing (difference in sleep SBP between groups = 1.1 mmHg, 95% CI -0.3 to 2.5, P = 0.14), (Table 3 and Figure 2) [47]. Similar results on nighttime BP were noted with either nebivolol or HCTZ in Ziegler, et al. [48] but not in Kario, et al. [46] who found a more pronounced decrease in mean nighttime BP with p.m. nifedipine than with p.m. carvedilol (BP = 137.3/86.4 mmHg at baseline decreased to 121.8/8.3 mmHg for the carvedilol versus 112.8/66.6 mmHg for nifedipine, P < 0.01), (Table 3 and Figure 2). They also confirmed that carvedilol was more effective at reducing sleep SBP surge, but the difference between carvedilol and nifedipine was not significant, [46] (Table 3 and Figure 2).
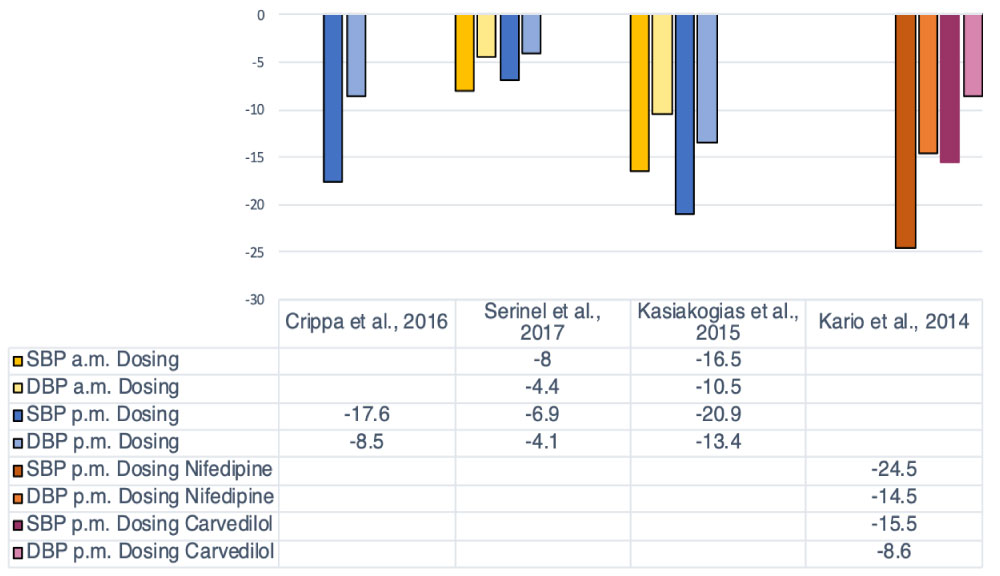 Figure 2: Changes from baseline in average nighttime BP between the treatment groups.
Figure 2: Changes from baseline in average nighttime BP between the treatment groups.
All values reported in mmHg. SBP: Systolic Blood Pressure; DBP: Diastolic Blood Pressure; HTN: Hypertension; HCTZ: Hydrochlorothiazide.
Crippa, et al. [49] - p.m. dosing only with Barnidipine 10 mg;
Serinel, et al. [47] - a.m. and p.m. dosing with Perindopril 10 mg;
Kasiakogias, et al. [50] - a.m. and p.m. dosing with Valsartan 160 mg for stage 1 HTN, Amlodipine/Valsartan 5/160 mg, 10/160
mg, or 10/320 mg for stage 2 HTN;
Kario, et al. [46] - Nifedipine ER 40 mg, Carvedilol 20 mg.
View Figure 2
The purpose of this systematic review was to determine the efficacy of nighttime versus daytime administration of antihypertensive medications among patients with HTN and OSA. Overall, the published evidence is insufficient to support or oppose the use of chronotherapy for the treatment of HTN in this population at this time. There is a considerable amount of variability in study design and drug class examined among the few currently available studies on the topic. Nonetheless, the results presented in these studies suggest that specific chronotherapy modalities may positively affect mean daytime BP, mean nighttime BP, and restore the circadian dipping pattern in this population, which may in turn reduce CV risk.
All antihypertensives used in the included studies produced a statistically significant decrease of daytime BP regardless of dosing time, [47,49,50] except for HCTZ in Ziegler, et al. [48] Kasiakogias, et al. [50] and Serinel, et al. [47] compared morning versus evening dosing of their respective medications, and observed similar results regarding the average daytime BPs between treatment groups. This demonstrates that the nighttime administration of some medications does not provide inferior daytime BP management when compared to daytime dosing of the same medications [47,48]. However, nighttime dosing may not be efficacious with all drug types or preparations, and is likely dependent on the pharmacokinetics of the chosen medication. It is important to consider that daytime BP is only one metric, and that drugs which do not produce a desired effect on daytime BP may produce more favorable outcomes on other measures associated with CV risk.
Nighttime BP was also substantially reduced across four of the five studies which examined this variable, regardless of medication used or the administration time, [46-49] (Figure 2). The greatest net decrease in average nighttime BP from the baseline was achieved with CCBs in a combination with valsartan (ARB) [50] or solo [46,49]. The differences demonstrated in two studies [46,50] involving CCBs were both statistically and clinically significant, as mean nighttime BP approached the 110/65 mmHg threshold for nocturnal HTN described in the 2017 ACC/AHA guidelines [44]. The final group of researchers examining a CCB [49] observed a statistically significant decrease in mean nighttime BP, but BP readings after the intervention were still higher than the ACC/AHA threshold (Table 3 and Figure 2).
It is important to note that only one group of researchers compared a.m. versus p.m. administration of a combination drug which included a CCB, and that the dose was adjusted based on the stage of HTN of each participant in this study [50]. The others used a CCB alone, but had only a p.m. treatment group and did not personalize the dose [46, 49]. All CCBs used in these studies were of the dihydropyridine class. As dihydropyridine CCBs are well known for their vasodilating effects, it is likely that the efficacy of these medications is due to the blockage of hypoxia-induced vasoconstriction. Interestingly, when compared to p.m. dosing of carvedilol (BB), a nighttime dose of nifedipine (CCB) produced a markedly greater decrease in mean nighttime BP (and mean daytime BP), [46] (Figure 1 and Figure 2). This suggests that vasodilation may be a more desirable effect than broad adrenergic blockade in this population. More research into the exact mechanism of CCBs in this setting is warranted.
The mechanism of action for BBs among this population is better studied. Nebivolol was used in one study [48]. This drug has the potential to block vasoconstriction due to norepinephrine secreted in response to apneic episodes in OSA. Additionally, it augments the nighttime dipping pattern of BP by supplementing the stimulation of nitric oxide release and causing peripheral vasodilation [48,52]. The beneficial effects of nebivolol on reducing vascular resistance facilitates the melioration of the endothelial dysfunction and vascular β-2 adrenergic receptors stimulation without affecting vasocontraction [53].
Serinel, et al. [47] report a statistically significant decrease in sleep BP with both treatment groups but observed a slightly higher nighttime mean BP with p.m. administration of perindopril (an ACEI) versus a.m. administration of the same drug, although this difference was not statistically significant (Table 3). These authors reemphasized the importance of pharmacokinetics and pharmacodynamics in drug efficacy during a 24-hour period. The minimal effect of perindopril in this study could be due to the long half-life of the drug metabolite pendoprilat (20-30 hours), although the authors report that other studies have shown chronotherapeutic effect of ACEIs with long half-lives on nighttime SBP in other populations demonstrated by Morgan [53]. Other pharmacokinetic factors may explain the reduced efficacy of perindopril in the Serinel, et al. study, such as the potential circadian variation in pharmacokinetics of the drug, or the possibility that these processes are different in patients with OSA [47].
Importantly, the use of diuretics at night will have a minimum effect on lowering nighttime BP because of the natural increase in secretion of atrial natriuretic peptide that occurs in response to hypoxia induced by OSA [48]. This effect was confirmed by the analysis of baroreflex sensitivity which was decreased when the subjects used HCTZ compared to those in the placebo and nebivolol groups in one study [46]. This reduction indicates that the natural feedback mechanism by which the body balances BP and heart rate was somewhat impaired with the use of HCTZ in this population, although the clinical significance of this magnitude change was not described in the study.
Four of the studies [47-50] examined in this review reported the effect on the prevalence of a dipping pattern among the participants. In agreement with the evidence presented by Hermida, et al. [54] bedtime administration of amlodipine/valsartan [50] was more efficient in converting participants to a dipping pattern than a.m. dosing. Bedtime dosing of barnidipine was also effective, converting 88% of non-dippers/risers to dippers [49] (Table 3). It is reported that p.m. dosing of ACEIs was generally effective in restoring this variable among hypertensive patients without OSA [54]. Considering that RAAS is overstimulated in OSA, [22] it follows that there is a physiologic basis for the use of this drug class for chronotherapy among this population. Several authors report that BBs had a greater effect on daytime BP, which promoted a non-dipping pattern among patients with HTN but not OSA [22]. Ziegler et al. [48] report that nebivolol had a somewhat similar effect on BP over the entire 24-hour period, and hypothesize that sympathetic stimulation could be increased during the day as well as during sleep in patients with OSA. This supports the therapeutic value of BB among this population, but care should be taken to ensure that the peak effect occurs during the nighttime hours as to avoid promotion of a non-dipping pattern. Additionally, the inferior effect of HCTZ in treating high BP in patients with OSA is attributed to the impaired baroreflexes activity juxtaposed to the increase sympathetic stimulation [48]. Additional trials will be necessary to determine the efficacy of these drug classes in chronotherapy among hypertensive patients with OSA.
It is hypothesized that the high BP produced in response to hypoxic episodes caused by OSA is part of the pathologic process behind sleep-onset CV events that are common among hypertensive patients with OSA [20,34, 46] Beta-blockers such as carvedilol are effective in lowering a.m. BP surge in hypertensive patients without OSA [54]. Conversely, Kario, et al. [46] found that nifedipine was more effective than the carvedilol in lowering the early morning surge. One potential explanation for this discrepancy is that Kario, et al. [46] administered sustained-release nifedipine and immediate-release carvedilol. If the medications are taken in the evening, the a.m. surge would occur near the end of the 12-hr duration of action for carvedilol, and in the middle of the 24 hr duration of action for the sustained-release nifedipine [56]. Future trials should explore the effect of sustained release β-blockers on BP surges in this population.
Yoshida, et al. [49] describe the use of a different sympatholytic agent in the treatment of a young man with a history of HTN, OSA, three sleep-onset strokes, and a sleep SBP surge that continued to be elevated after successful lifestyle modification and weight loss [51]. The patient was prescribed doxazosin 4 mg/day at bedtime, effectively lowering his sleep SBP surge and early morning average BP. The authors describe that at the time of publication the patient had not experienced another stroke during the two years he had maintained this treatment. Importantly, this is the only report that describes the effect of a chronotherapeutic intervention on direct measures of morbidity. Superior 24-hour BP control among hypertensive patients without OSA with p.m. dosing of doxazosin was also documented, [54,57,58] with more beneficial activity in the early morning hours [57].
To the authors' knowledge, this is the first systematic review to examine the use of chronotherapy for treatment of HTN among patients with comorbid OSA. The detailed examination of the theory behind the use of chronotherapy presented in this review intends to highlight the advantages this approach could provide for CV risk reduction in this challenging population. The use of ambulatory BP metrics allowed for an understanding of the effect of this intervention during times associated with increased CV risk. Ambulatory BP monitoring is a valuable tool that should be more widely utilized when predicting CV risk. The majority of the studies looked at the same variables (dipping pattern, daytime BP, nighttime BP), which allowed the authors to contrast which medications may have the greatest potential for this application. At the same time, the variety of study designs and medications used limits the comparability of the results. The different medications, dosing patterns, comparison groups, and variables used mean that the strength of the evidence is limited to single-study findings, and reproducibility cannot be considered. Nonetheless, this breadth of methodologies does allow for a more comprehensive exploration of this emerging field. Furthermore, the chronotherapy evaluation of Ziegler, et al. [48] on other variables expose valuable data reflecting a detailed picture of BP regulation as a method to improve the endothelial function and further to decrease the CV risk.
There are several important limitations to this systematic review. Firstly, there was an extremely small amount of literature for this type of intervention among individuals with both HTN and OSA. What studies have been published all had a fairly small sample size presenting little or no information of the baseline demographic characteristics of the study participants, thus reducing the generalizability of the results. Zeigler, et al. [48] did not report the BP measurements prior to initiating the trial therapy, thus it cannot be concluded if the addition of the trial therapy lowered BP to target levels. Additionally, there was a wide range in mean BMI between studies (25.8 ± 8 kg/m2 44 to 35 ± 8.6 kg/m2), [46] which is an important confounder. It is likely that the pathological effects of a higher BMI induce disturbance of the baroreflexes and impairs vascular dilatation further aggravating the OSA manifestation. This reflects that obese patients are at higher risk for hypoxemic events during sleep compared to those who have a normal BMI [59].
Another limitation is that only two studies [48,50] excluded subjects who were already taking antihypertensive medications, meaning that the observed effect in only these studies may be attributed to the regimen prescribed by the research physicians and not to a possible synergistic effect induced by augmenting the participant's original regimens. This is an important differentiation since both modalities may prove to be efficacious but hold different implications for prescribing clinicians.
Although Serinel, et al. [47] and Kasiakogias, et al. [50] investigated the use of CPAP in addition to chronotherapy, this was not addressed in the current review. CPAP is the gold standard treatment for OSA and has been associated with a small but statistically significant reduction in BP among hypertensive patients who use it consistently [60]. It is possible that CPAP could have an additive or synergistic effect when used with chronotherapy, but neither of the studies in this review observed an association between medication dosing time and decrease in BP related to CPAP. For this reason, it is not believed that CPAP has had a confounding effect on the results presented here. Additionally, suboptimal adherence to CPAP is multifactorial [61] and further augments the severity of symptoms. The authors argue that chronotherapy is a valuable tool as it may have better compliance among patients who are intolerant of CPAP and should therefore be investigated individually in order to determine its efficacy among this population.
There are also several potential sources of bias in the studies themselves that could influence the results of this systematic review. The methods used to identify and enroll subjects are not described by any of the researchers, leaving the question of potential selection bias unanswered. Loss of follow-up was pronounced in the study performed by Zeigler, et al. [48]. Ten out of 41 (24%) participants did not finish the study, thus diminishing the power and presenting an opportunity for attrition bias [48]. Most of the studies had a cross-over design, [44-46,48] and none had a separate control group [46-51] Kasiakogias, et al. [50] and Crippa, et al. [47] did not randomize participants into treatment groups. Kario, et al. [46] and Kasiakogias, et al. [50] performed an open-label study without blinding the participants. Most of the studies disclose the blinding of the researchers, which is important in helping reduce potential measurement and/or confirmation bias [46-48,50].
Based on current published evidence, it is not possible to draw robust conclusions about treatment recommendations until more studies have been completed. Meanwhile, clinicians are encouraged to follow current best practice recommendations for the treatment of patients with HTN and OSA. Chronotherapy may be considered on an individual basis, especially if ambulatory BP monitoring is an option at initiation of treatment to ensure adequacy and safety of the regimen. It is imperative to emphasize the relevance of the add-on CPAP therapy to amplify the chronotherapeutic effect of antihypertensive treatment in this target population.
Although the wide range of medications and variables used in these studies precludes the development of guidelines for using chronotherapy in clinical practice, it does provide a fairly broad preliminary picture of the potential use of chronotherapy among hypertensive patients with OSA. As such, the authors are able to make recommendations regarding the direction of future research. First, no large studies of any design type have been conducted with this particular population. The publication of studies with large sample sizes will help improve the power to detect any benefit of chronotherapy and, depending on the sampling techniques used, could also help provide a sample that is representative of the larger population of people with HTN and OSA. Second, studies utilizing a prospective design with long-term follow-up will be critical in determining if there is truly a reduction in CV risk with the use of chronotherapy. The current evidence only addresses intermediate variables that have been correlated with increased risk for CV events, but which cannot directly determine the effect on morbidity or mortality.
Future studies should use what is known about the circadian variation in pharmacokinetics of various drugs and drug classes, and the pathophysiology of HTN in the setting of OSA to decide which medications should be tested. The published evidence indicates that HTN with comorbid OSA is affected by increased nocturnal sympathetic tone and alteration of RAAS. This suggests a pathophysiologic basis for the chronotherapeutic use of sympatholytics, ACEIs, and ARBs, but it appears that the pharmacokinetics and pharmacodynamics of each medication are particularly important in determining their time-dependent efficacy. CCBs may also be efficacious in this application, but a better understanding of the pathology of HTN/OSA and of the chronopharmacokinetics would likely help determine which particular agents to investigate.
Additionally, more trials are warranted to investigate the potential adverse effects of antihypertensive medications administrated at night. Those with extreme dipping patterns have been shown to have a small but increased risk of total CV events compared to those whose nocturnal dipping pattern was within the normal range [62]. The effect of dose adjustments on the dipping pattern must be evaluated to avoid lacunar stokes, renal ischemia, or MI in patients treated with aggressive therapies. It is suggested that ambulatory blood pressure readings be considered when appraising the risk of CV morbidity and mortality.
Accumulating evidence regarding chronotherapy illustrates that p.m. administration of antihypertensive medications can be effective in regulating BP parameters associated with CV morbidity and mortality among hypertensive patients without OSA. Recent studies suggest that nighttime versus daytime administration of BP lowering drugs have a greater impact on improving ambulatory BP metrics in adults with coexisting comorbidities such as OSA and HTN. However due to heterogenic characteristics of the study designs and treatments included in this systematic review it is difficult to make robust conclusions about the effect of chronotherapy in the targeted population. This is a promising field of study for patients with HTN and OSA as HTN in this setting is often resistant to pharmacologic treatment. Chronotherapy is a cost-effective, simple intervention that if proven safe and efficacious could help improve outcomes for a particularly at-risk population.
We would like to thank to Dr. Rebecca Tsusaki, Dr. Rebecca Casarez, and Dr. Lisa Boss for helping with this paper and for encouraging and supporting to pursue our careers as nurse practitioners and researchers.
None.
None.