Diabetes mellitus (DM), a cluster of metabolic diseases, resulting in high blood glucose levels, is prevalent in today's world. The global costs of diabetes and its consequences are rising and are expected substantially increase by 2030, especially in middle- and lower-income countries. Evidence-based therapies, specifically targeting the reduction of high blood glucose levels, and minimizing diabetic complications, are currently the choice of treatment. Stem cell therapy offers a promising vision to treat DM. Although challenges are still posed with this line of therapy, studies have produced regenerative beta-cells which closely resemble insulin-secreting cells. A number of sources for stem cells have been explored, ever since the proof-of-concept for cell therapy was laid down. This review summarizes stem cell therapy in the treatment of DM.
Stem cells, Diabetes, Islets, Pancreas
According to the WHO report released in November 2017 the number of people with Diabetes mellitus (DM) has risen from 108 million in 1980 to 422 million in 2014 [1]. The prevalence of DM across all age-groups, worldwide was estimated to be 2.8% in 2000 and is expected to rise to 4.4% by 2030, with the total number of people with DM projected to rise from 171 million in 2000, to 636 million in 2030 [2].
There are two types of DM - type 1 and type 2. Type 1 diabetes, also known as insulin-dependent diabetes and juvenile diabetes, involves the immune system, which results from a cellular-mediated autoimmune destruction of the BETA-cells of the pancreas. It can occur at any age, but most often occurs in children and young adults. The etiology of DM type 1 is not fully uncovered, however in most cases, the body's immune system attacks and destroys insulin producing beta-cells. Family history is known to play a role, in about 10% to 15% of people with DM type 1. Type 2 diabetes, also known as adult-onset DM, which usually develops after the age of 40 but can appear earlier in obese patients. With DM type 2, the pancreas produces insulin, but the body cannot use it effectively. Insulin treatment is not always necessary, in these patients, as with DM type 1. Diabetes mellitus is associated with severe long-term micro- and macrovascular complications and carries a high rate of morbidity and mortality. Both DM type 1 and 2 are a significant public health concern with numerous debilitating complications. The global costs of diabetes and its consequences are rising and are expected substantially increase by 2030 [1], especially in middle- and lower-income countries. Evidence-based therapies, specifically targeting the reduction of high blood glucose levels, and minimizing diabetic complications, are currently the choice of treatment [3].
The pathogenesis of DM, whether type 1 or type 2, can be traced back to the dysfunction of the pancreatic beta-cells. Although approved therapies that enhance beta-cell function exist, there are none that lead to the regeneration of the lost or dysfunctional beta-cell [4]. Studies have shown that beta-cells can be reprogrammed, with certain molecules, such as GABA [5] and hormone [6]. However, these studies also bring to light a lot of uncertainties that need to be explored [4]. Stem cells for the treatment of DM come from a variety of biological sources like embryos, placenta, and bone marrow. Progenitor cells are another exciting avenue of research. Like stem cells, these cells can take on the form of a number of different types of mature human cells, however, unlike stem cells, they cannot divide indefinitely.
Progenitor stem cells have been used to grow insulin producing cells, under laboratory conditions, from intestinal cells and undeveloped pancreatic cells [7]. This article provides an overview of the various approaches used to regenerate pancreas in patients with diabetes, recent advances including our contributions and also a novel approach that may be explored in future.
Progenitor cell are very similar to stem cells. They are biological cells and like stem cells, they too have the ability to differentiate into a specific type of cell. However, they are already more specific than stem cells and can only be pushed to differentiate into its "target" cell. Whereas, stem cells have the ability to differentiate into many different types of cells as shown in Figure 1. Comparison between Stem Cell and Progenitor Cell are highlighted in detail in Table 1.
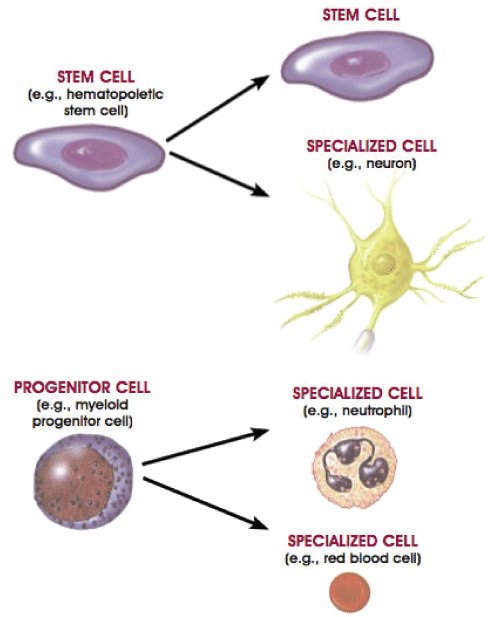 Figure 1: Formation of differentiated cells and specialized cells from stem cells and progenitor cells.
View Figure 1
Figure 1: Formation of differentiated cells and specialized cells from stem cells and progenitor cells.
View Figure 1
Table 1: Comparison between Stem Cell and Progenitor Cell. View Table 1
Pancreatic islets include the insulin producing beta cells, which are crucial in regulating blood glucose levels. Islet transplants are a safer option than whole pancreas transplants. The procedure to insert donor islet cells is far less critical than transplanting a complete pancreas. For any type of transplantation procedure, a balance is sought between efficacy and toxicity. With respect to islet transplantation a main concern was that many of the current agents' cause damage beta cells or induce peripheral insulin resistance [8,9]. Immunosuppressant drugs also can cause problems, as suppressing the immune system raises the risk of infection [10].
Shapiro, et al., reported insulin independence, with tight glycemic control and correction of glycated hemoglobin levels, in seven consecutive subjects treated with glucocorticoid-free immunosuppressive therapy combined with infusion of an adequate mass of freshly prepared islets from two or more pancreases from deceased donors [11]. This treatment came to be known as the Edmonton Protocol [12]. In continuation to this protocol, a phase I/II clinical trial was undertaken to demonstrate the feasibility and reproducibility of the outcomes of the Edmonton protocol. The trial concluded that though long-term endogenous insulin production and glycemic stability in subjects with type 1 diabetes mellitus was achieved, it was found that insulin independence was more than often not sustainable, and gradually lost in the long run [13].
Future studies/trials should focus on enhanced islet engraftment, less toxic immunosuppressive therapy, reduced metabolic stress, reduced apoptosis, enhanced regeneration, the use of living donors, and the induction of immunologic tolerance. This approach will ensure improved success rates in transplantation and sustained insulin independence [13]. In 2017, Westenfelder, et al., [14] reiterated the need for re-establishing endogenous insulin secretion without being limited by both the scarcity of organ donors and the life-long need for often-toxic antirejection drugs. He and his team argued that intrahepatic islet transplants were inefficient, due to the high number of donors required per treatment and was also associated with high early losses of islets [15]. They hypothesized that high numbers of mesenchymal stem cells (MSCs) in neo-islets (NIs) would enable islet cells to survive and re-differentiate into normally functioning endocrine cells.
This treatment led to long-term glycemic control in non-obese diabetic mice [14]. The NIs survived, engrafted and re-differentiated into functional insulin secreting cells in the well-vascularized omentum (via intraperitoneal administration), delivering insulin into the hepatic portal system. Simultaneously, re-expression of other islet-specific hormones occurred. Identical injection of Nis into nondiabetic animals resulted in omental engraftment without causing hypoglycemia, further demonstrating regulated islet hormone secretion [14]. Both allo- and auto-immune protection was also achieved [16,17]. In preparation for a pilot study in pet dogs with DM type 1, streptozotocin-diabetic nonobese diabetic/severe combined immunodeficiency (NOD/SCID) mice were treated in a similar manner with canine NIs (cNIs). In these, euglycemia was readily and durably induced and intraperitoneal Glucose Tolerance Tests (i.p. GTTs) were normalized by the exclusive release of canine-specific insulin [14].
Ongoing studies regarding this NI technology are focused on analogous studies using human NIs in diabetic NOD/SCID mice, as well as on the characterization of the NI-intrinsic microcirculation post-engraftment in the omentum, the long-term distribution of MSCs within the NIs in vivo, their potential differentiation into insulin-producing and vascular endothelial cells, the re-differentiation of alpha and other endocrine cells in vivo, in situ IDO (canine) and iNOS (murine) expression by MSCs, and a detailed analysis of the long-term histology and cell composition of functioning NIs [14]. In patients with DM type 1, glycemic control can also be achieved with intensive insulin therapy and pancreatic transplantation. Intensive insulin therapy does not normalize glycosylated hemoglobin values and may cause severe hypoglycemia. Pancreatic transplantation provides excellent glycemic control, and although the outcome of the procedure has improved dramatically over the past decade, it remains an invasive procedure with a substantial risk of morbidity. The findings indicated that islet transplantation alone is associated with a minimal risk and results in good metabolic control, with normalization of glycosylated hemoglobin values and sustained freedom from the need for exogenous insulin [18,19].
Autologous bone marrow contains hematopoietic stem cells, a mixture of mononuclear cells, a few mesenchymal cells, and other cells. Peripheral blood stem cells are mainly selected by their CD34 antigen positivity. Different preparations of the hematopoietic cells have been claimed to be effective in correcting hyperglycaemia, improvement of endogenous insulin production, and diminishing or eliminating the need for insulin and other diabetes controlling treatments [19-21]. Wang, et al., [22] used autologous bone marrow to treat 31 patients with stem cell infusion into the major arteries feeding the pancreas. The HbA1c dropped by > 1.5% within 30 days and the C-peptide increased at the 3-month follow-up mark. All patients were reported to have had a significant reduction of their anti-diabetic medications [23].
Bone marrow-Mononuclear Cells (BM-MNC) were used by Bhansali, et al., [19] in a prospective, randomised, placebo-controlled trial designed to treat 11 patients. Nine of the 11 patients (82%) achieved 50% reduction of the insulin requirements and 10 (91%) achieved a HbA1c < 7% in the intervention group.
These two recent meta-analysis of published trials concluded that both BM-MNC and peripheral blood mononuclear cell infusion may result in improvement of the HbA1C, fasting plasma glucose, C-peptide levels, and endogenous insulin production at 12 months in the majority of treated patients.
Embryonic Stem cells can differentiate in vitro and in vivo, to form a wide range of specialized cell types. Taken from the embryo at the blastocyst stage, ES cells are pluripotent. Their versatility is an asset over adult stem cells, but also a challenge. While ES cells can become insulin-secreting cells in culture, e.g., the cells are not as stable as adult stem cells. ES cells studied in vitro and in vivo can differentiate into tumor cells. Similarly, the rapid proliferation rate of ES cells, which is also greater than that of adult cells, carries greater risk of forming tumors in vivo [24-26]. The discovery of methods to isolate and grow human-Embryonic Stem Cells (ESCs) in 1998 renewed the hopes of researchers, clinicians and diabetes patients and their families that a cure for DM TYPE 1 and perhaps non-DM TYPE 1 as well may be within striking distance. In theory, ESC could be cultivated and coaxed into developing the insulin-producing islet cells of the pancreas. With a ready supply of cultured stem cells at hand, the theory is that a line of ESC could be grown up as needed, for anyone requiring a transplant.
The list of sources of stem cells advantages and disadvantages are cited in the Table 2.
Table 2: List of sources of stem cells. Their advantages and disadvantages. View Table 2
 Figure 2: Showing Stem cells for IDDM.
View Figure 2
Figure 2: Showing Stem cells for IDDM.
View Figure 2
1) It provides medical benefits in the fields of therapeutic cloning and regenerative medicine.
2) It provides great potential for discovering treatments and cures to a variety of diseases including Parkinson's disease, schizophrenia, Alzheimer's disease, cancer, spinal cord injuries, diabetes and many more.
3) Limbs and organs could be grown in a lab from stem cells and then used in transplants or to help treat illnesses.
4) It will help scientists to learn about human growth and cell development.
5) Scientists and doctors will be able to test millions of potential drugs and medicine, without the use of animals or human testers. This necessitates a process of simulating the effect the drug has on a specific population of cells. This would tell if the drug is useful or has any problems.
6) Stem cell research also benefits the study of development stages that cannot be studied directly in a human embryo, which sometimes are linked with major clinical consequences such as birth defects, pregnancy-loss and infertility. A more comprehensive understanding of normal development will ultimately allow the prevention or treatment of abnormal human development.
7) Stem cell research also benefits the study of development stages that cannot be studied directly in a human embryo, which sometimes are linked with major clinical consequences such as birth defects, pregnancy-loss and infertility. A more comprehensive understanding of normal development will ultimately allow the prevention or treatment of abnormal human development.
8) An advantage of the usage of adult stem cells to treat disease is that a patient's own cells could be used to treat a patient. Risks would be quite reduced because patients' bodies would not reject their own cells.
9) Embryonic stem cells can develop into any cell types of the body, and may then be more versatile than adult stem cells.
1) The use of embryonic stem cells for research involves the destruction of blastocysts formed from laboratory-fertilized human eggs. For those people who believe that life begins at conception, the blastocyst is a human life and to destroy it is immoral and unacceptable.
2) Like any other new technology, it is also completely unknown what the long-term effects of such an interference with nature could materialize.
3) Embryonic stem cells may not be the solution for all ailments.
4) According to a new research, stem cell therapy was used on heart disease patients. It was found that it can make their coronary arteries narrower.
5) A disadvantage of most adult stem cells is that they are pre-specialized, for instance, blood stem cells make only blood, and brain stem cells make only brain cells.
6) These are derived from embryos that are not a patient's own and the patient's body may reject them.
Pluripotent stem cells (PSCs) have the ability to self-renew and differentiate into three germ layers including ectoderm, endoderm and mesoderm, and hence can play an important role in regenerative medicine and cell therapy. PSCs are obtained from the inner cell mass of blastocyst (embryonic stem cells, ES) or from the foetal genital ridge (embryonic germ cells, EG). Human ES cell lines were first reported in 1998 by Prof. Thomson and his group [27] whereas human EG cell lines were reported by Prof. Shamblott in the same year [28].
Technology also exists to derive PSCs from adult somatic cells by reprogramming them to embryonic state using a cocktail of factors (induced pluripotent stem cells, iPS) or by allowing factors present in the oocyte cytoplasm to reprogramme somatic cells (therapeutic cloning). Prof. Yamanaka shared the Nobel Prize for Medicine in 2012 for iPS technology [29] whereas Prof. Mitalipov's group in 2013 [30] was the first to derive human ES cell line by somatic cell nuclear transfer (SCNT). Jiang, et al., [31] observed that 30% of transplanted mice showed reduction in hyperglycaemia on transplanting insulin positive cells, obtained by differentiating ES cells, for over a period of six months. Thus, proof of concept for use of human ES cells for diabetes was established; however, the process remains highly inefficient. Schulz, et al., [32] developed a scalable system for producing functional progenitors and Bruin, et al., [33] improved the differentiation protocol further which resulted in grafts containing > 80% endocrine cells and resulted in single hormonal cells expressing either insulin or glucagon or somatostatin in contrast to earlier polyhormonal cells. Kirk, et al., [34] demonstrated that human insulin was secreted by the seventh week after transplantation, of encapsulated pancreatic progenitors and by week 20 enough human insulin was produced to ameliorate alloxan-induced diabetic symptoms.
Mesenchymal stem cells (MSCs) are self-renewing multipotent cells that have the capacity to secrete multiple biologic factors that can restore and repair injured tissues. Preclinical and clinical evidence have substantiated the therapeutic benefit of MSCs in various medical conditions. Currently, MSCs are the most commonly used cell-based therapy in clinical trials because of their regenerative effects, ease of isolation and low immunogenicity. Experimental and clinical studies have provided promising results using MSCs to treat diabetes.
In 2015, investigators from Sweden were the first to evaluate the safety and efficacy of autologous MSC treatment in newly diagnosed DM type 1. Stem cells were harvested from the iliac crest bone marrow and the median systemic single dose was 2.75 × 106 cells/kg. They concluded that administration of MSCs did not result in adverse events in any of the ten patients and provided promising C-peptide concentrations at the 1-year follow-up. This phase I trial did not show any functional differences between the control and MSC group in hemoglobin A1c (HbA1c) or insulin dose.
Hu and coworkers conducted a single-center, double-blind study examining the safety, feasibility and preliminary outcomes of umbilical cord Wharton's jelly-derived MSCs for new-onset type I diabetics [35]. The MSC-treated group underwent two intravenous infusions (mean cell count of 2.6 × 107) separated 4 weeks apart. Postprandial glucose and HbA1c measurements were lower in the experimental cohort between 9 and 24 months after MSC infusion. Also, insulin usage and fasting C-peptide were significantly improved in the MSC group. The study authors concluded that in their small study, not powered to detect functional differences, the transplant of umbilical cord MSCs is feasible and safe. A pilot study in China involving delivering placenta-derived MSCs to patients with long-standing DM type 2 revealed the transplantation was safe, easy and potentially efficacious [36]. This investigation included ten patients with type 2 diabetes for a duration ≥ 3 years, insulin dependent (≥ 0.7 U/kg/day) for at least 1 year and poorly controlled glucose. The subjects received on average 1.35 × 106/kg placental stem cells on three separate occasions with 1-month intervals between intravenous infusions. Six months after treatment, the insulin dosage and HbA1c measurements for all the patients improved. Moreover, C-peptide and insulin release were also higher after MSC treatment. In addition, this study included a group of individuals that translate closer to actual clinical scenarios, as they also had other comorbidities, including heart disease, kidney disease and vascular complications. Lately, researchers have developed insulin-secreting MSCs and delivered them, in combination with hematopoietic stem cells, to patients with DM type I [37].
Both DM type 1 and type 2 are among the most amenable diseases for treatment. Functional restoration of existing beta-cells, transplantation of stem cells or stem cell-derived beta-like cells might provide new opportunities for treatment (Figure 3). However, the use of stem cells to generate a renewable source of beta-cells for diabetes treatment remains challenging, largely due to safety concerns. There has been a large number of small published studies that indeed do not constitute a solid scientific proof of the efficacy of different stem cells being tried. The introduction of pre-prepared or frozen cells, like the MSC of umbilical or bone marrow origin, by different pharmaceutical companies has proven extremely expensive at this point and definitely out of reach to the vast majority of individuals. Larger studies are needed to advance the field and understand the best way to realise its potential. We believe stem cell therapy should only be used within clinical trials at this time, until enough evaluable data becomes available. The laboratory methods, like culture conditions and methods of cell numbering, have to be better thought of and more uniformly standardized, and the interpretation of the results should be done critically. In summary, regenerative medicine remains a new and exciting field of research that holds much promise into the treatment of patients with endocronologic diseases of all ages. Evidence based clinical treatment of diabetic symptoms only adds to the disease burden. With the advent of stem cell therapy, the potential to eradicate diabetes seems to be on the horizon.
 Figure 3: Current therapies for Diabetes Mellitus.
View Figure 3
Figure 3: Current therapies for Diabetes Mellitus.
View Figure 3