Follicular lymphoma (FL) is an indolent lymphoma and is associated with a long survival time. However, there are patients who had an early progression after standard therapy. Our aim was to analyze the prognostic value of Ki67, CD10 and MUM1 expression in FL.
Immunohistochemical staining for Ki67, CD10 and MUM1 was performed on formalin-fixed paraffin-embedded biopsy tissues from 27 patients with FL. Patients were grouped according to the levels of the Ki67 proliferation index (PI). Survival times were analyzed and the cut-off value for Ki67 PI was determined. The patients were also divided into groups according to the presence of CD10, MUM1and MUM1 positive/CD10 negative.
Ki-67 PI levels were as follows: ≤ 20% in 14 patients, 21-40% in 6 patients, 41-60% in 4 patients, > 60% in 3 patients. A cut-off value of 60% revealed significantly different survival rates. OS, PFS in the Ki67 > 60% group decreased significantly for 5 years, and the difference was statistically significant (37.5% vs. 100%, p = 0.000; 0% vs. 66.7%, p = 0.016; respectively). The CD10 negative group had a less favorable outcome than the positive group in the OS rate at 5 years (72% vs. 100%, p = 0.049) and the PFS rate at 5 years (25.3% vs. 72.6%, p = 0.017). OS and PFS in the group with MUM1 positive/CD10 negative decreased significantly for 5 years than in the group without MUM1 positive/CD10 negative (4.7% vs. 100%, p = 0.002; 0% vs. 66.4%, p = 0.006; respectively).
Ki67 > 60%, CD10 negative, MUM1 positive/CD10 negative are poor prognostic factors in FL.
Ki67 proliferation index, CD10, MUM1, Follicular lymphoma, FL, Prognosis
Follicular lymphoma (FL) is a B cell neoplasm that is usually classified as an indolent type with a long overall survival (OS). However, there are patients who had an early progression after standard therapy [1]. Therefore, systems were proposed to classify the prognosis. The main systems used include: FLIPI, FLIPI2, m7-FLIPI.
FLIPI was developed on the basis of results of heterogeneous treatment, which was between different centers and varied over time. These included age > 60, Ann-Arbor stage III–IV, hemoglobin level < 120 g/L, lactate dehydrogenase (LDH) > upper limit of normal and more than 4 nodal sites [2]. However, FLIPI has been validated after the advent of rituximab-containing treatment. Subsequently, a second prognostic model was called FLIPI2, which was developed in the post-rituximab era and using PFS (progression-free survival) rather than OS (overall survival) as an endpoint. Five variables were selected: beta-2 microglobulin (β2M) above the normal upper limit, the longest dimension of the single lymph node greater than 6 cm, bone marrow involvement, hemoglobin less than 120 g/L and age greater than 60 [3].
Since the genomic landscape of FL is characterized by highly recurrent mutations, the m7-FLIPI model was developed after assessing the mutation status of 74 genes. The m7-FLIPI score was calculated by integrating the mutation of these seven genes with the FLIPI and ECOG performance status [2]. However, there was a study that suggested that the prognostic value of the m7-FLIPI model seems to depend on the therapeutic protocol [4].
However, risk factors continue to be studied, especially immunohistochemical markers that have diagnostic and prognostic value [5].
Ki67 recognizes a proliferative specific nuclear antigen. Several studies have suggested that high Ki67 expression is a poor prognostic factor in diffuse large B- cell lymphoma (DLBCL), mantle cell lymphoma (MCL) [6,7]. However, the effect of Ki67 on FL is controversial. Yukiko Kawaguchi, et al. suggested that high expression of Ki67 tended to be associated with short overall survival in FL [8]. Otherwise, the study by Tian Xue, et al. showed that a higher Ki-67 index appears to have a positive effect on PFS in patients with FL [9].
CD10 is a proteolytic enzyme expressed on the cellular membrane. During lymphocyte differentiation, CD10 first appears on pre-pre-B cells (pre-B-I cells) and is lost during maturation in naive B cells [10]. Although CD10 are expressed in the vast majority of FL, several studies are still looking for an association between CD10 expression levels and survival time [11,12]. However, studies such as those of Francisca I Camacho or Nurija Bilalovic agreed that the patient with CD10 positive had a better survival time, but did not show any correlation with expression level [11,12]. The recent study by Chen, et al. showed that loss of CD10 expression could play a role in the pathogenesis of leukemia change [13].
MUM1is a transcriptional factor and plays a role in cell proliferation, differentiation and survival. MUM1 is usually expressed in plasma cells and is rarely expressed in germinal center (GC) B cells. It was supposed to be absent in follicular lymphoma [14]. However, there are few studies interested in MUM1 expression as well as prognostic significance in FL. Luc Xerri, et al., Eric D His, et al. suggested that MUM1 had an unfavorable effect on survival time [15,16]. Kennosuke Karube, et al. indicated that FL with MUM1 positive/CD10 negative is different from typical FL with respect to biologic and clinical features [17].
In the present study, our objective was to determine the cut-off value of Ki-67 PI as a prognostic factor and to assess the prognostic value of CD10 and MUM1 in patients with FL.
From March 2016 to July 2021, in Bach Mai Hospital, Hanoi, Vietnam, a total of 27 patients with de novo confirmed FL according to the WHO 2008 classification of hematopoietic and lymphoid tumors were enrolled in our study [18]. Diagnoses of FL were confirmed by pathological examination of lymph node biopsies.
Immunohistochemical staining for CD20, CD10, CD3, CD5, CD23, Bcl2, Bcl6, MUM1 and Ki67 was performed on formalin-fixed paraffin-embedded tissue biopsy samples.
All patients were treated with conventional combination chemotherapy as follows: R-CHOP (rituximab- cyclophosphamide, doxorubicin, vincristine, prednisolone); R-COP (rituximab- cyclophosphamide, vincristine, prednisolone).
Classification of risk was defined according to FLIPI. Classification of stage was defined according to the criteria of Ann Arbor [19].
High tumor burden was defined according to the criteria of GELF (Groupe d'Etude des Lymphomes Folliculaires)1 [20].
The response to remission therapy was evaluated according to International Working Group consensus response evaluation criteria in lymphoma (RECIL 2017) [21].
CD10, Bcl6 and MUM1 staining were scored as negative (≤ 30% of cells) or positive (> 30% of cells) [22].
To determine the cut-off value for the Ki-67 proliferative index (PI), patients were grouped according to Ki-67 PI values (cut-off points: ≤ 20%, 21%-40%, 41%-60%, and > 60%) and the associated survival rates were analyzed. This cut-off value found was estimated to further divide Ki67 into low and high proliferative index. The patients were grouped according to this Ki67 PI value (cut-off points). The patients were divided into groups according to the presence of CD10, MUM1 and MUM1+/CD10- as well.
Overall survival (OS) was calculated from the day of diagnosis to death from any cause. Progression-free survival (PFS) was calculated from the day of achieving the best remission to disease progression, relapse, or death from any cause or remaining alive and disease free.
Overall survival and progression-free survival time were estimated using the Kaplan-Meier method and then compared by the log-rank test.
The characteristics of the patients are summarized in Table1. The median age of the patient was 58 years (range 34-78 years) and the male to female ratio was 0.93.
Table 1: Patients characteristics. View Table 1
The clinic pathological profiles of the patients are summarized in Table 2. Ki67 PI levels were as follows: ≤ 20% for 14 patients, 21-40% for 6 patients, 41-60% for 4 patients, > 60% for 3 patients.
Table 2: Clinicopathological profile of patients. View Table 2
A cut-off of 60% for Ki67 reveals significant differences in survival. OS and PFS in the group with Ki67 PI > 60% decreased significantly for 5 years, and the difference was statistically significant (p = 0.000; p = 0.016, respectively) (Table 3, Figure 1 and Figure 2).
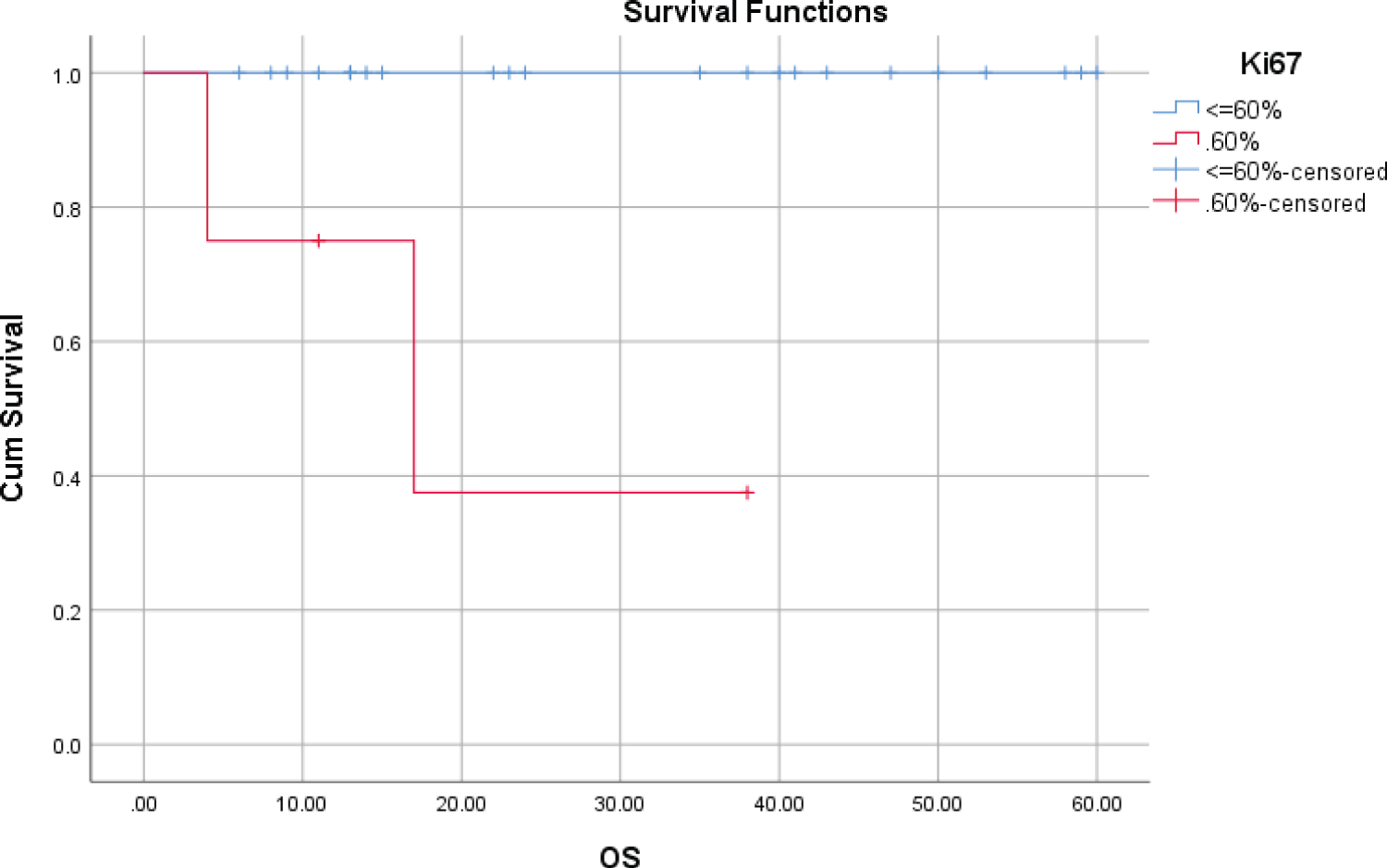 Figure 1: OS at 5 years according to presence of Ki67 > 60%.
View Figure 1
Figure 1: OS at 5 years according to presence of Ki67 > 60%.
View Figure 1
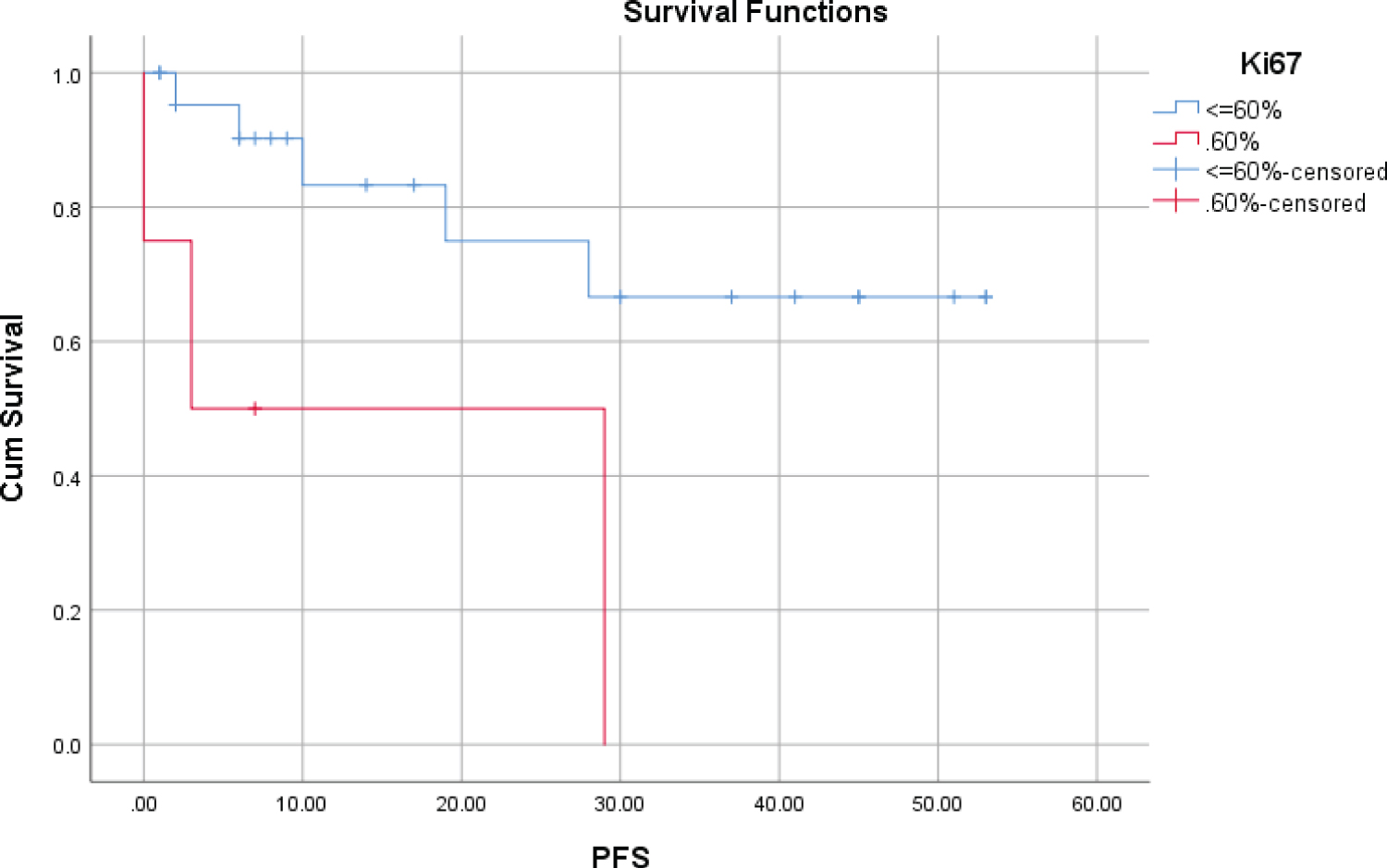 Figure 2: PFS at 5 years according to presence of Ki67 > 60%.
View Figure 2
Figure 2: PFS at 5 years according to presence of Ki67 > 60%.
View Figure 2
Table 3: Prognosis factor on overall survival and progression free survival. View Table 3
The CD10 negative group had a less favorable outcome than the CD10 positive group, such as the OS rate at 5 years (72% versus 100%) and the PFS rate at 5 years (25.3% versus 72.6%), with the statistically significant difference (Table 3, Figure 3 and Figure 4).
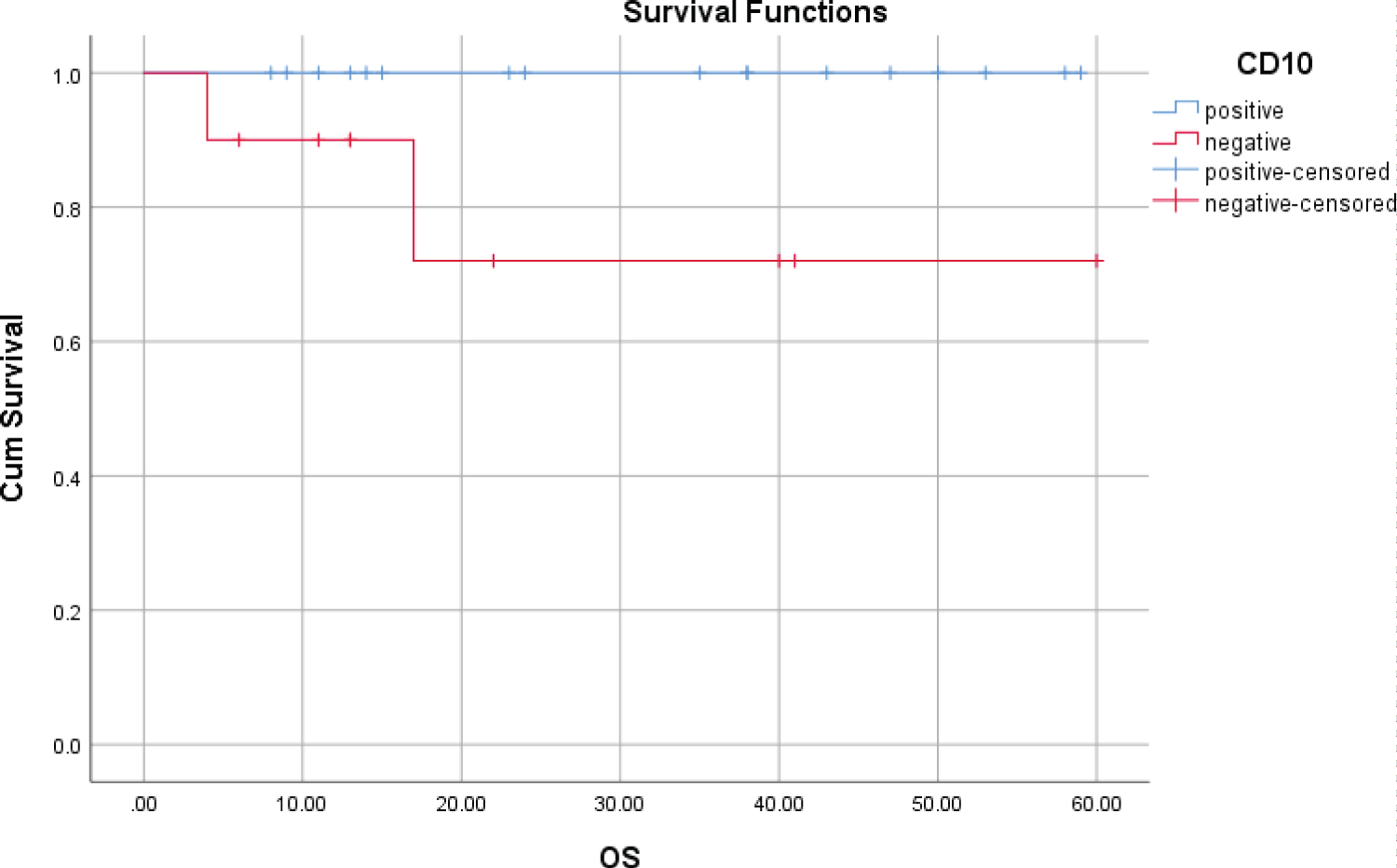 Figure 3: OS at 5 years according to presence of CD10.
View Figure 3
Figure 3: OS at 5 years according to presence of CD10.
View Figure 3
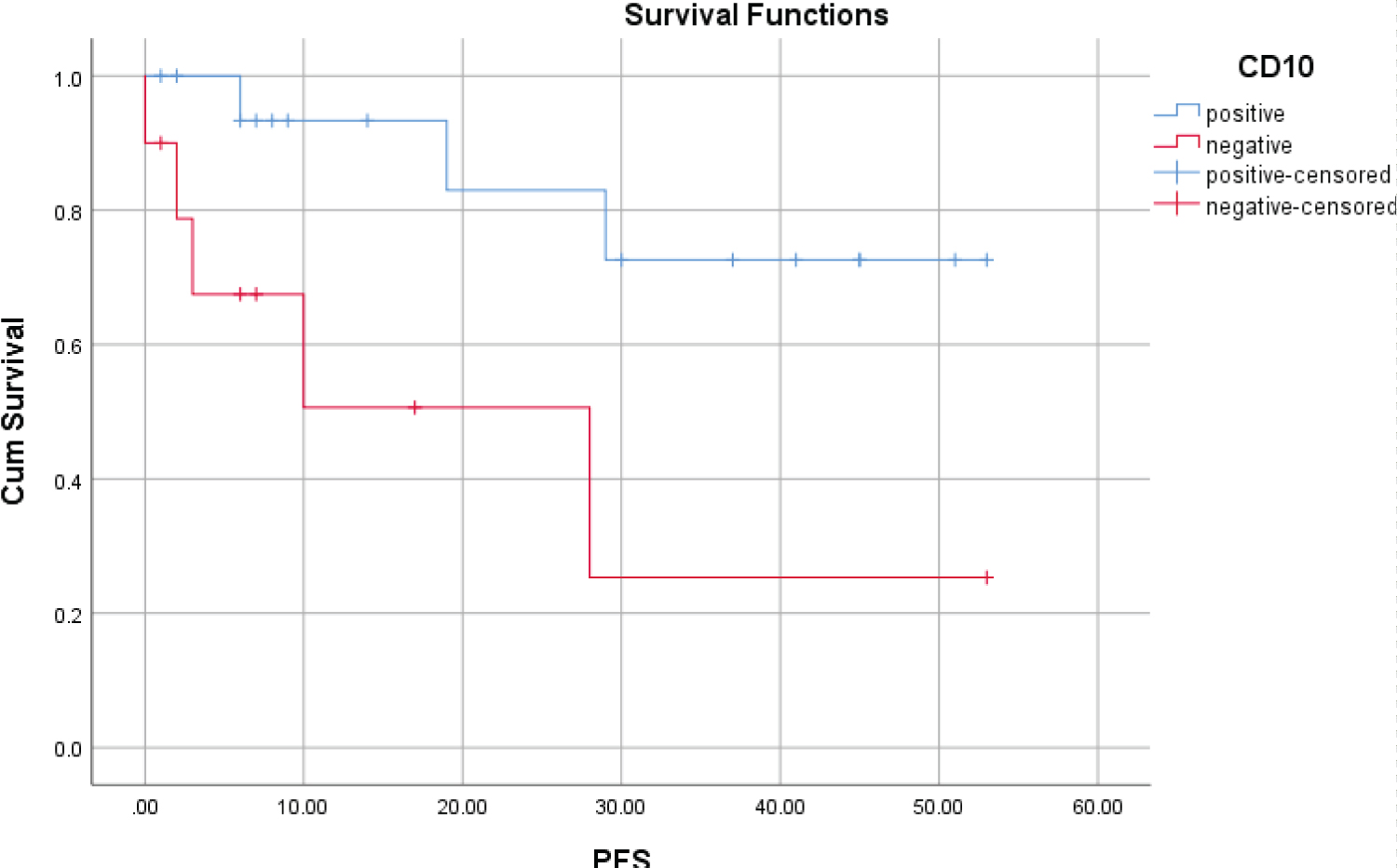 Figure 4: PFS at 5 years according to presence of CD10.
View Figure 4
Figure 4: PFS at 5 years according to presence of CD10.
View Figure 4
There were no statistically significant differences in OS, PFS at 5 years between the groups according to the presence of MUM1. However, MUM1 had an adverse effect in the patients with CD10 negative. OS and PFS in the group with MUM1 positive/CD10 negative decreased significantly for 5 years than in the group without MUM1 positive/CD10 negative, (p = 0.002; p = 0.006) (Table 3, Figure 5 and Figure 6).
 Figure 5: OS at 5 years according to presence of MUM1+/CD10-.
View Figure 5
Figure 5: OS at 5 years according to presence of MUM1+/CD10-.
View Figure 5
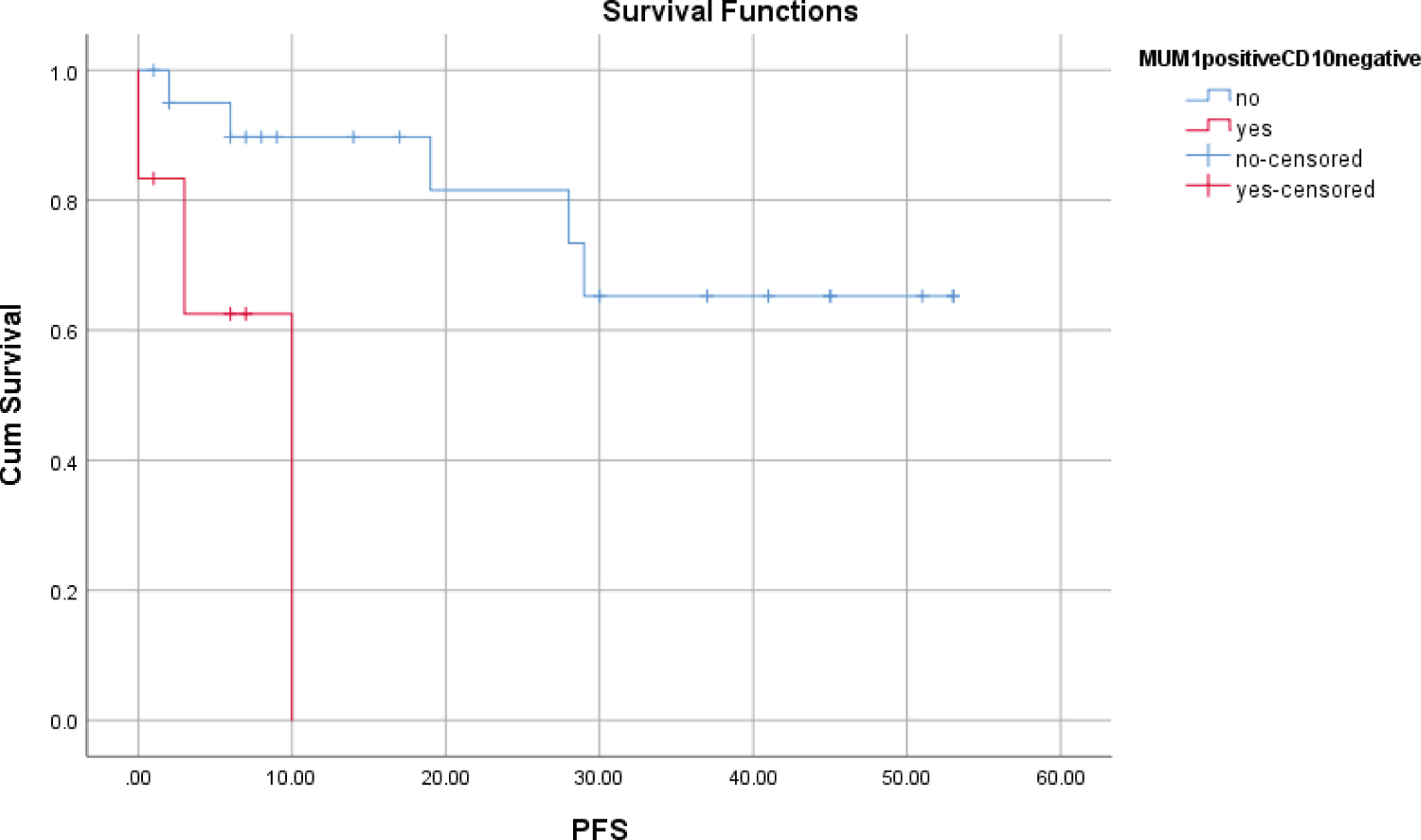 Figure 6: PFS at 5 years according to presence of MUM1+/CD10-.
View Figure 6
Figure 6: PFS at 5 years according to presence of MUM1+/CD10-.
View Figure 6
In our study, the median age of the patients was 58 years, equivalent to previous studies [23,24]. Immunohistochemical staining results showed a negative CD10 rate of 37%, which is quite higher compared to the other studies. Anna Szumera-Cieckiewicz, et al. had a rate of 22.4%, NM Almasri, et al. had a rate of 11% [25,26]. The positive rate of MUM1 was 44.4%, higher than in the studies of Kikeri N. Naresh (37%), Eric D Hsi (14.4%) [16,22]. The majority of patients had Ki67 PI ≤ 20%, only 11.1% of patients had Ki67 PI > 60%. This result is similar to the studies by Rukmini Berbaruah, et al., MT Sylvia, et al.: High Ki67 expression is rare in FL [23,24].
The prediction value of the Ki67 proliferation index in FL is controversial. Luc Xeri, et al. showed that high levels of Ki67 positivity (cut-off value of 10.25%) were significantly associated with shorter PFS [15]. Yukiko Kawaguchi, et al. suggested that a high expression of Ki67 (30%) tended to be associated with short OS in FL, but the difference was not statistically significant (p = 0.058) [8]. Francisca I Camacho, et al. did not see any differences between the groups defined by Ki67 expression [11]. The most recent study by Tian Xue, et al. had the opposite result: Patients with Ki67 ≥ 30% had significantly better PFS than patients with Ki67 < 30% [9]. Our study showed that at high expression level (> 60%), Ki67 has an adverse effect on OS and PFS for 5 years. Ki67 is a nuclear protein that is associated with cell proliferation. Therefore, it is difficult to assess the role of a proliferative factor in such a slow-growing tumor as FL.
CD10 is highly expressed in FL and is one of the markers used to diagnose FL. The absence of CD10 in FL was of interest to several authors and to investigate whether it had any association with survival time. Francisca I Camacho, et al. and Nurija Bilalovic, et al., showed that CD10 expression was associated with a significantly better outcome [11,12]. Chen, et al., in FL patients with leukemic presentation, showed that CD10 was detected in leukemic cells by flow cytometry in 38% of cases and in lymphoid tissue by immunoshistochemical staining in 77% of cases (p = 0.0471) [13]. This result suggested that CD10 gradually lost expression as cells progressed to more malignant. Our study also showed that the CD10 negative group had a less unfavorable outcome.
MUM1 is a rare marker in FL that is interested in relation to survival time. FL is defined as a neoplasm composed of germinal center B cells, so it is generally positive for CD10 and negative for MUM1. However, there have been some studies that refer to FL with MUM1 positive/CD10 negative. Kennosuke Karube, et al. indicated that the MUM1 positive/CD10 negative group is atypical FL with different biology and clinical characteristics [17]. Our study also indicated that MUM1 positive/CD10 negative expression was associated with poor OS and PFS. Luc Xerri also recommended that MUM1 can be used as a routine biomarker for the clinical management of patients with FL [15]. Otherwise, Anna Szumera-Cieckiewicz, et al. suggested that the lack of CD10 expression corresponded to the absence of Bcl2 and higher expression of MUM1, but had no impact on long-term outcomes [25]. Perhaps, for patients with FL with negative CD10, a further evaluation of MUM1 should be considered as an additional prognostic factor.
Markers Ki67, CD10 and MUM1 are valuable in the prognosis of follicular lymphoma.
No financial support was received for this study.
The authors declare no conflicts of interest.