Skin and soft tissue infections (SSTIs) with Nontuberculous Mycobacteria (NTMs) are increasingly encountered in recent times. We describe a case of a chronic non-healing ulcer on the foot of a young immunocompetent lady caused by Mycobacterium simiae. The diagnostic approach and successful management of the case have been reviewed. This is probably the first case report SSTI due to M. simiae from India.
A 28-year-old well nourished, healthy and educated lady (employee of a company), resident of Guntur, Andhra Pradesh, India, presented to the general Surgery out-patient clinic of Kamineni Hospitals, Hyderabad, Telangana State. She complained of a chronic non-healing ulcer, over the past 6 months, on the dorsum of the right foot. The lesion started as a small nodule that increased in size and later ulcerated. The patient noticed serosanguinous discharge from the ulcer. However, she had very minimal pain at the site of the ulcer and had no problem in walking. She did not give any history of trauma, surgery, insect or animal bite, cosmetic treatment for the feet, exposure to sea water or swimming pool water nor travel to other countries.
On examination, the ulcer was an open moist, 3-4 cm lesion on the dorsum of the right foot below the last toe (Figure 1). The edges were ragged, hard and indurated. The lesion was painful on pressure. A serosanguinous exudate was seen oozing out of the ulcer. There was no foul smell. The lesion was free from the underlying bone. And there was no wasting of the foot muscles. With a clinical impression of a chronic non-healing infected ulcer, the lesion was debrided extensively. The material was submitted for further analysis to histopathology and microbiology.
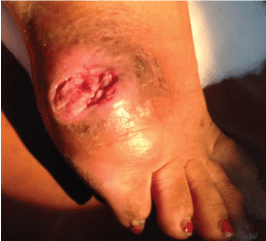 Figure 1: Chronic ulcer, dorsum of right foot.
View Figure 1
Figure 1: Chronic ulcer, dorsum of right foot.
View Figure 1
The histopathology of the material was reported as non-specific granulomatous inflammation with no organisms seen.
Gram's stain and bacteriological cultures were performed on the material. Gram's stain showed plenty of polymorphs and few thin, long Gram-negative bacilli. Ziehl-Neelsen stain (ZN) stain of the specimen was negative for Acid fast bacilli. A modified acid-fast stain with 1% acid was negative for Nocardia.
Specimen was inoculated on Blood agar and MacConkey agar and incubated at 37 ºC overnight. A heavy growth of Pseudomonas aeruginosa both on the 5% sheep Blood agar and MacConkey agar plates was observed the next day. This isolate was considered as a surface colonizer of the ulcer and was managed with topical polymyxin lotion.
Mycobacterial culture was performed after the material was decontaminated as per recommended procedure [1] and inoculated on a Lowenstein Jensen slant (LJ) (BD, India) and incubated at 37 ºC. The specimen was simultaneously inoculated in a Mycobacterial Growth Indicator tube (MGIT) (BD, USA) and incubated in the Bactec 320 automated system for Mycobacterial culture and susceptibility (BD, USA). The MGIT flagged positive by 3rd day of incubation. ZN stain of the MGIT broth showed Acid fast bacilli. However, there was no growth on the LJ slant even after 4 weeks of incubation.
The MGIT broth was sub-cultured on Blood agar and MacConkey agar and incubated at 37 ºC. After 2 days of incubation, very minute non-hemolytic colonies were seen to grow on the blood agar. While on the MacConkey agar, very minute pale colonies were seen after 2 days. Growth from both the plates was positive for acid fast bacilli by the ZN stain.
A TBc Identification Test (TBc ID) (BD, USA) (A rapid "card" test-differentiates M. Tb complex from Non TB mycobacterial species based upon detection of MPT64, a Mycobacterial protein secreted by M. Tb cells during culture) was performed from the colonies on blood agar. The result was interpreted as negative for M. Tb. Specimen was negative for M. Tb on the Gene expert MTB/RIF system (Cepheid, Sunnyvale, USA). With all these features, the isolate was considered to be an atypical Mycobacterium and the treatment was initiated. An extensive debridement of the ulcer was performed. She was advised oral Clarithromycin 150 mg twice daily and Intramuscular (I/M) Amikacin 500 mg twice daily for 2 months. The lesion healed completely with no recurrence (Figure 2) after 2 months.
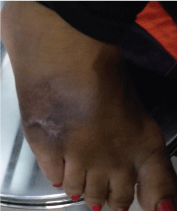 Figure 2: Healed ulcer, dorsum of right foot.
View Figure 2
Figure 2: Healed ulcer, dorsum of right foot.
View Figure 2
In view of the clinical presentation, the isolate was considered either to be M. ulcerans or M. abscessus. For further confirmation of the NTM isolate, it was sent for analysis by MALDI-TOF at GYD Diagnostics, Hyderabad, India. The isolate was also sent to the National Tuberculosis Institute (NTI), Bangalore, India, for HPLC analysis and identification at their national reference centre for NTMs. The isolate was identified as M. simiae by both the methods (Figure 3 and Figure 4).
 Figure 3: Isolate identified as M. simiae on MALDI TOF.
View Figure 3
Figure 3: Isolate identified as M. simiae on MALDI TOF.
View Figure 3
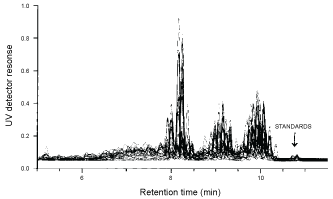 Figure 4: Isolate identified as M. simiae on HPLC.
View Figure 4
Figure 4: Isolate identified as M. simiae on HPLC.
View Figure 4
Mycobacteria related to M. simiae constitute the largest group or complex within the genus Mycobacterium [2]. M. simiae was first identified in Macaca rhesus monkeys by Karasseva in 1965 [2]. The first isolation from humans was in 1971. Since then it is an increasingly frequent clinical isolate in certain regions of the world, such as Israel, Cuba, Southern Texas. There were 400 reported cases of M. simiae isolated from clinical sites associated with a wide range of pathogenicity [2]. M. simiae is a ubiquitous organism with huge environmental reservoirs, such as natural and municipal water, soil, aerosols, besides animals and humans [3]. The M. simiae complex is comprised of 17 officially recognized species that are phylogenetically related and include M. triplex, M. genavense, M. heidelbergense, M. lentiflavum [4]. M. simiae, is the most common species associated with human pathology. M. simiae can be cultured on Loẅenstein-Jensen or Middle-brook media and typically requires 4 to 6 weeks to grow. It is a slow-growing photo-chromogen [3]. The colony appears rust-coloured after exposure to light. It is the only NTM that is niacin positive [2,3]. Though the M. simiae isolate, in our patient, was a slow grower in Middle-brook medium, it failed to grow on LJ medium, even after 6 weeks of incubation at 37 ºC. Hence, the photochromogenic nature of the isolate could not be assessed [5].
Since M. simiae is commonly found in nature, mainly in the aquatic environment, its role as a pathogen has been controversial [3]. Clinically significant disease occurs in 20% of patients. It can infect immunosuppressed and rarely immunocompetent patients. Most infections are associated with HIV infected patients, with disseminated respiratory and reticuloendothelial system involvement [3]. It is commonly isolated from respiratory specimen and considered as a colonizer especially in immunocompetent patients, [3]. Iatrogenic skin infection and lymphadenitis caused by M. simiae in immunocompetent person, were reported [3,6]. Other reported cases, of M. simiae infection include nosocomial pseudo-out-break suspected to have originated from a contaminated hospital water supply, central line related blood stream infection especially in immunosuppressed patients [3]. However, no cases were reported from India [7-9].
Since infection with M. simiae is uncommon, any case with M. simiae isolation should be defined in association with the histopathology features [2]. The present case, can be defined as definitely clinically significant as isolation of M. simiae is supported by histopathologic evidence of infection. And the alternate diagnostic modalities also confirmed the isolate as M. simiae.
SSTIs usually occur after a traumatic injury, surgery or cosmetic procedures, which may expose a wound to soil, water, or medical devices occasionally contaminated with environmental organisms including NTMs [9]. The diagnosis is challenging due to diverse and atypical clinical presentation, low yield from cultured specimens and nonspecific histopathology on tissue biopsy [10]. A high index of suspicion of NTM infection, especially with persistent non healing ulcers, is essential. Granulomatous inflammations are the most common presentation of infection by NTMs [8]. The lesion usually appears as a small superficial nodular lesion that later causes severe ulcerative skin disorder, as was seen in our patient. The gold standard for diagnosis is biopsy of the ulcer and culture for Mycobacteria [3]. The newer diagnostics such as the High-Performance Liquid Chromatography (HPLC) and Matrix Assisted-Laser Desorption/Ionization (MALDI TOF), have improved the definitive identification of the mycobacterial species, especially the NTMs [10,11]. The mycolic acid profile via HPLC enables discrimination between M. simiae, M. tuberculosis and M. avium complex [3,10]. The widespread adoption of MALDI-TOF by routine laboratories has potential to simplify and expedite the laboratory identification of NTMs [11,12].
SSTIs caused by nontuberculous mycobacteria (NTM) are generally underrecognized and difficult to treat [12]. The NTMs are usually resistant to the anti-tuberculous drugs. Agents reported to have activity against members of the M. simiae complex include clarithromycin, ethambutol, ethionamide, fluoroquinolones, amikacin, and cyclomerize [3]. Extensive surgical debridement along with aggressive medical treatment with Clarithromycin and Amikacin, resulted in a remarkable healing of the ulcer in the present case.
NTM skin and soft tissue infections are frequently underdiagnosed, mainly due to failure to perform relevant cultures [12]. A high index of suspicion of NTM infection, especially with persistent non-healing ulcers, is essential. A good communication between the clinician and microbiology is essential to diagnose these cases and manage successfully.