Although oxaliplatin is one of the main anticancer agents used against colorectal cancer, data on the pharmacokinetics of oxaliplatin in patients on hemodialysis are limited. In this case study, we evaluated the pharmacokinetics and safety of oxaliplatin in a patient on hemodialysis treated for colon cancer.
A 68-year-old Japanese woman receiving chronic hemodialysis was treated for metastatic colon cancer with modified FOLFOX6 plus bevacizumab every 2 weeks. Initial oxaliplatin dose was reduced to 42.5 mg/m2, which was 50% of the standard dose. 4-hour hemodialysis was begun 1.5 hours after the end of oxaliplatin infusion. Serial plasma concentrations of total and free platinum were measured before and until the first 48 hours after oxaliplatin infusion. Adverse events were evaluated using Common Terminology Criteria for Adverse Events version 4.0.
The pharmacokinetics of free platinum showed a bimodal pattern. Maximum concentrations were 0.73, 0.80, and 1.03 μg/mL after a dose of 42.5, 42.5, and 50 mg/m2 oxaliplatin, respectively. The area under the curve in the first cycle was 14.2 μg*h/mL. No clinically meaningful accumulation of free platinum was seen. No new safety signals were identified, and no Grade 2-4 adverse events were observed.
These pharmacokinetics data for oxaliplatin in a hemodialysis patient were comparable with those in patients with normal renal function. Modified FOLFOX6 plus bevacizumab every 2 weeks could be safely given in a hemodialytic colon cancer patient with a reduction in oxaliplatin dose to 50 mg/m2, if hemodialysis was performed 1.5 hours after the end of oxaliplatin infusion.
Oxaliplatin, Hemodialysis, Pharmacokinetics, Colorectal cancer, Modified FOLFOX6 plus bevacizumab
The number of patients with end-stage renal disease is increasing rapidly worldwide. These patients require dialysis treatment or renal transplantation for survival [1]. In Japan, hemodialysis patients have been increasing yearly [2], and the survival rate of patients with end-stage renal disease has shown a gradual improvement. The risk of cancer is increased in patients on hemodialysis, and the need for chemotherapy in hemodialysis patients is accordingly increasing [3-5].
Worldwide, colorectal cancer was the third-most common malignancy in men (746,000 cases; 10.0% of total cancer) and the second-most common in women (614,000 cases; 9.2% of total cancer cases) in 2012 [6]. Standard treatments for primary chemotherapy now include the addition of a molecular-targeted agent to oxaliplatin- or irinotecan-based chemotherapy, and mean survival time in patients with recurrent or metastatic colorectal cancer has improved to over 30 months [7,8]. Oxaliplatin is eliminated mainly by the kidneys, and its pharmacokinetics are reported to change in patients with renal dysfunction [9]. Although one report noted that a dose reduction is not necessary if creatinine clearance is more than 20 mL/min [10], it is not clear whether oxaliplatin can be administered in patients with severe renal dysfunction or hemodialysis. Furthermore, while some reports have described the administration of oxaliplatin to hemodialysis patients [11-13], dosing standards for this population have also not been determined.
Here, we evaluated the pharmacokinetics and safety of oxaliplatin administration in a hemodialysis patient.
A 68-year-old Japanese woman who had been maintained on hemodialysis because of chronic nephritis since 2011 was diagnosed with recurrent colon adenocarcinoma in 2015. She was treated with modified FOLFOX6 plus bevacizumab after irinotecan-based chemotherapy. Initial oxaliplatin dose was 42.5 mg/m2, or 50% of standard dose for modified FOLFOX6 (oxaliplatin 85 mg/m2 IV over 2 hours plus l-leucovorin 200 mg/m2 IV over 2 hours, followed by 5-fluorouracil 400 mg/m2 IV bolus, followed by 5-fluorouracil 2,400 mg/m2 over 46 hours continuous infusion; repeat every 2 weeks). After safety was confirmed, the dose of oxaliplatin was increased to 50 mg/m2. 4-hour dialysis, with a dialysate (Kindaly AF-3), was performed at 1.5 hours after the end of oxaliplatin infusion in accordance with a previous study [11]. Blood flow rate was 220 mL/min, bicarbonate-buffered dialysate flow rate was 500 mL/min, and dialysis water removal was 1.7 L. Low molecular weight heparin was infused for anticoagulation. Blood samples for pharmacokinetic evaluation were collected in tubes containing sodium heparin before oxaliplatin infusion; 1 and 2 (end of infusion) hours after the start of oxaliplatin infusion; and 15, 30 min, 1, 1.5 (start of hemodialysis), 3.5, 5.5 (end of hemodialysis), 7.5, 22, and 46 hours after the end of infusion. The samples were centrifuged at 1700 g at 4 ℃ for 10 min, and a portion of the plasma was stored in a deep freezer at -70 ℃ until assay of total platinum. The remaining plasma was ultra-filtrated using an ultrafiltration tube (Amicon® filter with a molecular cut-off of 30,000 Da) at 4 ℃ and stored at -70 ℃ until assay of free platinum. Total and free platinum concentrations were analyzed by inductively coupled plasma mass spectrometry (ICP‑MS) using an Agilent 7700 × ICP-MS (Agilent Technologies, Inc., Santa Clara, CA, USA) [14], and the area under the curve from 0 to 48 hours (AUC0-48) after oxaliplatin infusion was calculated using the linear trapezoidal method. Free platinum concentration of oxaliplatin in our patient was compared with that in historical patients with normal renal function treated with 85 mg/m2 oxaliplatin (Japanese package insert). Adverse events were monitored at least bi-weekly throughout the treatment and evaluated using Common Terminology Criteria for Adverse Events version 4.0.
The pharmacokinetics of free platinum showed a bimodal pattern (Figure 1). Free platinum concentration reached a peak at the end of infusion and decreased immediately in the next 30 minutes. After a slight increase caused by free platinum moving from tissue to blood, it decreased from the start to the completion of hemodialysis, then increased up to 24 hours and maintained a plateau thereafter.
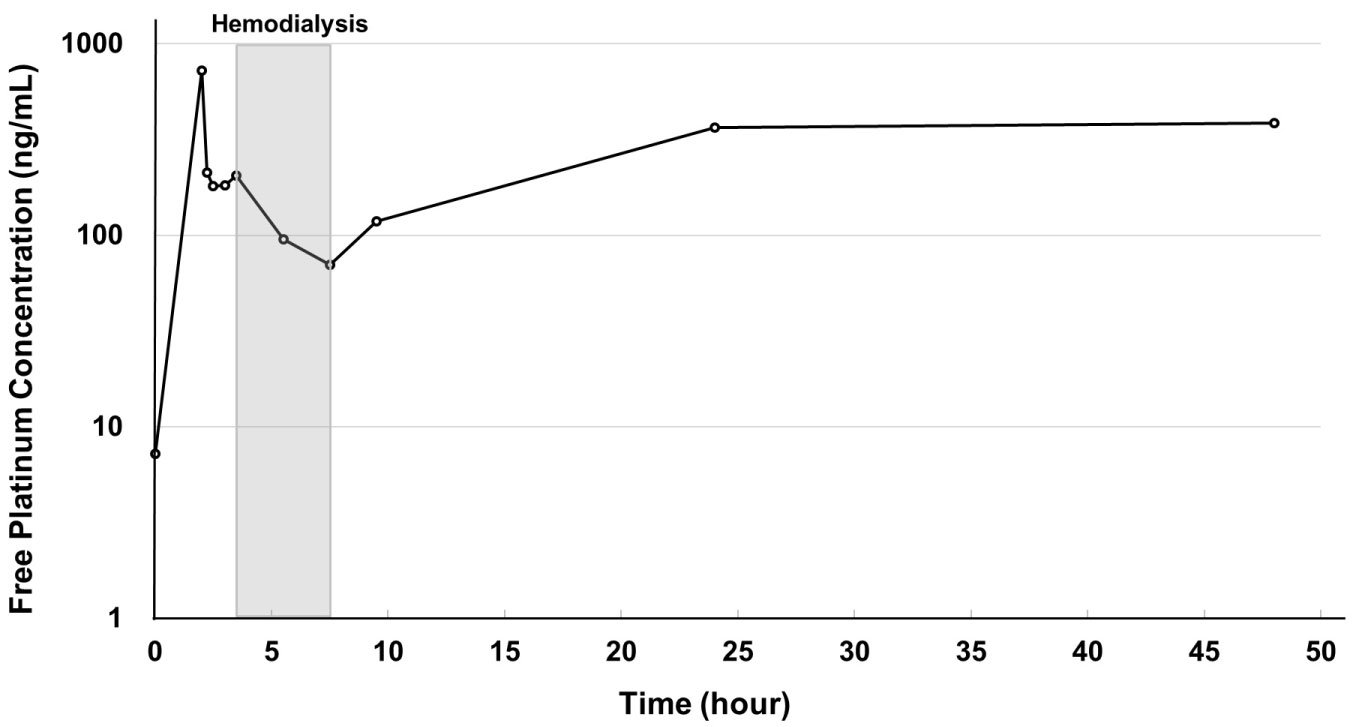 Figure 1: Time curve of plasma free platinum concentration.
Figure 1: Time curve of plasma free platinum concentration.
Free platinum concentration reached a peak at the end of infusion and decreased immediately in the next 30 minutes. After a slight increase caused by free platinum moving from tissue to blood, it decreased from the start to the completion of hemodialysis, then increased up to 24 hours and maintained a plateau thereafter. View Figure 1
The pharmacokinetics of total platinum also showed a bimodal pattern (Figure 2). Total platinum concentration reached a peak at 15 minutes after the end of oxaliplatin infusion and decreased immediately in the next 15 minutes. After maintaining a plateau for one hour, it decreased from the start of hemodialysis to 2 hours after the completion of hemodialysis, then increased up to 24 hours and decreased thereafter.
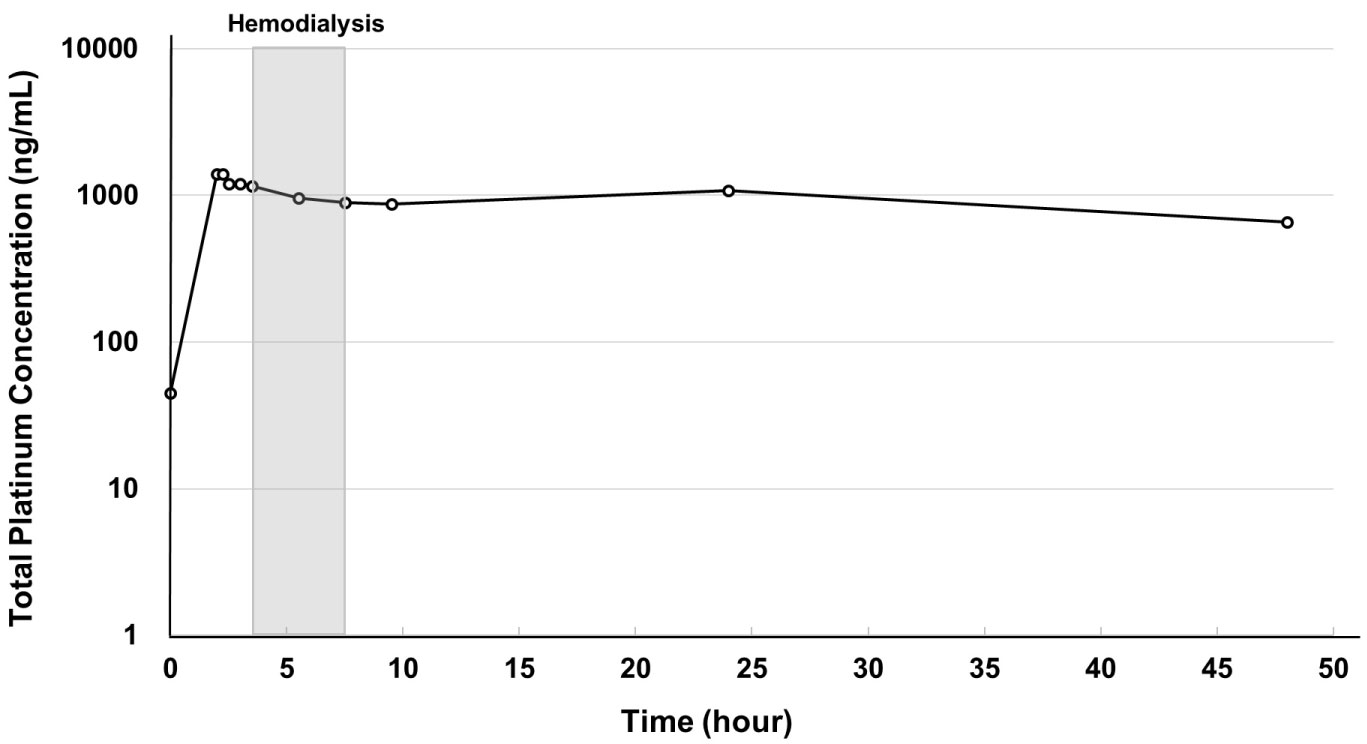 Figure 2: Time curve of plasma free platinum concentration.
Figure 2: Time curve of plasma free platinum concentration.
Free platinum concentration reached a peak at the end of infusion and decreased immediately in the next 30 minutes. After a slight increase caused by free platinum moving from tissue to blood, it decreased from the start to the completion of hemodialysis, then increased up to 24 hours and maintained a plateau thereafter. View Figure 2
Results for the pharmacokinetic parameters of initial concentration (C0), maximum concentration (Cmax) and AUC0-48 of plasma total and free platinum are shown in Table 1. Although C0 slightly increased with each successive cycle, none of these increases was clinically significant. Compared with normal renal function, Cmax was slightly lower on administration at 42.5 mg/m2, but slightly higher at 50 mg/m2. Free platinum AUC was 14.2 μg*h/mL at a dose of 42.5 mg/m2 in the first cycle, or about 1.3 times the value obtained from historical patients with normal renal function treated with 85 mg/m2 oxaliplatin. AUC in the second and third cycles could not be calculated because the free platinum concentration after 48 hours could not be measured.
Table 1: Pharmacokinetic parameters. View Table 1
With regard to safety, grade 1 adverse events included nausea and anorexia during the first cycle; nausea, anorexia, and mucositis during the second; and nausea, anorexia, mucositis, diarrhea, and peripheral sensory neuropathy during the third. No grade 2-4 adverse events were observed. Regarding efficacy, a CT scan performed after five cycles of modified FOLFOX6 plus bevacizumab was evaluated as progressive disease by RECIST ver1.1.
Modified FOLFOX6 plus bevacizumab every 2 weeks was safely administered to a hemodialysis patient with colon cancer by reducing oxaliplatin dose to 50 mg/m2 and conducting hemodialysis 1.5 hours after the end of oxaliplatin infusion. AUC0-48 of free platinum showed a slightly high value with a bimodal pattern, while Cmax showed comparable results to those from patients with normal renal function (Table 1). As for toxicity, only grade 1 adverse event was observed, and no grade 2-4 or new adverse events were observed.
Oxaliplatin changes into three kinds of active form (dichloro-, monochloro-, and diaquo-diaminocyclohexane platin) capable of interaction when entering the blood. Pharmacokinetics shows three phases, namely α (distribution phase), β (elimination phase), γ (redistribution phase). Free platinum concentration rapidly decreases in α-phase, gradually decreases in β-phase, and decreases quite gently in γ-phase [15,16]. However, because most free platinum after oxaliplatin infusion is inactive low molecular weight platinum-amino acid conjugates (glutamine, L-methionine, and L-cysteine) capable of renal elimination [9,16], free platinum in β - and γ - phases is mostly the inactive form.
Consistent with previous reports [11,12,16], plasma free platinum concentration in our patient showed a bimodal pattern. With normal renal function, the second peak caused by free platinum moving from tissue to blood does not appear due to prompt elimination of free platinum by the kidney. In contrast, patients on hemodialysis are unable to eliminate platinum from the kidney, resulting in a second peak which affects the value of AUC. However, since the free platinum at the second peak, corresponding to the γ-phase, is mostly an inactive form, it is considered that it does not affect efficacy or toxicity [9]. Giacchetti, et al. [17] reported that the toxicity and efficacy of oxaliplatin were not related to the AUC of free platinum, but that hematological toxicity was related to free platinum Cmax. Even in our patient, although AUC was slightly high despite a comparable Cmax versus normal renal function, no obvious increase in adverse events was seen compared with previous studies [8,18]. Given the increase in AUC caused by the second peak, some authors have suggested that administration interval should be extended in consideration of toxicity [12,13,19]. On the contrary, we do not consider that extension is necessary because the second and third peaks of free platinum largely consist of inactive forms. Indeed, our study dose was administered safely, albeit that we did not confirm the relationship between free platinum Cmax and toxicity. The possibility that oxaliplatin doses should be further increased until the maximum tolerable Cmax is reached may be worth considering.
The timing of hemodialysis requires an interval to allow sufficient distribution to tissue after oxaliplatin infusion in order for the drug to elicit its effect. If hemodialysis is performed immediately after the end of oxaliplatin infusion, the increase in Cmax can be suppressed, but oxaliplatin is removed before it is sufficiently distributed. Conversely, when dialysis is started 1.5 hours after the end of infusion, oxaliplatin is thought to be well distributed to the tissues. In our patient, free platinum Cmax with a reduced oxaliplatin dose was equivalent to that in patients with normal renal function when hemodialysis was performed 1.5 hours after the end of oxaliplatin infusion (Table 1).
This study has some limitations. First, the data were derived from a single patient. Additional examination in a large number of patients is required to evaluate the safety and efficacy of this treatment. Second, the optimal dose could not be confirmed. Although our study dose could be given safely, determining the optimal dose requires a dose escalation study, as is commonly performed in early phase trials.
In conclusion, pharmacokinetics data for oxaliplatin in this hemodialysis patient were comparable to those in patients with normal renal function. This modified FOLFOX6 plus bevacizumab regimen every 2 weeks could be safely given in a hemodialysis patient with colon cancer by reducing oxaliplatin dose to 50 mg/m2, provided hemodialysis was performed 1.5 hours after the end of oxaliplatin infusion. Allowing that further studies are necessary to confirm optimal dose and efficacy, we propose that the administration strategy used in our patient is one option that can be safely administered to patients receiving hemodialysis.
No funding was received.
All data generated or analyzed during this study are included in this published article.
This prospective study was approved by the Kobe University Hospital Ethics Committee (No.1481) and conducted in accordance with the Declaration of Helsinki. The patient provided written informed consent before study entry.
Not applicable.
Naomi Kiyota has received a research grant from Chugai Pharmaceutical Co., Ltd. outside the submitted work. Hironobu Minami has received research grants from Chugai Pharmaceutical Co., Ltd., Yakult Honsha Co., Ltd., and Kyowa Hakko Kirin Co., Ltd. and has received personal fees from Chugai Pharmaceutical Co., Ltd., and Kyowa Hakko Kirin Co., Ltd. outside the submitted work. The other authors declare that they have no conflict of interest for this study.