Trypanosoma brucei brucei in mice, the experimental model of African Trypanosomiasis, is known to begin crossing the blood-brain barrier between 9-13 days post-infection, which is also considered the commencement of the late stage of the disease. A few recent reports however, reported traversal as early as two hours to a few days. In these reports laboratory strains were used thus this study sought to find out if this early brain invasion is phenomenon of laboratory strains or also field isolates.
In this study the blood-brain barrier traversal of three Trypanosoma Brucei Brucei Strains: A field strain, a laboratory strain and an uncharacterized strain, was compared. Three groups of mice were infected with each strain respectively, and then treated with Diminazene aceturate at 3, 6 and 9 days post-infection. When no parasites could be seen in the blood, mice brains were excised out, homogenized in Phosphate Saline Glucose and used to re-infect new groups of mice. The infectivity of the brain homogenate was used to determine the traversal time-point.
The data showed that all strains traversed the blood brain barrier much earlier than what was previously reported, with the earliest traversal time point being 3 days post-infection.
Taken together these data indicate that early blood-brain barrier traversal is not just a phenomenon of laboratory strains but all strains and may have an implication on the control of African Trypanosomiasis.
Trypanosoma brucei brucei, Early traversal, Blood brain barrier traversal, Laboratory strains, Field isolates
Trypanosoma brucei causes a zoonotic disease called African Trypanosomiasis. African trypanosomiasis causes huge economic losses since it affects animal production and negatively impacts the livelihood of people in this area. The disease in humans, called Human African Trypanosomiasis (HAT) or Sleeping sickness is caused by Trypanosoma brucei gambiense (T.b.g) in West and Central Africa or Trypanosoma brucei rhodesience (T.b.r) in East Africa [1] with Uganda being the only country affected by both subspecies [2].
Classically, HAT is known to have two stages; the early hemolymphatic stage whereby parasites are confined to the blood and lymphatics and the second stage (meningoecenphalitic stage) where an increased number of trypanosomes and lymphocytes accumulate in the brain parenchyma [3,4]. The second stage which is the most severe and difficult to treat, is characterized by a number of neurological syndromes such as severe headaches, disruption of the sleep pattern characterized by disturbances of consciousness and of the circadian rhythm of sleep, represented typically by diurnal somnolence and nocturnal insomnia [5,6], progressive mental deterioration, psychiatric manifestations, tremors, finally culminating in coma and death if untreated [7]. In animals Trypansomoma brucei brucei (T.b.b) causes a disease in cattle similar to that in humans [8-10] characterized by the acute phase where parasites are seen in the blood and the chronic stage where parasites disappear from blood but are present in the brain parenchyma [11]. In the chronic stage cattle have been seen moving around in circles indicating a defect in their nervoussystem (Ann Nanteza, COVAB,Makerere University, Kampala Uganda, personal communication).
The most effective available way of controlling the disease is chemotherapy. In humans few drugs are registered for treating the disease according to the stage and the type of the disease. For the first stage, suramin and pentamidine are used for treatment againstT.b.randT.b.ginfection respectively [6]. These are not used for the treatment of the second stage due to their limited ability to traverse the blood brain barrier. Melasoprol is the most effective drug for bothT.b.randT.b.gstage two infections but has very terrible side effects with 5-10% of the patients dying due to post-treatment reactive encephalopathy [12]. A combination of eflornithine and nirfutimox was introduced, which is cheaper, but it is only effective againstT.b.ginfection [13]. Recent studies have reported the emergence of resistance to both eflornithine and melarsoprol making the search for novel drugs even more urgent.
However to be able to develop new interventions, understanding the timing of brain invasion and the mechanism by which Trypanosoma brucei invades the brain parenchyma and leads to the development of the central nervous disease is key. A number of mouse models have been used to study the development of the central nervous disease as well as examine the mechanism by which the trypanosome crosses the blood brain barrier [4,14]. Two of the earliest studies [4,14] using immunofluorescence assay, whereby anti-GLUT-1 antibodies and anti-Trypanosoma brucei brucei VSG antibodies were used to visualize the small blood vessels and trypanosomes respectively, showed that Trypanosoma brucei brucei (T.b.b) begins to cross the blood brain barrier around 13 days post-infection (dpi) in mice and rats. Following these studies other studies studying different aspects of the development of the stage two disease and using advanced techniques also reportred on the timing of brain parenchyma invasion which they found to commence around 9-13 dpi [15-17].
A few other reports however, have shown that laboratory strains are able to invade the brain parenchyma as early as a few hours to 4 days post-infection (dpi) [18,19]. In these studies different techniques were used to determine invasion of the brain parenchyma by T. brucei. In the study done by Sanderson and colleagues, mice were infected with T.b.b GVR35 and the development of parasitemia monitored. At peak parasitemia, mice were treated with Berenil (Diminazene aceturate) and parasitemia further monitored. After 7 days post-treatment when parasites could no longer be seen in the blood, these animals were sacrificed, their brains excised and homogenized in 1 ml of Phosphate Saline Glucose (PSG). The homogenate was used to re-infect a new batch of mice. Their experiments showed that parasites begin to penetrate the brain parenchyma as early as four days post-infection. Diminazene aceturate has been proven to effectively clear the parasites from the blood even in areas in the devoid of the blood brain barrier [17]. Thus when mice are treated with this drug the ony parasites that persist in the brain are those that have exited the blood vessels and crossed the blood brain barrier. In another study, using a mouse model and intravital imaging, Frevert and colleagues [18] showed that trypanosomes penetrate the brain parenchyma within hours of infection. In the latter study intravenous and intraperitonealy routes were used to infect mice with relatively high doses of parasites. It is also possible that the route of infection used could have influenced the timing of traversal. Having trypanosomes traversing the blood brain barrier within hours of infection would have a big implication on the control or management of African Trypanosomiasis. This would imply that the treatment regime would have to be changed once this finding is confirmed for field strains via further investigation with a number of different Trypanosoma brucei brucei (T.b.b) strains. Most studies on the development of the CNS disease in micehave been done with laboratory strains. No work has been done with field isolates. In the present study therefore we compared brain parenchyma invasion by a field isolate and an uncharacterized isolate, with that of a laboratory strain with the aim of substantiating or determining whether if early brain invasion is a phenomenon of laboratory strains or a common phenomenon for field isolates as well.
In this study, one field isolates, T.b.b APC098, an uncharacterized field isolate (uncharacterized in the sense that the date of its isolation is not clear), T.b.b 729X and one laboratory strain T.b.b GVR35 were used. T.b.b APC098 was isolated on 31st July 2010 from a cow in Apach District, Uganda, by a team from our lab and was passaged about five times in mice prior to this study. The date and place from which T.b.b 729X was isolated from is not clear. T.b.b GVR35 is a laboratory strain originally isolated from a wildebeest, in Tanzania in 1966 [20]. This strain has been cultured in vitro and passaged in mice numerous times. To confirm that the isolates were indeed T.b.b species, ITS, SRA and TgsGP PCRs were carried out on them. Mice were infected with a stabilate of each of the field strains. At peak parasitemia, the mice were anesthetized and blood drawn from the heart by cardiac puncture. Two hundred microlitres of this infected blood was used for extraction of DNA using the Zymogen genomic DNA extraction kit. ITS, SRA and TgsGP PCRs were then performed. All strains were confirmed to be Trypanosoma Brucei Brucei Strains on the basis of band size on Agarose gel electrophoresis where a band of about 480 bp was obtained for ITS PCR and no band was obtained for SRA or TgsGP PCR for both isolates.
To determine the onset of blood brain barrier traversal a method used by [19] Sanderon, et al. 2008 with modification was used. Briefly, for each strain, nine 6-8 weeks-old Swiss Webster mice were infected intramuscularly with 3 × 103 trypanosomes respectively. The infected mice were monitored for parasitemia using the Herbert and Lumsden matching method [21] and their pre-patent period, parasitemia at 6 dpi and persistence spans recorded. Three mice in each group were treated with Diminazene aceturate at 3 days post-infection (dpi), 6 dpi and 15 dpi respectively to clear parasites from the peripheral blood. To determine whether indeed parasites have been cleared, the mice were monitored for parasitemia post-treatment for 10 days. After 10 days parasites could no longer be seen in blood, thus the mice were sacrificed; their brains excised out and immediately placed in Phosphate Saline-Glucose (PSG). These were homogenized with a hand homogenizer, centrifuged at 200 g for 5 minutes and the supernatant used to re-infect two mice per group to determine which brain material contained parasites and could therefore initiate a new infection. The newly infected mice were then monitored for development of parasitemia and those which were positively infected suggested that parasites had traversed the fist level of the blood brain barrier (BBB) by the time of treatment. Diminazene aceturate has abeen shown to successfully clear parasites from blood in 7 seven days and in 2 days in the brain areas devoid of the blood brain barrier [17]. Thus when using this method one does not need to perfuse the mouse brain before homegenisation since the only parasites that will be in the brain are parasites that have exited the blood vessels and crossed the first level of the blood brain barrier. This makes this an easy and simly method for determining if parasites have crossed the blood brain barrier or not. This experiment was repeated three times and the onset of BBB traversal (infectivity of brain matter) recorded as the time (days post-infection) when mice which were initially infected were treated. In each repeat, the 'infectivity' at each time point was given a value of one (1) and the average 'infectivity' for the three experiments determined. If the 'infectivity' was greater than 0.5, it was taken to imply that trypanosomes were more likely to begin crossing the BBB at that time point.
This study was part of one of the aurthors doctoral thesis and was approved by the Scientific or Doctoral Committee. At that time the College Research Ethics committee had not yet been established that is why it was approved as such.
In this study the infectivity was used to determine the onset of blood brain barrier traversal. An infectivity greater than 0.5 was taken to imply that trypanosomes were more likely to begin crossing the blood brain barrier at that time point. The results show that at 3 dpi the 'infectivity' or 'occurrence' of infective trypanosomes in the brain parenchyma for T.b.b GVR35, T.b.b 729X and T.b.b APC098 was 0.33, 0.67 and 0 respectively (Figure 1). This implies that only T.b.b 729X was more likely to begin crossing the BBB at 3 dpi. At 6 dpi the 'infectivity' of T.b.b GVR35, and T.b.b APC098 was 0.67 while that of T.b.b 729X was 1 suggesting that the onset of BBB traversal for T.b.b GVR35 and T.b.b APC098 is 6 dpi. Thus the onset of BBB traversal for T.b.b 729X remains the earlier one (3 dpi) whereas that of T.b.b APC 098 andT.b.b GVR35 is 6 dpi.
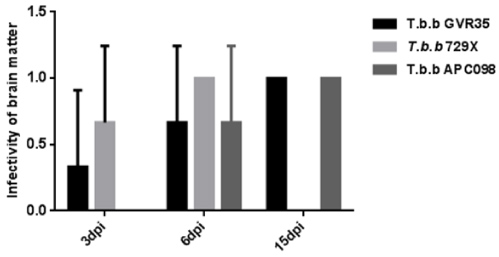 Figure 1: Infectivity of brain matter of treated mice as a measure of the timing of the onset of BBB traversal.
View Figure 1
Figure 1: Infectivity of brain matter of treated mice as a measure of the timing of the onset of BBB traversal.
View Figure 1
Previously infected mice were treated at 3, 6 and 15 dpi and infectivity of their brain matter as a measure of the likely-would of occurrence of trypanosomes in the brain at 3, 6 and 15 dpi determined and used to deduce the timing of the onset of BBB traversal for each strain. The data ± SD represent three replicate experiments.
In order to estimate the number of parasites in the brain at the timing deduced as the onset point of traversal, the pre-patent period of mice infected with the appropriate brain homogenate was determined (Table 1). Though the pre-parent period does not directly represent the number of parasites in the mouse brain it can give an indication of the number of pasites in the brain and thus can be used as an indicator of parasite load in the brain. From previous experimental studies (data not published) we observed that when parasites are too few no infection occurs but the mouse clears off the parasites. However when a high dose is given for the same strain the pre-patent period is usually shortened. Thus based on this observation the pre-patent period was used as an indicator of parasite load in the brain. In this study, the magnitude of parasites initiating the infection was assumed to be inversely proportional to the pre-patent period. The days post-infection (dpi) whereby the infectivity was found to be greater than 0.5 was taken as the earliest onset of BBB traversal for each strain. And pre-patent periods of strains were compared for infective brain matter obtained at their earliest timing of BBB traversal of the respective strain. That is 3, and 6 dpi for T.b.b 729X and T.b.b APC098 and T.b.b GVR35 respectively. There was no significant difference (P > 0.05) in pre-patent period among T.b.b 729X, T.b.b APC098 and T.b.b GVR35 (Table 1). This implies that there was more or less the same number of trypanosomes in the brain at the traversal time points deduced for each strain. Since all the strains had two time points where the infectivity was greater than 0.5, the pre-patent periods at the two time points were compared for each of the strains to substantiate the magnitude of parasites in the brain at these time points. There was no significant difference (P > 0.05) in the pre-patent period at 3 dpi and 6 dpi for T.b.b 729X and at 6 dpi and 15 dpi for both T.b.b GVR35 and T.b.b APC098 thus no significant difference in parasite numbers in the brain at the time of onset BBB traversal and other time points (Table 1). Therefore the magnitude of traversal may remain the same irrespective of the timing of the onset of traversal.
Table 1: Determination of pre-patent period to compare the number of trypanosomes in the brain parenchyma for different strains at the onset of invasion or for the same strain at different time points. View Table 1
Mice infected with the appropriate strain were treated at the earliest onset BBB traversal and their brain used to infect new mice and the pre-patent period (indicator of parasite load) determined and compared for the three strains used. The pre-patent period of T.b.b 729X at the onset of BBB traversal (3 dpi) was compared with that at subsequent point (6 dpi) and the onset of T.b.b GVR35 and T.b.b APC098 (6 dpi) also compared with the subsequent ppint (15 dpi).
This study was unable to conclusively substantiate why T.b.b 729X may begin crossing the BBB earlier than the other strains but it was found to be more virulent than the other two (Figure 2).
 Figure 2: Virulence of various strains as measured by the pre-patent period and other infection progression monitoring parameters.
View Figure 2
Figure 2: Virulence of various strains as measured by the pre-patent period and other infection progression monitoring parameters.
View Figure 2
Different groups of mice were infected intramuscularly with 3 × 103 trypanosomes respectively and their pre-patent period (panel a), parasitemia at 6 dpi (panel b) and persistence spans (panel c) recorded. The data ± SEM represent three replicate experiments and were analyzed using One-way Anova or Unpaired t-test. T.b.b 729X had a pre-patent period significantly lower than T.b.b GVR35 (*P < 0.05) or T.b.b APC 098 and presented the highest parasitemia in the first six days and also a significantly (*P < 0.05) shorter persistence span and thus considered to be the more virulent than the other two. There was no significant difference between the pre-patent period and persistence span of T.b.b APC 098 and those of T.b.b GVR35 (P > 0.05). Thus T.b.b 098APC is a mildly virulent field isolate and its virulence is comparable to the lab strain (T.b.b GVR35).
Taken together these results show that all the strains started crossing the blood brain barrier earlier than 9 dpi as reported by Laperchia and colleagues [15] or 13 or 14 dpi that as previously reported [4,17]. Other researchers have shown that trypanosomes are seen extravasculature in the meninges as early as 5 days post-infection but cross the blood brain barrier starting 13 dpi and more extensively at 21 dpi [17]. In their study, Myburgh and colleagues showed that extravascular trypanosomes in the the menigenes were cleared by Diminazene acteturate. This is because the meninges generally lack the blood brain barrier [22] thus the drug is able to clear them. In another study scientists reported trypanosomes in the brain as early as 7 dpi and more at 14 dpi and 21 dpi. However the methods used in this study could not precisely locate the trypanosomes in respect to the blood vessels, they however showed significant blood brain barrier dysfunction starting at 14 dpi [23].
In our study, the three strains started crossing the blood brain barrier at different time points; 3 dpi and 6 dpi for T.b.b 729X and T.b.b APC098 and T.b.b GVR35 respectively. Our data seem to suggest that strains of the same subspecies do not begin crossing the blood brain barrier at the same time. So far, studies looking at BBB traversal have only compared onset of trypanosome BBB traversal at subspecies level but not at isolate/strain level. At subspecies level a difference in the efficiency of traversal was observed whenT.b.gwas compared with T.b.b [24-26]. The difference in BBB traversal at isolate level observed in our study is further backed up by other studies that looked at disease progression but not focusing on BBB traversal, which showed differences in disease progression by different isolates of the same subspecies [24,27,28].
The earliest time of traversal in our study was 3 dpi. Frevert and colleagues indicated traversalas early as two hours post-infection. They were able to detect this probably because they used intravital microscopy which was beyond the scope of this study thus it could still be possible that traversal began within 24 hrs especially for T.b.b 729X. In their study they also used the intraperitoneal and intravenous routes but this study used the intramuscular route. It is possible the intramuscular route we used influenced the outcome. The intramuscular route can be expected to delay the trypanosome in getting into the blood flow thus resulting in slower traversal of the BBB. This however needs to be substantiated in a route comparative study. What is clear though, is that traversal began much earlier for all isolates including the the field isolate. In conclusion one can say that the phenomenon of early brain parenchyma invasion could indeed be a phenomenon of any strains including laboratoryand field strains and may thus have implications on the control of African Trypanosomiasis. Further investigations using more field isolates and laboratory strains are recommended to further substantiate these findings.
We would like to thank Dr. Ann Kazibwe and (Makerere University, Kampala,Uganda) for providing the field isolates and Dr. Claire Mugasa (Makerere University, Kampala Uganda) for critically reading this article.
Monica Namayanja designed the study, participated in conducting the experiments, analysed the data and wrote the manuscript. Gloria Akurut was Monica Namayanja's Research Assistant and helped the conducting the experiments. George W Lubega was Monica Namayanja's Supervisor and critically reviewed the manuscript.