Background: Head and neck cancer (HNC) patients undergoing chemo/radiation (C/RT) commonly experience severe and persistent distress associated with treatment related fear and physical side effects such as xerostomia, dysphagia, and dryness of mouth. Cortisol, a stress sensitive hormone, can be easily measured in saliva to reflect biobehavioral responses to such stressors. Unfortunately, it has not been used in this population due to concerns associated with C/RT related xerostomia.
Methods: In a proof-of-concept study, we explored the feasibility of collecting salivary cortisol as a marker of fear and distress in HNC patients. Ten HNC subjects undergoing C/RT provided saliva samples for 3 consecutive days across three timepoints (pre-treatment, 3-weeks and 1-month post-treatment) and completed concurrent depression, anxiety and swallowing related fear measures.
Results: Salivary cortisol collection adherence was between 60-80%. It was not impacted by xerostomia. Diurnal cortisol pattern's demonstrated dysregulation at pretreatment in 62%, and flattened aberrant slopes continued at 3-weeks and beyond in 50% of subjects.
Conclusions: Our study supports the feasibility and utility of salivary cortisol measurement in HNC patients across the treatment trajectory. Diurnal cortisol measures may be a valuable tool to detect and monitor treatment distress during C/RT in this population.
Salivary Cortisol, HPA dysregulation, Depression, Anxiety, Head Neck Cancer
Cortisol is a stress sensitive hormone known to reflect changes in biobehavioral responses e.g., depression and anxiety in patient groups [1]. It is released by the adrenal medulla and is a useful indicator of poor functioning of the Hypothalamic - Pituitary - Adrenal (HPA) axis [1,2]. Cortisol follows a diurnal rhythm i.e. highest level around the time of awakening followed by progressively reduced levels throughout the day [1]. Cortisol dysregulation can occur due to both acute stressors (lasting for a few minutes to a few hours such as during a cancer diagnosis) and chronic stressors (such as during the treatment trajectory) [1].
A diagnosis of cancer provokes a strong bio-behavioral response, and may be exaggerated over the treatment trajectory causing dysregulated levels of cortisol [3,4]. Head and neck cancer (HNC) patients are well recognized as having higher levels of distress (anxiety and depression) along with social isolation as compared to other patient groups [5,6]. However, the profile of cortisol production in this group has not been evaluated. It is reported that because this cancer affects the most visible part of the body along with its impact on basic functions such as eating, breathing, and speaking, distress is high in this group [7]. Recent data suggests that swallowing related fear may also be another distinct stressor related to ongoing behavioral feeding burden in HNC [8]. Stressors such as depression and anxiety which are highly prevalent in HNC patients have also been purported to be associated with dysregulation of neuroendocrine hormones such as cortisol [4,9,10]. Further, research in breast cancer survivors has shown that elevated cortisol awakening response measured via salivary cortisol is a significant risk factor for depression [9,10]. Recent studies have shown that a stress related bio-behavioral response can impact tumor biology via mediation to tumor signaling pathways showing strong links to tumor growth, metastasis and recurrence [11,12]. In concert with this, monitoring of salivary cortisol has been shown to be useful in other fields for identifying individuals that may need further input to support coping or behavioral management of stressors [13-17]. Data also suggests that blunted/flattened and dysregulated cortisol rhythms in those groups was related to poorer memory function, lower perceived social support, and higher perceived stress [1,16,17]. Despite the negative and persistent impact of distress in the HNC population, studies examining the modulation of cortisol by fear, depression, anxiety, or tumor biology in HNC patients are scarce.
Treatment of HNC (chemotherapy with or without radiation (C/RT)) commonly involves a range of oral side effects such as xerostomia (dryness of mouth), mucositis, dysphagia, dental caries, and trismus (inability to open mouth) among others [6-8]. Consequently, a frequently suggested barrier for the lack of investigation into salivary cortisol in HNC has been the perceived impact of xerostomia limiting saliva sampling. This issue of oral side effects impacting saliva collection feasibility, along with the perceived additional treatment-related burdens from the disease, has resulted in a significant gap in exploration of cortisol as a valuable marker of distress within this group. To date no study has evaluated the value of salivary cortisol collection in HNC. Consequently, our preliminary proof-of-concept study sought to investigate the following 1) the feasibility of collecting viable salivary cortisol specimens across time points in HNC undergoing C/RT, 2) to evaluate the pattern and concordance of salivary cortisol slope to perceived distress measured by standard and valid biobehavioral measures in HNC patients, 3) to compare the differences in cortisol levels before and after treatment in a sample of HNC patients.
This proof-of-concept study included 10 patients with HNC cancer confirmed by clinical history and exam, who were undergoing C/RT at a university hospital cancer clinic. They were recruited over a 6-month period. Patients were included if they were receiving HNC treatment at our facility, had no previous history of cancer - related illness or history of non-oral feeding for any reason and were not taking any medications known to adversely affect cortisol secretion or interfere with cortisol measurement. Eligible patients were identified by the attending physicians and referred for evaluation by the study coordinator. The study was approved by the local institutional review board (IRB201700078).
Priori determined longitudinal study time points included baseline (prior to C/RT; T1), 3 weeks into C/RT (T2), and 1-month post C/RT (T3). Two saliva samples (awakening and 9pm) were collected for 3 consecutive days at each time point, resulting in 6 cortisol samples collected at each time point. At baseline, participants completed saliva kits and survey demographics including cancer related information. Self-rated bio-behavioral stress measures included: Center for Epidemiologic Studies Depression Scale [CESD] (depression), State trait anxiety inventory [STAI] (anxiety), and Swallowing Kinesophobia Scale [SWKS] (swallow-related fear) also completed at each assessment point.
Salivary cortisol measurement was completed by an independent external lab (using the ELISA (enzyme-linked immunosorbent assay)) and results returned for analysis. Cortisol rhythms were measured by individual linear regression. The slope (β) of the regression line predicting cortisol level from time of day was used to represent each participant's cortisol rhythm. The regression of the 6 cortisol values on the hour of sample collection was calculated, with data pooled over the 3 days for each participant for each day. A typical negative or a steep diurnal rhythm (smaller β coefficients indicating a rapid decrease in cortisol) was defined as decreasing cortisol levels over the course of the day, and an atypical flat diurnal (larger β coefficients) indicating slower decreases or abnormally timed peaks) rhythm was defined as a flat pattern with no/limited decline. The consistent diurnal rhythm was defined as cortisol secretion rhythms (β) that were highly similar over 3 days, whereas the inconsistent diurnal rhythm had variable patterns of rhythm across 3 days. Mean salivary concentration at 3 time points were correlated with depression, anxiety, and fear of swallowing.
Three primary domains of behavioral stress in HNC (depression, anxiety, and swallow-related fear) and two feeding related outcomes were measured at each time point using patient rated outcome measures. These included:
Center for Epidemiologic Studies Depression Scale (CESD): The CESD is a 20-item self-report tool developed to screen for depression scored on a 5-point Likert scale. It has been validated for use in HNC populations with a cut off of ≥ 16 reported to detect depression [6].
State Trait Anxiety Inventory (STAI): The STAI is a validated 20 item self-report tool to measure anxiety. Each item is evaluated on a 4-point Likert scale. A raw score cutoff point of >40 has been suggested to detect clinically significant symptoms for this scale [18,19].
Swallowing Kinesiophobia scale (SWKS): The SWKS is a validated 17 item self-report tool to measure fear of swallowing and swallowing related movement in patients with HNC. A cutoff point of ≥29 has been suggested to detect clinically significant fear [8].
Functional Oral Intake Scale (FOIS): The FOIS is a 7-point ordinal scale that describes the typical oral intake with consideration for alternate food sources, modifications in food/liquid, and required adjustments used by the patients to sustain a particular level of intake. This scale has demonstrated strong validity and inter-rater reliability. The score is derived by patient/family interview and/or medical chart review [20,21].
Non-oral Feeding: The presence or absence of non-oral feeding sources was noted (+/-) at each assessment point.
Descriptive and graphic analyses were used to evaluate the data distributions, search for outliers, and describe sample characteristics and cortisol secretion level. Outliers with correct values were retained. Data was summarized using the means, median, standard deviation, and range where appropriate. Non-parametric correlations using Spearman’s Rho were used to evaluate relationships among continuous variables. Mann Whitney U test was used for the evaluation of ranks among categorical and continuous variables. The non-parametric Wilcoxon signed rank test was used to analyze associations among salivary cortisol and the three domains of distress. Given the smaller sample size for this study, missing data were retained at each timepoint analysis where possible using pairwise analysis approach. Statistical significance was set apriority α = 0.05. The Statistical Package for the Social Sciences (SPSS version 28.0; SPSS Inc, Chicago, Illinois) was used for all analyses.
In total 10 Caucasian patients (mean age 59 (SD = 9.2); 8 males, 2 females) were enrolled in this pilot study. All study patients were diagnosed with squamous cell carcinoma with tumor location in hypopharyngeal (30%) and oropharyngeal sites (70%), Participant tumor stages were T1-T3 (mode = 3) with the most common tumor site in the tonsil. Not all patients had the same treatment regime, 53% of the cohort received chemoradiotherapy plus radiotherapy which was provided by intensity-modulated radiation therapy (IMRT) in all cases. Most of the patients were employed (70%) with 50% having an education status of lower than a high school degree. At baseline, 6 (60%) were modifying their diets to accommodate feeding. Additional baseline characteristics of the sample are displayed in Table 1.
Table 1: Participant characteristics View Table 1
At 3 weeks into C/RT (T2), one subject had withdrawn, and three subjects had a reactive percutaneous gastrostomy (PEG) feeding tube placed. At this timepoint 22% (2) patients demonstrated oral mucositis (grade 2) on the NCI common toxicity criteria [22] and 33% (3) self-reported dry mouth. Average weightloss for the group was 4lb (SD: 5.7)
By 1-month post C/RT (T3) two patients had been hospitalized briefly for dehydration and an additional subject was receiving alternate feeding however, at this time point only 3 subjects were consuming no oral nutrition. All of these patients were receiving C/RT. Dry mouth was reported by 44%. Mean weight loss for the group at this timepoint was 10 lbs (SD:10.4).
At baseline, the mean self-rated depression score for the group was 11.1 (SD: 10). The number of patients who self-rated above 16 was 4 (40%). Baseline mean self-rated score for anxiety was 10.6 (SD: 4.8). The number of patients who self-rated above the cut point for significant stress (>40) was 2 (20%). Baseline mean self-rated SWKS score was 34.7 (SD: 6.53). The number of patients rating in the significant fear range was 8 (80%). Depression (CESD) was strongly and positively correlated with anxiety (STAI) at all three time points (Baseline: r = 0.88, p = 0.01; during C/RT r = 0.77, p = 0.02; 1-month post C/RT r = 0.78, p = 0.03). Swallowing fear (SWKS) was correlated to both depression (r = 0.762, p = 0.047) and anxiety (r = 0.759, p = 0.048) at the 1-month post C/RT time point.
Overall response rate for cortisol collection across the whole study was 83.3%. Response rate at baseline was 100% for surveys and 80% for saliva collection. During treatment, survey response rate was 80% and cortisol collection was 60%. Lastly, at 1-month post C/RT, survey response rate was 70% and cortisol collection was 50% (Figure 1). Data loss was frequently seen in patients with unexpected hospitalizations (n = 2) and lower SES (n = 3).
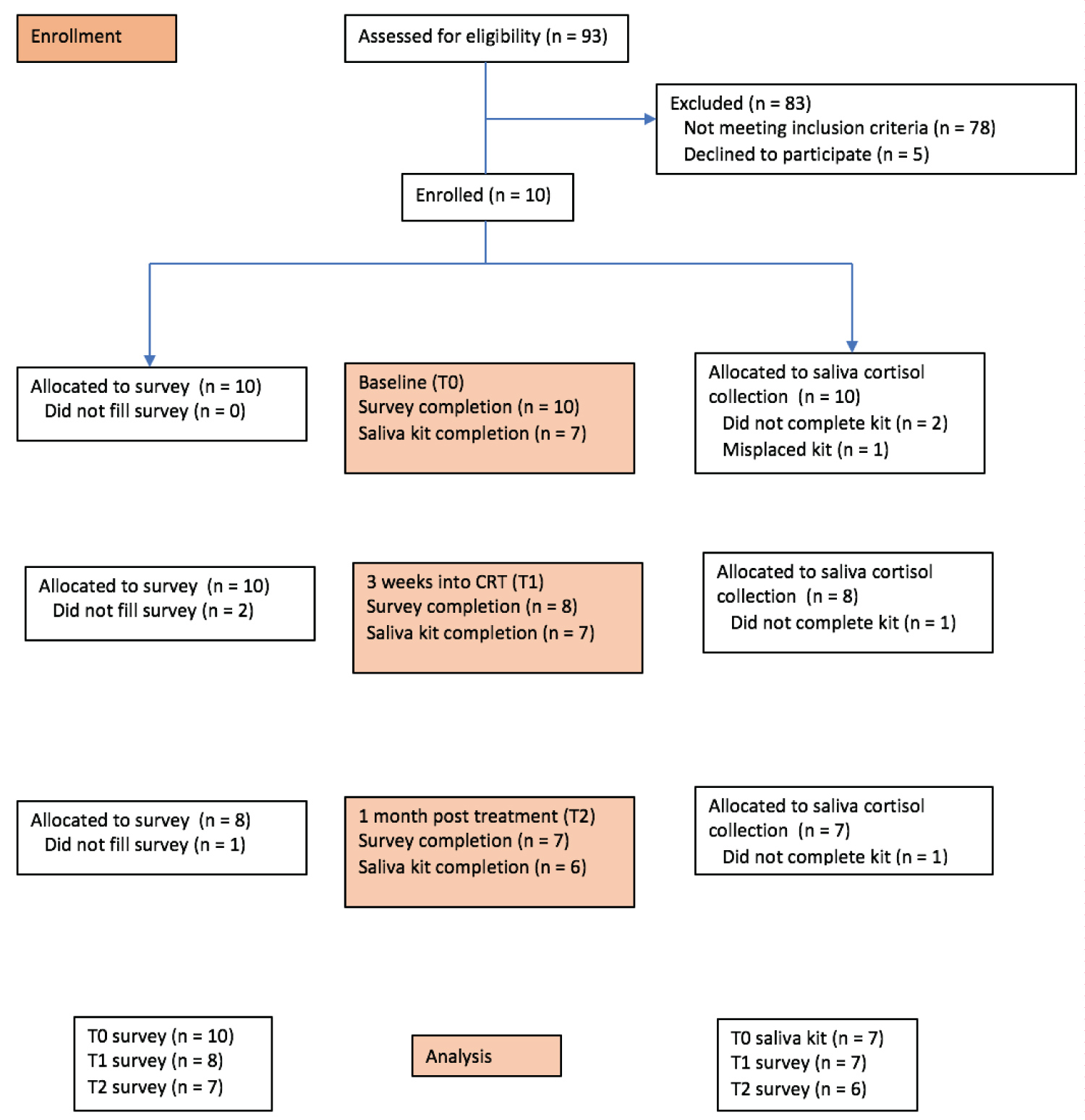 Figure 1. Study flow chart
View Figure 1
Figure 1. Study flow chart
View Figure 1
Evaluation and comparison of Log cortisol slopes across participants for morning and evening collections demonstrated dysregulated circadian diurnal patterns in 5 patients at baseline (62.5%), 4 (57%) at 3 weeks (i.e. during treatment) and 3 (50%) at one-month post C/RT (Figure 3, Figure 4 and Figure 5). Moreover, flattened aberrant patterns (larger mean β coefficient values ± SD) were more commonly noted (57%) at the 3-week (within treatment) time point (Figure 2).
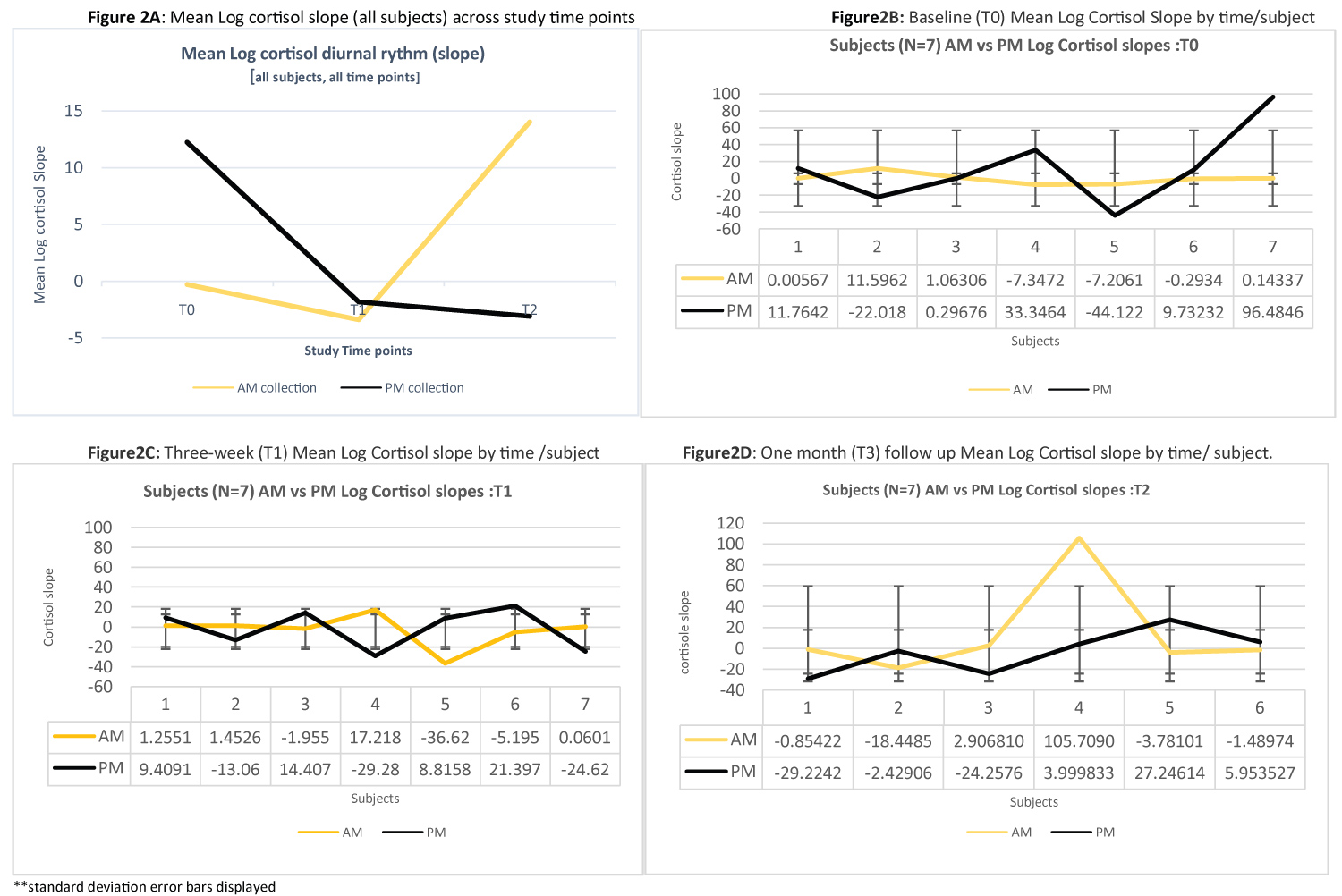 Figure 2: Mean log cortisol slope for total group, and by subject over the study time points.
View Figure 2
Figure 2: Mean log cortisol slope for total group, and by subject over the study time points.
View Figure 2
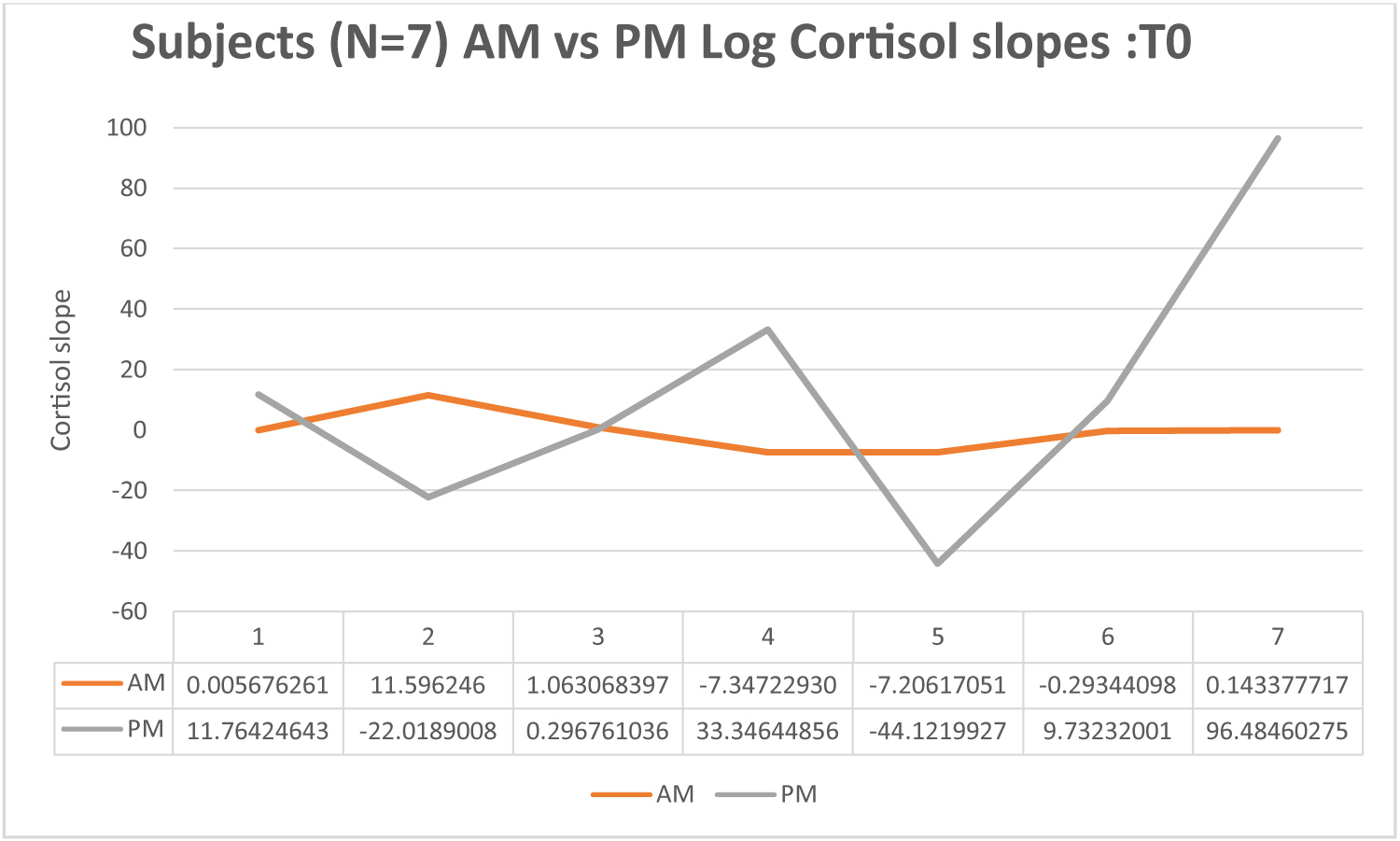 Figure 3. Baseline Log Cortisol Slope by Time
View Figure 3
Figure 3. Baseline Log Cortisol Slope by Time
View Figure 3
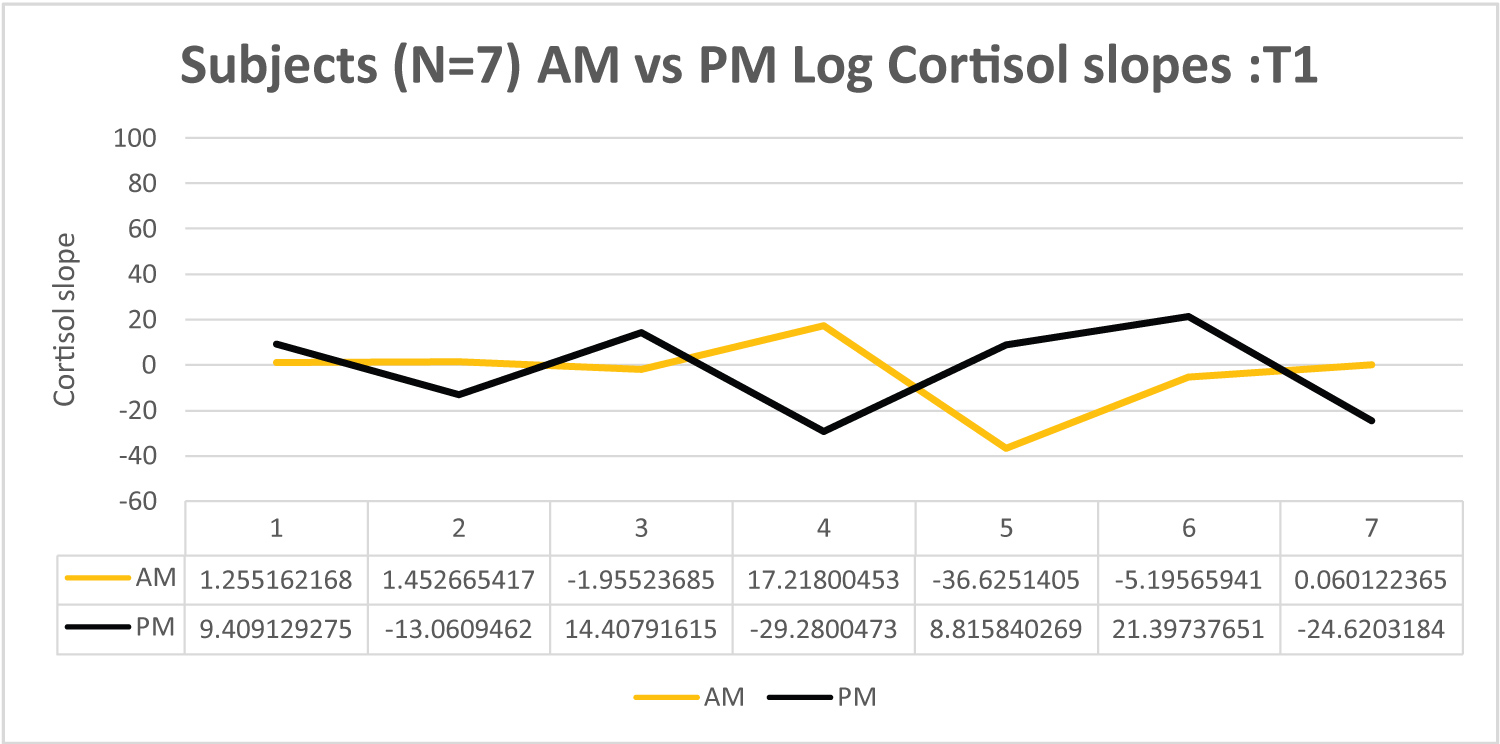 Figure 4. Within treatment (3-weeks) Log Cortisol slope by time
View Figure 4
Figure 4. Within treatment (3-weeks) Log Cortisol slope by time
View Figure 4
 Figure 5. One month follow up Log cortisol slope by time.
View Figure 5
Figure 5. One month follow up Log cortisol slope by time.
View Figure 5
Results revealed no relationship between mean log salivary cortisol slope at any time point to age, gender, education, tumor size, tumor site, weight loss, presence of PEG tube feeding, reported oral dryness or mucositis. Presence of dysphagia (as measured by the surrogate FOIS level) was significantly related to level of swallowing related fear both during C/RT (p = 0.024) and at 1-month post C/RT (p = 0.048). Mean Log Cortisol slope was significantly related to swallowing fear (SWKS) at both 3 weeks (U = -2.366, P < 0.018) and 1-month (U = 2.201, p < 0.028) with significantly fearful patients demonstrating more dysregulated profiles. Mean Log cortisol slope was also found to be significantly different between patients receiving IMRT (-0.1322) vs C/RT (-0.0330) during the treatment period (U = -2.490, p = 0.028), with C/RT patients demonstrating more dysregulated patterns.
Univariate relationships between mean salivary cortisol level and the domains of stress were performed using non-parametric correlations at each time point. Results revealed that anxiety (STAI) and depression (CESD) were both strongly correlated with mean cortisol level at all time points baseline (Z = 2.366, p = 0.018), during C/RT (Z = -2.366, p = 0.018), and at 1-month post C/RT (Z = -2.201, p = 0.028). Moreover, swallowing related fear (SWKS) was also significantly related to mean cortisol level at baseline (Z = -2.366, p = 0.018) and during C/RT (Z = -2.197, p = 0.28).
At all timepoints cortisol measurement demonstrated stability of sampling in subjects. No significant difference within timepoint sampling variability was demonstrated. Descriptively our subjects showed a limited time of day effect on cortisol. The magnitude of difference between AM and PM cortisol levels was only statistically significant at the 3-weektime point. At baseline, only 28% (2) demonstrated elevated AM samples (near the time of suspected circadian peak). In contrast at 3 weeks 42% demonstrated elevated AM samples. At this time point (3-weeks) median rank for AM samples was significantly higher than PM (0.19) [Z = -2.837, p < 0.005]. At 1-month post C/RT AM samples were not statistically higher than PM, despite 50% descriptively demonstrating a more elevated AM pattern at 1-month post treatment (Figures 2, Figures 3, Figures 4 and Figure 5).
This study has confirmed that the collection of saliva and analysis of salivary cortisol in patients with HNC across the three study time points was both feasible and informative. Even when considering reported attrition rates in similar behavioral research for this population (15 -20%), [23] more than 50% of our subjects were able to complete the salivary collection protocol with little difficulty.
The HPA axis is considered highly responsive. Moreover, salivary cortisol measurements are considered robust enough that both circadian effects and stressor effects can be identified in patient populations [1,16]. In general, healthy salivary cortisol rhythms are illustrated through high morning and low evening levels, while dysregulated cortisol rhythms show more flattened or erratic daytime profiles [4,16]. Considering this, our study showed both stressor and flattened/dysregulated slopes in 75% (6) of our HNC subjects. Our results indicated that the magnitude of difference between AM and PM cortisol levels was irregular with only the one-month comparison demonstrating evidence of normal circadian rhythms. Our results suggest that circadian cortisol levels were in this group were initially dysregulated at baseline, prior to the start of C/RT treatment, and had flattened by the 3-week timepoint. An explanation for this finding could be due to the heightened stress, fear and additional burden of appointments at baseline associated with a diagnosis of HNC. The chemoradiation treatment process for HNC is intense and can be an exhausting experience for the patient [7]. Moreover, even the diagnosis of this form of cancer can initiate significant fear and distress in patients [6,8,21]. This could help explain the dysregulated cortisol we experienced at baseline.
The use of salivary cortisol sampling to measure and monitor “distress” from HNC treatment offers major advantages to the field. It is reliable, replicable, and non-invasively sampled, and can be self-collected by patients at their homes or in the clinic. It does not involve subjective interpretation by clinicians, or the interpretation of “self-perceived” biobehavioral impacts which often plague current tools used for measuring depression, anxiety or fear. Altered cortisol slopes have been linked to poor prognosis and lowered survival in several other cancer groups [24-27]. These dysregulated slopes in conjunction with added psychosocial distress may disrupt other endocrine and immune responses. The inclusion of this neuroendocrine measure into regular monitoring regimes may facilitate earlier identification of normal vs mal-adaptive responses to exacting HNC interventions. Moreover, the documentation of these cortisol rhythm alterations among HNC patients may aid modulation of treatment schedules as well as predict negative prognoses.
None-the-less, the findings reported in this study should be considered exploratory. Our pilot sample was small, and we experienced some attrition across the study in salivary cortisol sample completion. While attrition rates in measurements within the HNC research are generally ascribed to disease and treatment burden along with side-effects such as dryness of mouth that could interfere with study protocol compliance [7,21]. A study by Edmonds et. al. investigated the issues with adherence and compliance in HNC patients undergoing C/RT [28] and reported various physical, psychological, and psychosocial factors impacting compliance. Similar findings could not be supported in our work. Loss to follow up at 3 weeks during treatment in our study could not be directly explained by physical side-effects of C/RT such as dryness of mouth. This may have been a direct result of our study design which attempted to account for xerostomia, requiring saliva collection at 3 weeks into treatment and not at end of treatment. Moreover, in descriptively reviewing our subjects reported morbidity profiles, weight loss, mucositis, presence of PEG tube, reported oral dryness and saliva change did not appear to affect the saliva collection for these persons. It seems that in this small group attrition may be related to other factors not directly measured.
Despite these weaknesses we believe our study offers novel information to direct future work and reveals that saliva collection for cortisol is both practical and informative for HNC populations. Our study successfully sampled diurnal salivary cortisol from subjects with varying severity of HNC diagnosis and from those undergoing both IMRT and CRT. Moreover, cortisol secreted in a diurnal pattern was routinely collected by our subjects, through multiple samples taken at a similar time of day. The majority of our subjects provided 3x daily samples consistently and followed explicit instructions for the avoidance of interfering substances/behaviors, despite the ongoing burden of curative intent HNC treatment. In this way our study supports the feasibility of clinical salivary cortisol collection in HNC patients. Although cortisol levels alone are not diagnostic, and patients may have day to day variations, we feel that the inclusion of salivary cortisol as a monitor for distress in addition to patient self-reported measures may facilitate improved identification of patients in need of support. Future studies should incorporate larger study samples and a range of surveillance points to explore multivariate relationships more fully between the timing of diurnal cortisol change and the severity of HNC psycho-behavioral morbidities.
In summary, this investigation has provided data supporting the feasibility and potential value of salivary cortisol collection and monitoring in HNC. In this sample salivary cortisol demonstrated both diurnal abnormality at baseline and flattened aberrant patterns at 3 weeks into treatment. Results suggest that noninvasive sampling of salivary cortisol may be a simple and useful marker by which to monitor distress related impacts from diagnosis of cancer and treatment burden in HNC patients.