With the growing prevalence of type 2 diabetes in the United States, the use of insulin has increased along with the incidence of needle-stick injuries [1]. The Institute for Safe Medication Practices (ISMP) published guidelines in 2017 on how to optimize safe subcutaneous insulin use in adults. One of the recurring themes of these guidelines is education, education, education. This is essential for self-management by patients with diabetes and also for the safety and effectiveness of the care they receive from healthcare professionals [2].
Insulin is a drug that can save a patient's life, but it can also cause life-threatening injury or even death when not used appropriately. In April of 2016, the Centers for Medicare and Medicaid Services (CMS) published evidence-based guidelines that categorized hypo- and hyperglycemia as preventable conditions. When these guidelines are followed, CMS subsequently denies payment for the treatment of diabetic ketoacidosis, hypoglycemic coma, and other serious conditions that result from poor glycemic control [3].
Errors that have been associated with insulin therapy include, but are not limited to, administration of the wrong insulin product, under- and overdosing, dose omissions, incorrect use of insulin delivery devices, wrong route (intramuscular versus subcutaneous) and improper patient monitoring. Many of these errors result in serious hypo- or hyperglycemia [2].
Since insulin is considered a high-alert medication, the ISMP guidelines recommend that certified diabetes educators (CDEs) or others trained in diabetes education be available to support and educate patients with diabetes. ISMP encourages healthcare organizations to define the roles and responsibilities of staff for educating patients to ensure completion of this potentially life-saving task [2].
Several studies have noted that insulin was implicated in 33% of medical errors that caused death within 48 hours of the error with patient safety incidents, of which 24% resulted in patient harm. For older Americans, insulin was one of four medications that caused 67% of all emergency admissions due to adverse drug events [2]. In an article published by Dr. Pandya, et al. in Geriatric Nursing, long-term care facility recommendations include the assessment of current practice, education of staff, control of diabetes, implementation of new practices, and resident self-management education during discharge planning [4]. Educating healthcare professionals, patients and caregivers about safe subcutaneous insulin injection can minimize and/or prevent adverse consequences [2].
Dr. Pandya, et al. also make recommendations for insulin delivery in post-acute and long-term care facilities that stress using the shortest needle possible to minimize risk of intramuscular injection, and also obviate the need for pinching up the skin when administering the injection. In fact, when using short needles, the pinch-up technique is not recommended, since it would increase the risk of intradermal injection. Moreover, use of short needles reduces the risk of a needle-stick injury and/or an accidental intramuscular injection that could cause unwanted hypoglycemia. The exception to this would be a cachectic individual, for whom pinching up the skin is necessary to avoid an intramuscular injection. In this situation, since the area of pinchable skin is limited and the injection area is small, care must be taken to avoid subcutaneous induration. Figure 1, derived from findings of the Hirsch, et al. study [5], illustrates that the risk of an IM injection of insulin decreases as the length of the needle used decreases. In the United States, the 5 mm needle is the shortest currently available in safety injection pen devices, and the 4 mm needle is currently only available in conventional products.
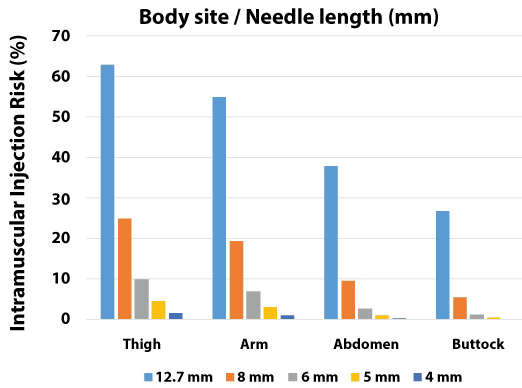 Figure 1: IM Injection risk versus needle length. This figure shows the marked reduction in risk of intramuscular (IM) injection with decreasing needle length at all common injection sites. This risk is clearly minimized by use of the 5 mm needle (shortest currently available in safety injection pen devices commonly used in healthcare settings) and the 4 mm needle (currently only available in conventional products). Shorter needles are particularly relevant for long-term care residents, approximately 20% of whom have been reported to have a BMI < 20 kg/m2 and are therefore at a greater risk for intramuscular injection [5].
View Figure 1
Figure 1: IM Injection risk versus needle length. This figure shows the marked reduction in risk of intramuscular (IM) injection with decreasing needle length at all common injection sites. This risk is clearly minimized by use of the 5 mm needle (shortest currently available in safety injection pen devices commonly used in healthcare settings) and the 4 mm needle (currently only available in conventional products). Shorter needles are particularly relevant for long-term care residents, approximately 20% of whom have been reported to have a BMI < 20 kg/m2 and are therefore at a greater risk for intramuscular injection [5].
View Figure 1
The Occupational Safety and Health Administration (OSHA) recommends the use of safety-engineered devices for skin injection that include syringe needles, pen needles, and lancets. There are safer options commercially available for pen needles that incorporate safety engineered designs for both front and back ends. Employers using pen needles for delivery of insulin or other medications to patients or residents should evaluate these devices in accordance with the OSHA Blood-borne Pathogens Standard [1].
It is also important to consider the increased risk of infections and blood-borne illnesses that is seen with the improper use or reuse of needles and syringes used for blood glucose testing and/or the administration of insulin. Lancets, blood-glucose testing devices, needles and syringes, as well as pen needles may transmit secondary illnesses including hepatitis B and C viruses. The decision to move to safer devices for all insulin delivery must consider potential patient and employee exposure to, and avoidance of, potential illness or infection from multiple pathogens [1].
Patient and direct caregiver education regarding safe subcutaneous insulin use and diabetes self-care is a crucial step in the overall management of diabetes across the healthcare landscape. Whenever a patient with diabetes using insulin is transitioned from one healthcare setting to another, post-discharge education is a key factor in preventing harm. All practitioners caring for patients with diabetes who are prescribed insulin should have easy access to complete patient information and medical records to provide care with the safest devices possible, minimizing the risk of unwanted needle-stick injuries [2]. All frontline healthcare professionals, caregivers and patients should be involved in the evaluation process of all safe insulin delivery devices for each patient with diabetes.
Dr. Manzi has been a licensed pharmacist since 1990 and a Board Certified Geriatric Pharmacist since 1998. She is currently an independent consultant pharmacist, and a previous Board Member and recognized Fellow of the American Society of Consultant Pharmacists (ASCP), USA.
Francis M. Siri, PhD of Paradigm LTC, assisted in the consolidation and editing of this article.