To choose an appropriate protocol for treatment of Wilms tumor in conditions of limited resources.
We analyzed outcomes, advantages and disadvantages in application of SIOP 2001 and NWTS 5 protocols in our hospital. Patients with nephroblastoma admitted to National Children's Hospital in Hanoi, Vietnam, had been treated according to the NWTS 5 protocol from 2000-2005, and from 2008-2013 according to the SIOP 2001 protocol.
33 patients, stages I-III treated with NWTS 5 had estimated 5 year EFS 90.1% and OS 96.7% (mean follow up time 30.4 months); 58 patients, stages I-V, treated with SIOP 2001 had estimated 5 year EFS 71.5% and OS 80.9% (mean follow up time 27 months). Patients stage I-III treated with SIOP 2001 protocol had estimated 5 year EFS 77.5% and OS 84.1%. The complication and side effects were acceptable except in one patient treated with SIOP 2001 protocol who died as a complication of treatment. For patients treated with SIOP 2001, imaging diagnosis was confirmed by pathological examination in 78.3% cases.
Patients treated with NWTS 5 and SIOP 2001 protocols had good outcomes in a limited resources situation. Imaging and pathological diagnoses are challenging for medical staff in developing countries. Treatment approach should be based on medical staff competence and patient's condition. We recommend immediate operation for operable tumors and preoperative chemotherapy for inoperable cases.
Nephroblastoma or Wilms tumor (WT) is one of the most common solid malignant tumors in children. With a huge progress in diagnosis and treatment, as a result of NWTS/COG (National Wilms Tumor Study/Children Oncology Group) and SIOP (International Society of Paediatric Oncology) protocols, the outcome in developed countries is excellent, with 85-90% 5 year EFS [1,2]. In developing countries, these protocols were also applied but usually under difficult circumstances due to lack of resources, training and funding [3]. There is a need to identify which protocol is most appropriate for institutions with limited resources [4,5]. There were studies carried out in developing countries to define outcome's influencing factors [6] and apply adapted protocol for developing countries based on SIOP protocol in order to estimate its capacity in limited resources condition [7]. In National Children's Hospital in Hanoi, we applied both NWTS5 and SIOP2001protocols for treatment of patients with nephroblastoma. In this study we analyzed advantages and disadvantages of these protocols under our conditions.
Our first study using the NWTS5 protocol was carried out from July 2000 to August 2005. All patients had immediate nephrectomy, and staging and histological classification were done according to the NWTS5 criteria. We exclude all patients who abandoned treatment immediately after nephrectomy from our study because in these cases their families had made a decision before operation that if the diagnosis proved to be of a malignant tumor or in advanced stages, the child would have no further treatment.
Post-operative treatment (chemotherapy and radiotherapy) was given as follows:
We had no patient requiring treatment with regimen I (Table 1).
Table 1: Post-operative treatment for patients treated with NWTS 5 protocol. View Table 1
Patients under 12 months of age received chemotherapy reduced to 1/2 of calculated doses on the basis of body weight.
From July 2008 to June 2013, we conducted a second study with the SIOP 2001 protocol. All patients with clinical diagnosis of retroperitoneal or abdominal mass had abdominal ultrasound and CT. On tumor board meeting, we discuss and decide the tumor is WT or not. The decision based on clinical findings, patient's age, imaging characters of tumor. If the imaging diagnosis was nephroblastoma, patients were given preoperative chemotherapy according to stages assigned by imaging findings: stage I-III, IV and V. If the imaging diagnosis was not nephroblastoma or patients were under 6 months of age or there was tumor rupture, patients had immediate nephrectomy. We exclude the patients who received preoperative chemotherapy but died or abandoned treatment before operation because we could not confirm the diagnosis of WT. Post-operative treatment was based on tumor's stage and histological classification as follows (Table 2 and Table 3):
Table 2: Post-operative treatment for patients treated with SIOP 2001 protocol that had preoperative chemotherapy. View Table 2
Table 3: Post-operative treatment for patients treated with SIOP 2001 protocol that had immediate operation. View Table 3
We did not randomize patients in stages II and III with intermediate risk tumors for administration of Doxorubicin as per SIOP2001 protocol (the authors of protocol designed it to test their question if added Doxorubicin will improve patient's outcome or not). We used Doxorubicin only for patients in stage III. After the SIOP 2011 Congress, we applied SIOP recommendation that these patients do not need Doxorubicin for their treatment [8].
The cases with stage IV with immediate operation received 6 weeks of chemotherapy as preoperative chemotherapy for stage IV and further treatment depended on re-evaluation of metastatic response after this course.
Patients with body weight less than 12 kg received chemotherapy with dose reduction to 2/3 of calculated doses.
For both studies, all patients had abdominal ultrasound and CT to evaluate the tumor, and chest x-ray for examination of lung metastasis. If there was metastasis, chest CT would be done later.
In our 1st study which followed the NWTS5 protocol there were 33 patients (6 in stage I, 12 stage II and 15 stage III with 1 case of focal anaplasia in stage III) treated with NWTS 5 protocol. Follow up time ranged from 10 to 61 months with mean of 30.4 months. EFS estimated at 5 years by Kaplan-Meier was 90.1% and OS 96.7% (Figure 1). One patient with stage II and 2 patients with stage III WT relapsed, and one of stage III patients died. Two patients (6.06%) had episodes of neutropenic fever (grade III). Other side effects include stomatitis grade I (most of the cases) and II and skin infection grade I.
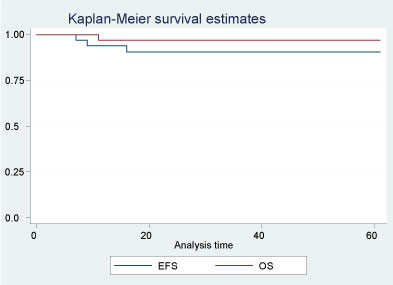 Figure 1: Treatment outcome of patients treated with NWTS 5 protocol. View Figure 1
Figure 1: Treatment outcome of patients treated with NWTS 5 protocol. View Figure 1
The results of the 2nd study which followed the SIOP 2001 protocol are summarized in Figure 2. There were 60 patients with nephroblastoma including 47 who had preoperative chemotherapy and 13 who had upfront nephrectomy. Out of the 47 patients who received preoperative chemotherapy, 43 had localized tumors (stage I-III), one had a metastatic tumor (stage IV with lung metastasis), and three had bilateral tumors (stage V). After preoperative chemotherapy and surgery, the stage distribution was as follows: 18 (38.3%) stage I, 19 (40.4%) stage II, and 10 (21.3%) stage III; the patient with stage IV had local stage III, and patients with stage V had local stage II.
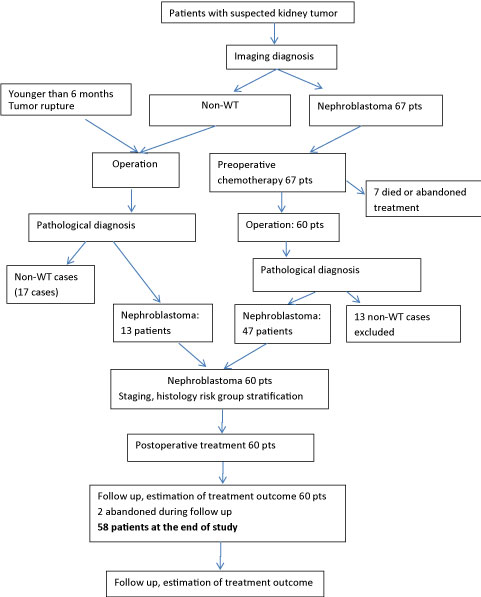 Figure 2: The algorithm of study with SIOP 2001 protocol is as following. View Figure 2
Figure 2: The algorithm of study with SIOP 2001 protocol is as following. View Figure 2
For the 13 cases of nephroblastoma requiring immediate surgery, 10 were due to incorrect imaging diagnosis of non Wilms tumors, two had tumor rupture and one was 4 months old at presentation. Following immediate surgery, three patients (23.1%) were stage I, five (38.5%) stage II, four (30.7%) stage III, and one (7.7%) stage IV with liver metastasis. Stage IV patient at presentation had immediate nephrectomy due to tumor rupture. Out of the 13 non-WTs who had received preoperative chemotherapy, there were 10 clear cell sarcomas of kidney, two neuroblastomas and one renal cell carcinoma. WTs which had received preoperative chemotherapy were stratified as low risk WTs in 2.1%, intermediate risk in 85.1% and high risk in 12.8% cases.
Two of the 60 patients abandoned follow up examination after end of treatment in EFS condition (both had preoperative chemotherapy and were staged as stage I and II, intermediate risk). Follow up time for the remaining 58 patients ranged from 2-57 months with a mean of 27 months. At the end of study, there were 44 patients in EFS, one death related to treatment, and 13 patients relapsed of whom eight died. Kaplan-Meier estimated EFS rate for all stages together at 5 years was 71.5% and OS 80.9% (Figure 3). Patients in stages I-III (53 patients: 41 had preoperative chemotherapy and 12 had immediate nephrectomy) had estimated 5 year EFS was 77.5% and OS 84.1%. Most severe side effects included one treatment related death, and seven patients (11.7%) had neutropenic fever (grade III). The other side effects included hematologic toxicity, skin infection, stomatitis, and fever in absence of infection with most of the cases in grade I and some in grade II and did not required special treatment and self-resolved after cease of chemotherapy.
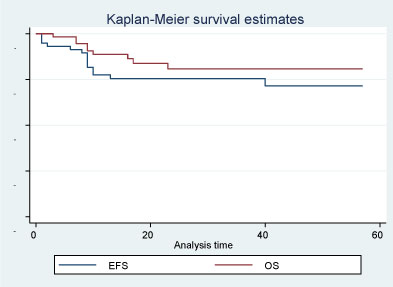 Figure 3: Treatment outcome of 58 patients treated with SIOP 2001 protocol. View Figure 3
Figure 3: Treatment outcome of 58 patients treated with SIOP 2001 protocol. View Figure 3
There are two main approaches to treatment of Wilms tumor in the world: one based on immediate operation (NWTS/COG) and the other based on preoperative chemotherapy (SIOP). Both are followed by chemo- and radiotherapy depending on tumor's staging and histological classification. They both have excellent results in developed world but they still have advantages and disadvantages, especially in countries with limited resources.
During the time of our study with NWTS5 protocol (patients enrolled to study from July 2000 to October 2004 and follow up to August 2005), the families had to pay for treatment. Because of social and financial reasons, a number of patients in stage II had not enrolled in our study. Patients with stages III, IV and V refused treatment also because their parents considered that the disease was incurable. The abandonment rate was around 50% after nephrectomy but no more abandonment when chemotherapy had commenced. However, since 2005, Health Insurance covered treatment expense for our patients. As a result, there was no abandonment of treatment and follow up when treatment became free for families. We used this protocol up to June 2008, (before we started to use SIOP 2001) but we don't have data of follow up of all patients treated with this protocol from August 2005 to June 2008.
Patients who were treated with SIOP 2001 preoperative chemotherapy had higher proportion of stage I and lower proportion of stage II and III in comparison to the ones who had immediate surgery, i.e. preoperative chemotherapy had a desired "down-staging" effect. We had lower proportion of stage I patients (38.3%) than SIOP data (52-62%) but the number with stage III (21.3%) was similar [9]. This means that the number of patients with stage III, who require radiotherapy, was reduced. The histological risk groups distribution was similar to SIOP data [10].
We did not compare the patients' outcome of our two studies because they had been carried out at different times, with different cohort of patients and different overall circumstances. Our outcome of treatment is much lower than that from developed countries. Patients treated with NWTS 5 protocol had high EFS and OS rates, but less than half the patients in stage III and no patients in stage IV or V that were enrolled and had full treatment.
In SIOP 2001 protocol, if a patient is older than 6 months and imaging diagnosis is that the tumor originates from kidney, the patient will have preoperative chemotherapy without confirmatory histological diagnosis. That is why the imaging diagnosis was not considered incorrect as long as the tumor was of a renal origin [11]. However, we think that approach is suitable for populations where nephroblastoma comprises more than 90% of renal tumors. In our study, only the cases with the imaging diagnosis of WT received preoperative chemotherapy, and not to those where the imaging features were not diagnostic of WT. We chose that approach because previous reports from our Department of Pathological Anatomy in 1990s showed that patients with WT contributed only around 70-75% of all renal tumors. During the time that we used NWTS5 protocol, WT contributed 76.5% of all renal tumors diagnosed in our hospital. So if we had given chemotherapy to all patients with renal tumors, a high proportion of patients with non-WT would have had received non-effective preoperative chemotherapy. This approach was confirmed by data from Department of Pathological Anatomy: nephroblastoma represented only 68% of all renal tumors admitted to our hospital during the period when we followed the SIOP 2001 protocol (unpublished data). The data from European countries showed that 93% of renal tumors were confirmed to be nephroblastoma [12]. However, in our study only 78.3% (47/60) of tumors diagnosed pre-operatively as nephroblastoma were confirmed as such on histological examination. The discrepancy may be due to less experience of our imaging specialists compared with SIOP institutions or due to a genuine higher incidence of rare renal tumors in our Vietnamese population. But, significant discrepancies between imaging and pathological diagnosis have been reported in the UK and German studies too, where 12% and 7.8% of cases, respectively, with typical imaging findings of WT proved to be non-WTs [13,14]. A study in the USA which included children of all ages showed that the proportion of WT among renal tumors was only 73.9% (68/92) and CT studies had diagnostic accuracy of 82% [15]. Smets, et al. have stated that imaging studies cannot distinguish between clear cell sarcoma of kidney and nephroblastoma [16]; and rhabdoid tumors of kidney and renal cell carcinoma also have imaging findings similar to nephroblastoma [16], despite the fact that nephroblastoma have some typical imaging findings [17]. In our series of 60 cases with the imaging diagnosis of WT, 10 (16.6%) were clear cell sarcomas and one renal cell carcinoma. We experienced a higher incidence of 18% of clear cell sarcomas and rhabdoid tumors during the time of the SIOP 2001 protocol, compared with 3.6% (113/3134 patients) [12] reported in Europe during 1988-1997 period. In two studies from Ho Chi Minh City, clear cell sarcoma of kidney comprised 21.2% and nephroblastoma 66.7% of all renal tumors (articles published in 2005 and 2007, only in Vietnamese). So all data from Vietnam are showing that in renal tumours, nephroblastoma is less frequent than in SIOP and NWTS data, and other tumors (clear cell sarcoma of kidney and rhabdoid tumor of kidney), which have similar ultrasound and CT finding as nephroblastoma, are more frequent. This is a likely reason for discrepancy between imaging and pathological diagnoses in our study and it makes their reconciling impossible. In China, a neighborhood country with us, the rate of clear cell sarcoma and rhabdoid tumor of kidney reported is much higher than in Europe and similar with our data, comprise in total 14.7% of all renal tumors of childhood [18]. In Japan, also a country in the Far East, WT contributed only 75.1% and the proportion of clear cell sarcoma and rhabdoid tumor of kidney were 16.2% [19]. So it is clear that the incidence of WT are not the same in all over the world and in some places the distribution of renal tumors a different than in Europe and United States.
From our study, it is clear that if we used preoperative chemotherapy based on imaging diagnosis, we could not avoid giving it to a significant proportion of patients with non-Wilms tumors, especially clear cell sarcoma or rhabdoid tumors, which need to be treated with more intensive treatment than nephroblastoma. In our study with SIOP 2001 protocol, the proportion of patients had immediate nephrectomy was high (21.7%), these patients had no benefit from "down-staging" effect of preoperative chemotherapy and also make the interpretation and analysis of treatment outcome of all patients complicated.
Histological diagnosis of Wilms tumor treated with preoperative chemotherapy is a very big challenge for pathologists in both developed and developing countries since chemotherapy significantly alters the histological feature of Wilms tumor, resulting in different histological patterns and distribution of subtypes from those treated with immediate surgery [20]. Staging is also more difficult because it is harder to evaluate the extent of tumor [20].
In order to avoid wrong diagnosis leading to inappropriate treatment, the SIOP have established a system of rapid central pathology review [21]. In SIOP 2011 Congress, the SIOP reviewers reported that there were 25% of discrepancies between institutional pathologists and central pathology review including 9.5% with diagnostic and 15.5% with staging differences. However, the vast majority of cases had rapid central pathology review so the patients received appropriate treatment. We think our pathologists are at the same risk of making mistakes without central pathology review. From the beginning of our study and up to present, our colleagues have been helped by Bengt Sandstedt (Stockholm, Sweden) for training and reviewing of cases but we cannot have rapid central pathology review. We can send the images of tumors by internet but in that case the specialists can only see limited and selected images. This would be even more difficult for other hospitals in our country, which have less facilities, resources and international collaborations than our hospital.
From our point of view, the staging and histological classification according to NWTS criteria are easier for our pathologists than SIOP criteria. It is due to the facts that tumor's structure is not altered by preoperative chemotherapy and NWTS criteria for histological subtyping are simplier, since tumors are classified as non-anaplastic (favorable) or anaplastic (unfavorable) only. According to NWTS 5 protocol, non-anaplastic WTs stage I or II have the same treatment - regimen EE-4A, and patients with stage III and IV – regimen DD4A. So miss-staging between stage I and II, III and IV was not treatment relevant. In our study with NWTS 5 protocol, we think there was no error in staging related to treatment regimen when our pathologists had no help from specialists from developed countries. All patients with stage III tumors had clear evidence of this stage which means that there was no over-staging. Patients with stage I and II did very well so we think there was no under-staging and consequent under-treatment. All patients with stages I and II have favorable histology WTs so there was no problem if there was miss-staging between stage I and II. The cases with anaplastic feature are not common in SIOP and NWTS/COG data as well as in our hospital. We had 3 cases with focal anaplasia (only 1 had full treatment)/65 WTs in our study with NWTS 5 protocol and 4 cases with focal anaplasia, 1 case with diffuse anaplasia/60 WTs in the study with SIOP 2001 protocol.
In the developing world, treatment of children with WTs according to both SIOP and NWTS/COG protocols have their advantages and disadvantages. In our opinion, institutions with limited resources will have difficulties with preoperative chemotherapy based on imaging finding and histological diagnosis when using SIOP protocol. On the other hand, they will have difficulties in avoiding intra-operative rupture when operating on big tumors treated according to the NWTS/COG protocol. Nowadays SIOP and COG are focusing on reducing the doses of chemotherapy and radiotherapy for most of patients while maintaining the achieved excellent outcome, and improvement of outcome of patients with poor prognosis. For that, more risk factors are being using or will be used for risk-stratification and risk-directed therapy [22,23]. Institutions in developing countries cannot apply all of those risk factors in practice. The recognition of loss of heterozygosity 1p and 16q as a prognostic factor which have a role in assignment of risk groups is an additional technological challenge for developing countries for application of new NWTS/COG protocols. We think the current NWTS/COG approach is quite flexible: if the tumor is inoperable, patient will have preoperative chemotherapy and the tumor is classified as stage III if there is no sign of distant metastases to make it stage IV. We agree with the opinion of Julio G. D'Angio [24] "What would the writer recommend if a child in a family were to develop a large flank mass? If near a major pediatric oncology center, early surgery. If not, preoperative chemotherapy with dactinomycin and vincristine". For developing countries it can be interpreted as: if the tumor is operable, immediate nephrectomy is recommended; if it is inoperable, then preoperative chemotherapy is the first treatment. Based on our experience, we recommend using NWTS 5 protocol for the cases with immediate nephrectomy and SIOP 2001 for the cases with preoperative chemotherapy. Localized tumors in stage I and II are operable and will have immediate nephrectomy, whereas tumors in stage IV and V will have preoperative chemotherapy. Tumors in stage III may be operable or inoperable, depending on discussion at tumor board meeting with attendance of doctors from all related specialties. The cases with tumor's rupture will have immediate nephrectomy as an urgent operation. By this approach, we think that we can combine most of advantages of these protocols in our condition. The inoperable cases will have down staging effect from preoperative chemotherapy, which means their post-operative treatment will be milder. This is the advantage of SIOP approach that tumors in advanced stages at presentation may have post-operative treatment that not requires Doxorubicin and radiotherapy. The operable cases should have a majority proportion: in our study with SIOP 2001 protocol the rate of WTs in stage IV and V was similar to data in developed countries. So, in the majority cases we have no problem with miss-matching between imaging and pathological diagnoses and we can avoid giving preoperative chemotherapy designed for WTs for a significant proportion of non-WTs. The pathological diagnosis will be easier for our pathologists and treatment relevant mistakes will be fewer in condition that we have no rapid central pathology review.
The outcome for patients treated with NWTS/COG and SIOP protocols have been similar for decades. We think that medical staffs in developing countries have to weigh their competence (skill and equipment) and patient's condition before deciding which approach is most suitable under their condition. We think both approaches can be used flexibly. We recommend using NWTS 5 protocol for the operable tumors and SIOP 2001 for inoperable cases. We think that way of treatment approach is flexible and appropriate for our hospital as well as for developing countries.
I am very grateful to Professor Henry Ekert; Hematology and Oncology Department, The Royal Children Hospital, Melbourne, Australia; not only for his help in writing this article but also for his help and teaching during my training course in his department and in my work for many years. I would like to express my sincere thanks to Professor Gordan Vujanic; Department of Cellular Pathology, University Hospital of Wales, Cardiff, UK; for his help in writing this article.
The author declares that there is no conflict of interest regarding the publication of this article.