Introduction: Monitoring and diagnosis of benign or premalignant breast lesions as it progresses to a malignant condition is very necessary for its ability to predict or curb the progression of a breast lesion to the malignant condition and Immunohistochemistry has been proven to be a useful tool in achieving this as immunohistochemical markers are corresponding and specific in nature.
Objectives: The primary objective of this case-control study is to study the expression of KI67, E-cadherin, and P53 in normal breast tissues, fibroadenomas, and Invasive Ductal adenocarcinoma and to determine the degree of expression of these biomarkers.
Materials and methods: A total of 65 formalin-fixed, paraffin-embedded tissue blocks including 15 normal breast tissues, 25 benign fibroadenoma of the breast and 25 malignant invasive ductal adenocarcinoma were retrieved from the Pathological Archives of the Obafemi Awolowo University Teaching Hospital Complex Ile-Ife (OAUTHC). Immunohistochemical analysis was carried out on the samples. Immunohistochemistry (IHC) was performed according to the streptavidin-biotin immunoperoxidase technique (Dako-cytomation). Multiple slides were evaluated. Immunohistochemical (IHC) profile of the tumor was assessed by subjecting slides from each case and a representative biomarker to an immunostain.
Results: Nuclear Ki-67 staining was expressed; normal cases showed a positivity rate of 20%, fibroadenoma showed a positivity rate of 36%, invasive ductal adenocarcinoma had a positivity rate of 100%. Membrane staining of E-cadherin was expressed; normal cases expressed a positivity rate of 100%, fibroadenoma expressed a positivity rate of 100%, Invasive ductal adenocarcinoma had a positivity rate of 40%. Nuclear P53 staining; normal cases showed a positivity rate of 46%, fibroadenoma showed a positivity rate of 84%, invasive ductal adenocarcinoma had a positivity rate of 100%. There was an upregulation of Ki-67 and P53 and downward regulation of E-cadherin in progression to the malignant condition.
Conclusion: The degree of expression of Ki-67, E-cadherin, and P53 in Normal, Fibroadenoma and Invasive ductal carcinoma have confirmed the ability of immunohistochemistry and tumor markers to predict the progression of breast cancer hence, more than one tumor biomarker must be employed for accurate diagnosis of benign and malignant breast lesions.
Breast cancer, Fibroadenoma, Ki-67, E-cadherin, P53
Breast cancer has persisted as an overall general wellbeing situation and is as of now, the most well-known cancer in the globe with a significant effect on populace wellbeing as it is the most well-known intrusive harm and the second most continuous reason for death from cancer in ladies [1]. Breast cancer is a diversified group of tumors with variable arrangement, behavior, response to therapy, and molecular profiles [2]. Fibroadenoma is the most well-known benign breast tumor in youthful women. They present most regularly between ages 20 and 50 with top frequency revealed at 20 to 24 years. They represent 68% of all breast masses and a huge level of breast biopsies [3]. Malignancy in fibroadenomas is uncommon, with announced rates going from 0.002% to 0.125%. Carcinoma in situ inside a fibroadenoma is normally found unexpectedly when tumors are extracted. Invasive ductal adenocarcinomas are usually preceded by a fibroadenoma phase [4]. Fibroadenomas emerge from the lobular stroma of the terminal duct lobular unit. They are an expansion of epithelial and stromal segments, likely identified with estrogen. After some time, whenever left in situ, they go through hyalinization of the stromal segment with relapse of the epithelial segment. They are chemical responsive masses and may go through cyclic changes in size and manifestations with menses [3]. Invasive ductal adenocarcinoma (IDC) begins in the milk ducts of the breast and metastases' to the wall of the duct, invading the fatty tissue of the breast and possibly other regions of the body. Invasive ductal carcinoma is the most common type of breast cancer, accounting for 80% of breast cancer diagnoses [5]. Invasive ductal adenocarcinomas have cancer cells that invade and spread outside of the normal breast ducts, growing into the surrounding breast stromal tissue. Invasive ductal adenocarcinomas have the potential to unfurl to various other sites of the body, such as the lymph nodes or other organs and to form metastases [6].
Immunohistochemistry (IHC) is utilized to describe intracellular proteins or different cell surfaces in all tissues. There is a developing rundown of accessible items (antibodies) or antigen recovery procedures, which all add to the more extensive utility of Immunohistochemistry for tackling indicative issues or for deciding guess and reaction to treatment in breast pathology [7]. Antigen KI67 (KI67) MH167 a marker of cell proliferation is a non-histone nuclear protein expressed throughout the active phase of the cell cycle, except G0 and early G1 [8]. Ki67 index is a significant biomarker of breast cancer as higher ki67 is associated with higher tumor grade [9]. In normal tissues, the declaration of Ki67 is for the most part lower but significantly upregulated in cancerous tissues. The expression levels increments continuously with the improvement of the infection [10]. A critical component of malignant growths is their uncontrolled capacity to multiply [11]. Ki-67 staining can be utilized as a kind of perspective file for the visualization and customized therapy of breast cancer patients [6]. E-cadherin (E-CD) is a significant cell grip atom in typical epithelial cells serving functions including cell modification, tissue morphogenesis, the foundation of cell extremity and support of tissue engineering [12]. Amassing proof recommends that high-grade intrusive ductal carcinomas are portrayed by genomic unsteadiness with the deficiency of an expanding number of cancer silencer qualities during the carcinogenesis interaction that adds to their forceful conduct [13]. Decreased/loss of E-cadherin articulation is perceived as a feature of the fundamental sub-atomic occasions driving loss of cell-cell attachment and hence working with malignant growth intrusion and metastasis and its down regulation during the time spent epithelial-mesenchymal change in cancer movement [14].
P53 is a nuclear transcription factor with a pro-apoptotic function that assumes a basic part in forestalling human malignancy development [15]. In light of an assortment of stress signals, p53 becomes initiated and prompts cell cycle capture and additionally apoptosis. Nearly one-third of breast tumors possess mutations in the p53 gene [16]. Although P53 mutations are common in invasive breast carcinomas, few have been identified in intraductal breast carcinomas [17]. P53 gene may be correlated as a diagnostic marker of breast cancer as the loss of its normal function can lead toward the breast cancer progression and it's over expression associated with breast cancer [18].
This study was a retrospective analysis evaluating the expression of KI67 (Table 1b), E-cadherin KI67 (Table S2b), and P53 KI67 (Table S3b) in normal breast tissues, fibroadenomas, and Invasive Ductal adenocarcinoma and determining the degree of expression of these biomarkers.
Immunohistochemistry study was performed at Obafemi Awolowo University Teaching Hospital Complex Ile-Ife (OAUTHC). Study Analysis was performed at Afe babalola University. The study period was from February to April 2021.
Samples were collected only from confirmed cases of breast cancer.
A total of 65 formalin-fixed, paraffin-embedded tissue blocks including 15 normal breast tissues, 25 benign fibroadenoma of the breast and 25 malignant invasive ductal adenocarcinoma were retrieved from the Pathological Archives of the Obafemi Awolowo University Teaching Hospital Complex Ile-Ife (OAUTHC).
Results were presented in figures and tables; micrographs were also used where necessary. Ki67, E-cadherin and P53 staining were evaluated using a regular light microscope at mg x40 and x10 (Figure S1, Figure S2, Figure S3 and Figure S4).
Expression of KI-67, E-cadherin and P53 were determined through a semi-quantitative method. The immunoreactivity of these markers was set on by assessing the staining intensity and percentage of stained cells per field. The staining intensities were graded as mild, moderate and severe. Percentages of positive cells were graded as follows:
0.1%-10% are stained = negative (-), grade 0.
10.1%-39% are stained = positive (+), grade 1.
40.0%-79% are stained = positive (++), grade 2.
80.0%-100% are stained = positive (+++), grade 3 [19].
Factual examination of acquired outcomes would be done utilizing the Graph Pad Prism programming program.
The procedures followed were found to have compiled substantially to the stipulated standards for the conduct of human specimen research. Ethics Committee Approval was obtained from the Institutional Ethics Committee with reference number FBMS/EC/12012021 dated February 22nd 2021. Waiver of informed patient consent was obtained from the ethical committee due to the retrospective nature of the research study.
Table 1a showing the semi-quantitative expression of KI67 in normal, fibroadenoma and invasive ductal adenocarcinoma. Normal cases showed a positivity rate of 20% where expression on the slide was weak, fibroadenoma showed a positivity rate of 36% where sixteen (16) cases showed inadequate staining, nine (9) cases showed weak staining, invasive ductal adenocarcinoma had a positivity rate of 100% as all of the cells showed significant expression.
Table 1a: Expression of KI67 in normal breast tissue, fibroadenoma and invasive ductal adenocarcinoma. View Table 1a
A Table 2a showing the semi-quantitative expression of E-Cadherin in normal, fibroadenoma and invasive ductal adenocarcinoma Normal cases showed a positivity rate of 100% where five (5) slides showed moderate positivity and ten (10) showed strong positivity, fibroadenoma showed a positivity rate of 100% as they all showed significant expression; where ten (10) cases showed mild staining, seven (7) cases showed moderate staining and eight (8) cases showed severe staining. Invasive ductal adenocarcinoma had a positivity rate of 32% where 17 slides showed inadequate expression, 8 showed weak expression.
Table 2a: Expression of E-Cadherin in normal breast tissue, fibroadenoma and invasive ductal adenocarcinoma. View Table 2a
A Table 3a showing the semi-quantitative expression of P53 in normal, fibroadenoma and invasive ductal adenocarcinoma Normal cases showed a positivity rate of 26.7% where the expression was mildly significant and mostly negative, fibroadenoma showed a positivity rate of 76% where six (6) cases showed negative staining, nine (9) cases showed mild staining, and ten (10) showed moderate staining, invasive ductal adenocarcinoma had a positivity rate of 100% where five (5) showed weak reactions, twelve (12) showed moderate expression and eight (8) showed severe expression.
Table 3a: Expression of P53 in normal breast tissue, fibroadenoma and invasive ductal adenocarcinoma. View Table 3a
Table 4 shows the mean percentage reactivity of KI67, E-Cadherin and P53 in normal, fibroadenoma and invasive ductal adenocarcinoma. It shows an increase in reactivity in KI67 and P53 from normal to invasive ductal adenocarcinoma and a decrease in E-Cadherin (Graph 1).
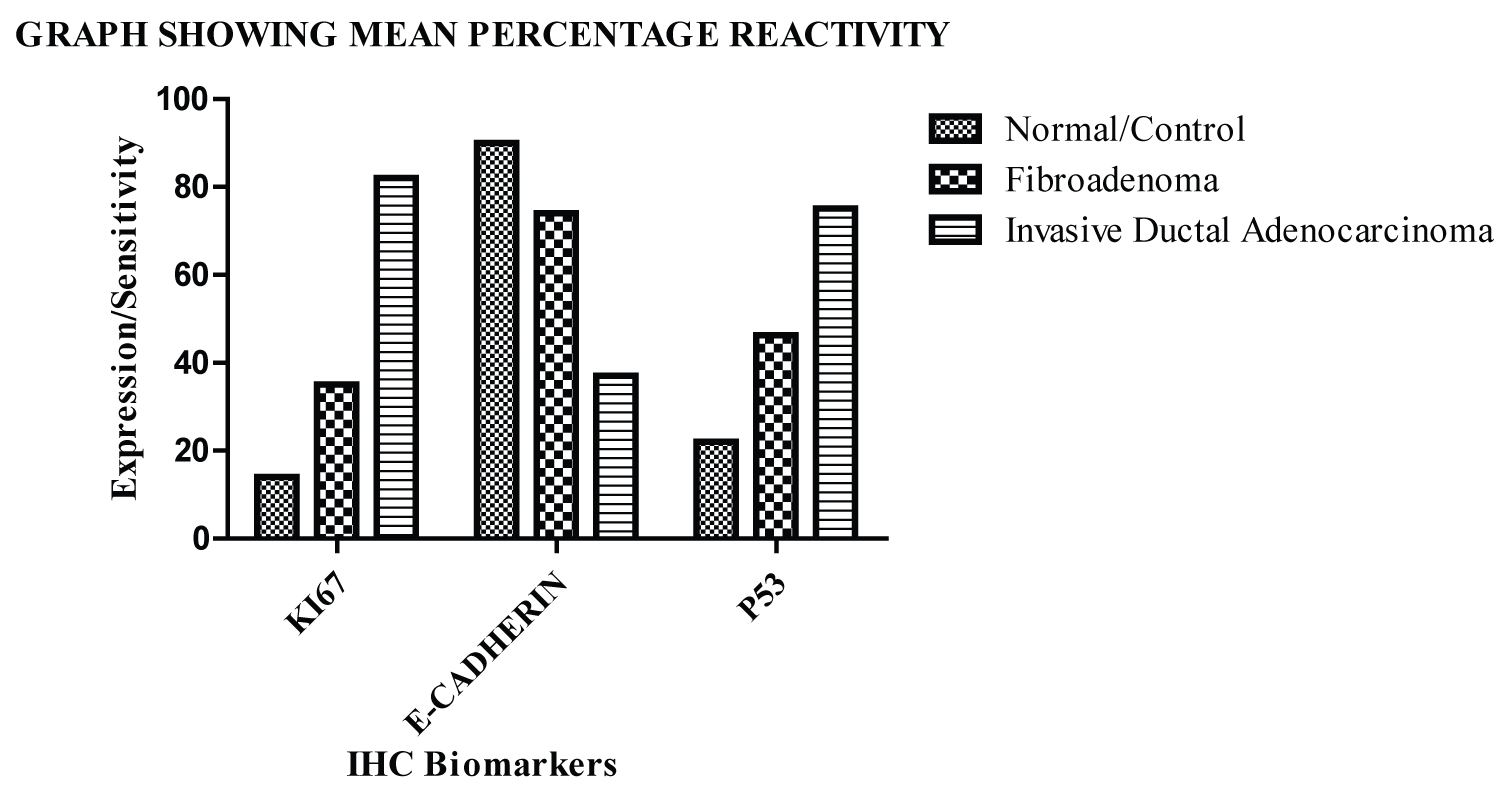 Graph 1: The graph above shows the gradual increase in percentage reactivity in KI67 and P53 from normal to invasive ductal adenocarcinoma and a decrease in E-Cadherin.
View Graph 1
Graph 1: The graph above shows the gradual increase in percentage reactivity in KI67 and P53 from normal to invasive ductal adenocarcinoma and a decrease in E-Cadherin.
View Graph 1
Table 4: Mean percentage reactivity of immunohistochemical markers in normal breast tissue, fibroadenoma and invasive ductal adenocarcinoma. View Table 4
The use of immunohistochemistry in the study and diagnosis of malignant lesions has been widely studied and serves as an efficient mode of diagnosing mutations that lead to the progression of benign lesions to malignant. With the constant advancement of early determination and medical procedure, medical procedure joined with postoperative radiotherapy and chemotherapy has turned into the best therapy for bosom malignancy patients, which can broaden the endurance season of patients [20]. Breast cancer is an illness where dangerous cells structure in the tissues of the breast [18]. Of patients who receive an early diagnosis of breast cancer after receiving adjuvant therapy, ~30% will eventually develop recurrence or metastasis [21]. Breast cancer recurrence and metastasis are severe clinical problems.
Ki67 is an antigen that subsists in the atomic network of fundamentally multiplying cells. It is found on chromosome 10 of human DNA and is viewed as a significant marker for measuring the proliferative action of cancer cells in later years [22]. From this study, our findings reveal that KI67 is expressed significantly in invasive ductal adenocarcinoma with high immunohistochemical expression and degree of reactivity while in the normal breast tissue and fibroadenoma; weak immunohistochemical staining was observed within the nucleus and classified as negative and mild respectively. The positivity rate of KI67 among the cases was twenty (20%) in normal, Thirty-six percent (36%) in fibroadenoma and one hundred percent (100%) in invasive ductal adenocarcinoma and the mean percentage reactivity was 14%, 35% and 82% in normal, fibroadenoma and invasive ductal adenocarcinoma respectively. According to Ahmed, et al., 2018, Ki67 has proven to be a useful predictive marker and is upregulated in breast cancer patients. The outflow of Ki67 is altogether diminished and this articulation level advances step by step with the proceeded with improvement of the disease as reported by [10]. Most of fibroadenomas were related with low Ki-67 cell immunoexpression demonstrating a reformist reduction in cell cycle restraint, yet not really high proliferative movement as in carcinoma as reported by [23]. The higher Ki-67 expression is associated with high grade, the more aggressive the cancer is, and thus, jeopardizing the survival of patients with early recurrence and metastasis stated by [24]. The nuclear protein Ki67 is an affirmed prognostic and prescient marker for the assessment of biopsies from patients with cancer [25]. The positive expression Ki67 is firmly connected to the development, attack, and lymphatic metastasis of breast cancer.
E-cadherin adhesive movement is controlled at the cell surface by a back to front instrument likely including allosteric guideline of the homophilic cement bond, practically equivalent to integrin guideline [26]. From this study, E-cadherin immunohistochemistry was found to be highly expressed within the normal epithelium and in Fibroadenoma, but less expressed in invasive ductal adenocarcinoma. The positivity rate for E-cadherin was 100% in normal cases, 100% in fibroadenoma cases and 32% in invasive ductal adenocarcinoma and the mean percentage reactivity was 90%, 74% and 37% in normal, fibroadenoma and invasive ductal adenocarcinoma respectively. E-cadherin has proven to be a useful predictive marker and is down regulated in breast cancer as stated by [27]. According to our findings which are in agreement with [28], the normal breast epithelium and fibroadenoma case showed a higher degree of E-cadherin expression, showing that the epithelium is still intact in normal and fibroadenoma and the integrity of the epithelium are upheld but in invasive ductal adenocarcinoma, there is a significant reduction in the expression of E-cadherin allowing a metastasis of the cells from the epithelium further invading the surrounding tissues. According to our results, E-cadherin is downregulated in the progression of the malignant condition and its expression within the breast epithelium shows the gradual progression of tumors from benign to malignant and has been reported by [14] to become unregulated showing cellular adhesion deterioration and gain of invasive properties and metastasizes. Downregulation of E-cadherin is identified with the deficiency of epithelial aggregate and the securing of the mesenchymal aggregate, which is related with obtrusive conduct in breast malignant growth. Abnormal E-cadherin articulation is more normal in invasive ductal adenocarcinomas with the possibility to create far off metastases [25].
P53 is the most frequently mutated gene in human cancer. Protein levels of p53 are controlled by the E3 ubiquitin protein ligase MDM2, which targets p53 for ubiquitylation and degradation [28]. The P53 gene is the most usually changed quality in different growths including breast cancer malignancy. About 30% of all breast cancer cases are described by changes in the P53 gene [29]. The greater majority of P53 mutations occur within the DNA-binding domain, which is situated between amino acids 120 and 300. Only one-tenth of them prompt deficiency of p53 capacities and the rest are missense transformations that outcome in the declaration of a mutated protein [30]. These mutations display predominant negative action over wild-type p53 if there should be an occurrence of heterozygosity just as it gives new oncogenic properties to the protein hence furnishing it with "gain of capacity" (GOF) features [31]. The p53 GOF structures were emphatically steadier on the protein level in malignant growth cells in contrast with their ordinary b counterparts [32]. From this study, P53 is expressed in fibroadenoma and invasive ductal adenocarcinoma with invasive ductal adenocarcinoma having higher immunohistochemical expression and degree of reactivity while in the normal breast tissue; weak immunohistochemical staining was observed within the nucleus and classified as negative. The positivity rate of P53 among the cases was Twenty-six point seven (26.7%) in normal, seventy-six (76%) in fibroadenoma and one hundred percent (100%) in invasive ductal adenocarcinoma and the mean percentage reactivity was 22%, 46.3%, and 75.1% in normal, fibroadenoma and invasive ductal adenocarcinoma respectively. Although P53 mutations are common in invasive breast carcinomas, few have been identified in intraductal breast carcinomas as reported by [17]. P53 gene may be correlated as regards a diagnostic marker of breast cancer as the loss of its normal function can lead toward the breast cancer progression and its overexpression associated with breast cancer [33].
From this study, some limitations of the IHC markers were observed.
For instance, KI67 can cause interobserver discrepancies therefore it may be used in adjunct to other auxiliary markers.
P53 as a hallmark characteristic of cancer can be expressed in varying degrees in the premalignant condition.
This study has shown that to maximize both predictive and diagnostic abilities of these IHC markers, they should be used in adjunct with other similar markers to eliminate all possible limitations.
The use of other molecular techniques such as RT-PCR and Western blot together with these predictive biomarkers offers more reliable results and as such may be employed.
KI67 showed majorly nuclear staining across the epithelium while E-cadherin showed consistent membranous staining across the epithelial cells and P53 showed strong nuclear staining in the epithelial cells. This study has established the usefulness of KI67, E-cadherin and P53 immunohistochemical markers in studying the breast tissue from normal to fibroadenoma and then to Invasive ductal adenocarcinoma. KI67 and E-cadherin were found to be more useful in studying the progression as characterized by KI67's increase and E-Cadherin's decrease along the progression.
The authors acknowledge Olalekan Adegoke Aremu from the department of Morbid Anatomy and Forensic Medicine Obafemi Awolowo Complex (OAUTH) for his contribution to this study in the sample immunohistochemical process.
None.
The authors declare no conflict of interest.