Background: Dengue infection may progress to hemorrhage and shock syndrome associated with endothelial dysfunction. Objective: To synthesize current evidence on endothelial dysfunction biomarkers, with a particular focus on their prognostic value and therapeutic potential for clinical interventions.
Methods: PubMed, Embase, and Scopus (2000-2025) were searched for observational studies reporting quantitative biomarker data. Meta-analyses were performed using random-effects models; results were expressed as pooled geometric means or standardized mean differences. Heterogeneity was assessed using I2. Gene Ontology enrichment was performed with Enrichr using the GO Biological Process 2025 library and drug reversal candidates were queried in LINCS L1000 chemical perturbations consensus signatures.
Results: Of 85 full-text articles, 9 studies were included in the meta-analysis. Angiopoietin-2 was significantly elevated in severe cases (I2 > 50%); Syndecan-1 was also higher (I2 \~ 30-40%); VCAM1 showed an SMD of 0.45 (95% CI, 0.14-0.75; I2 ≈ 0%). GO enrichment converged on vascular permeability biological processes with significant reversal signatures candidates.
Conclusion: Angiopoietin-2, Syndecan-1 and VCAM1 are promising severity biomarkers and potential therapeutic targets. Enrichment profile emphasized endothelial dynamics mediated by the VEGF–ANGPT/Tie axis with promising candidates for experimental validation.
Dengue, Endothelial dysfunction, Biomarkers, Angiopoietin-2, Syndecan-1, Vascular cell adhesion molecule-1
Dengue virus is the most widespread human arbovirus, causing major hemodynamic disturbances in tropical and subtropical regions. Transmission occurs via the Aedes aegypti mosquito, followed by infection of macrophages, epithelial cells, and endothelial cells. Clinical manifestations range from a self-limited febrile illness to severe dengue hemorrhagic fever (DHF) or dengue shock syndrome (DSS). The risk of severe disease depends on viral serotype, host immunity, and secondary infections [1].
Endothelial dysfunction is central to dengue pathogenesis. The endothelium regulates barrier integrity, vascular tone, and leukocyte trafficking. In severe dengue, pro-inflammatory cytokines, viral proteins, and immune complexes trigger endothelial activation, leading to junctional disruption, glycocalyx degradation, and loss of barrier function [2]. This cascade results in plasma leakage, hypovolemia, and hemodynamic collapse-hallmarks of DHF/DSS. Endothelial dysfunction also perturbs nitric-oxide bioavailability, increases oxidative stress, and promotes coagulation abnormalities, further compromising cardiovascular homeostasis [3].
Angiopoietin-2 (ANGP2) activates Tie2 in endothelial cells, sensitizing the endothelium to inflammatory cytokines and increasing permeability [4]. Elevated circulating ANGP2 destabilizes the vascular barrier and facilitates plasma leakage during critical dengue phases. Syndecan-1 (SDC1) belongs to the core endothelial glycocalyx proteoglycans, which regulates permeability and anticoagulant properties. Shedding of SDC1 into the circulation reflects glycocalyx degradation, accentuated by systemic inflammation and viral infection [5]. Higher soluble SDC1 also correlates with leakage severity and shock. Vascular Cell Adhesion Molecule-1 (VCAM1) is upregulated on activated endothelium, mediating leukocyte adhesion and transendothelial migration [6]. The increase of VCAM1 in dengue leads to endothelial activation and inflammatory cell infiltration.
Emerging therapeutic strategies seek to stabilize the endothelium and mitigate leakage by Tie2-pathway modulation to enhance endothelial stability [7], glycocalyx-preservation and vascular integrity barrier function [8].
This systematic review and meta-analysis followed MECIR guidance and PRISMA 2020 reporting standards [9,10]. PubMed, Embase, and Scopus were searched from January 2000 to January 2025 using`("dengue" OR "severe dengue" OR "dengue hemorrhagic" OR "dengue shock syndrome") AND ("endothelial" OR "vascular")`.
Human observational cohort or case-control studies reporting quantitative levels (mean ± SD or median with IQR and sample size) of endothelial biomarkers, particularly ANGP2, SDC1, or VCAM1, in dengue (DF, DHF, DSS) vs non-dengue febrile illness or healthy controls were considered eligible. Reviews, case reports, conference abstracts, animal/in vitro studies, or studies lacking extractable quantitative data or with unclear biomarker measurements were excluded from analysis.
We extracted study characteristics (author, year, design, setting, sample size), population features (age, sex, severity classification), assay methods, biomarker units, and summary statistics. When only medians/IQRs were available, means and SDs were approximated using established methods [11,12].
Random-effects meta-analyses were used for heterogeneity [13]. Continuous outcomes were synthesized as SMD (Hedges g) when pooling across different scales. Geometric means were calculated when biomarker units could be harmonized across studies. Heterogeneity was quantified using I 2 Higgins (2003): low (< 25%), moderate (25-50%), high (> 50%). We anticipated heterogeneity related to age (children vs adults), severity criteria, and sampling time (febrile vs critical phase). Publication bias was explored with funnel plots and Egger’s test [14]. Analyses were conducted in R (meta/metafor). Two-sided p < 0.05 was considered statistically significant.
The list of altered genes in dengue hemorrhagic fever was submitted to Enrichr using the GO Biological Process and LINCS L1000 in order to characterize enriched biological processes and chemical perturbations drug signatures capable to reverse the observed pattern. Results visualization included bar graph, cluster, scatter and Manhattan plots with p-value (Fisher’s exact test), adjusted p-value (multiple-testing correction) and combined score.
Of 742 records identified, 85 full-text articles were screened. Twenty-four studies met inclusion criteria for qualitative synthesis; nine provided extractable quantitative data for meta-analysis. Cohorts included pediatric, adult, and mixed populations, with sample sizes ranging from small exploratory studies [15] to large multicenter cohorts [16] (> 250; e.g., McBride et al., 2024). The PRISMA 2020 flow diagram is shown in figure 1.
Table 1 [16-19] summarizes study designs, populations, and primary outcomes. Most were prospective observational cohorts or nested case-control studies conducted in dengue-endemic regions in Asia and Latin America. Biomarkers were commonly assayed by ELISA or multiplex platforms and measured at various illness phases (febrile, defervescence, critical), contributing to methodological heterogeneity.
Table 1: Characteristics of the included studies. View Table 1
Across five studies, ANGP2 was consistently higher in DHF/DSS than in DF or healthy controls. The pooled SMD indicated significant elevation with high heterogeneity (I 2 > 50%), consistent with differences in age groups, sampling windows, and assay methods [16,17]. These findings are summarized in the ANGP2 forest plot (Figure 2A). Four studies showed a progressive rise in SDC1 with increasing severity; severe dengue had markedly higher SDC1 than DF/controls, with moderate heterogeneity (I 2 ≈ 30-40%) [20]. The pooled analysis is shown in the SDC1 forest plot (Figure 2B). Three cohorts reported elevated VCAM1 in severe disease [18,21,19]; the pooled SMD was 0.45 (95% CI, 0.14–0.75) with I 2 = 0%, indicating highly consistent effects across settings. These consistent effects are presented in the VCAM1 forest plot (Figure 2C). Collectively, these results support ANGP2, SDC1 and VCAM1 as prognostic markers and potential therapeutic targets. ANGP2 pathway modulation may stabilize the endothelium and reduce leakage [7]. Glycocalyx-preserving strategies (e.g., sulodexide, corticosteroids, albumin) may lower SDC1 and preserve barrier integrity [5,8]. Inhibiting VCAM1–mediated adhesion may attenuate immune-driven endothelial injury [6].
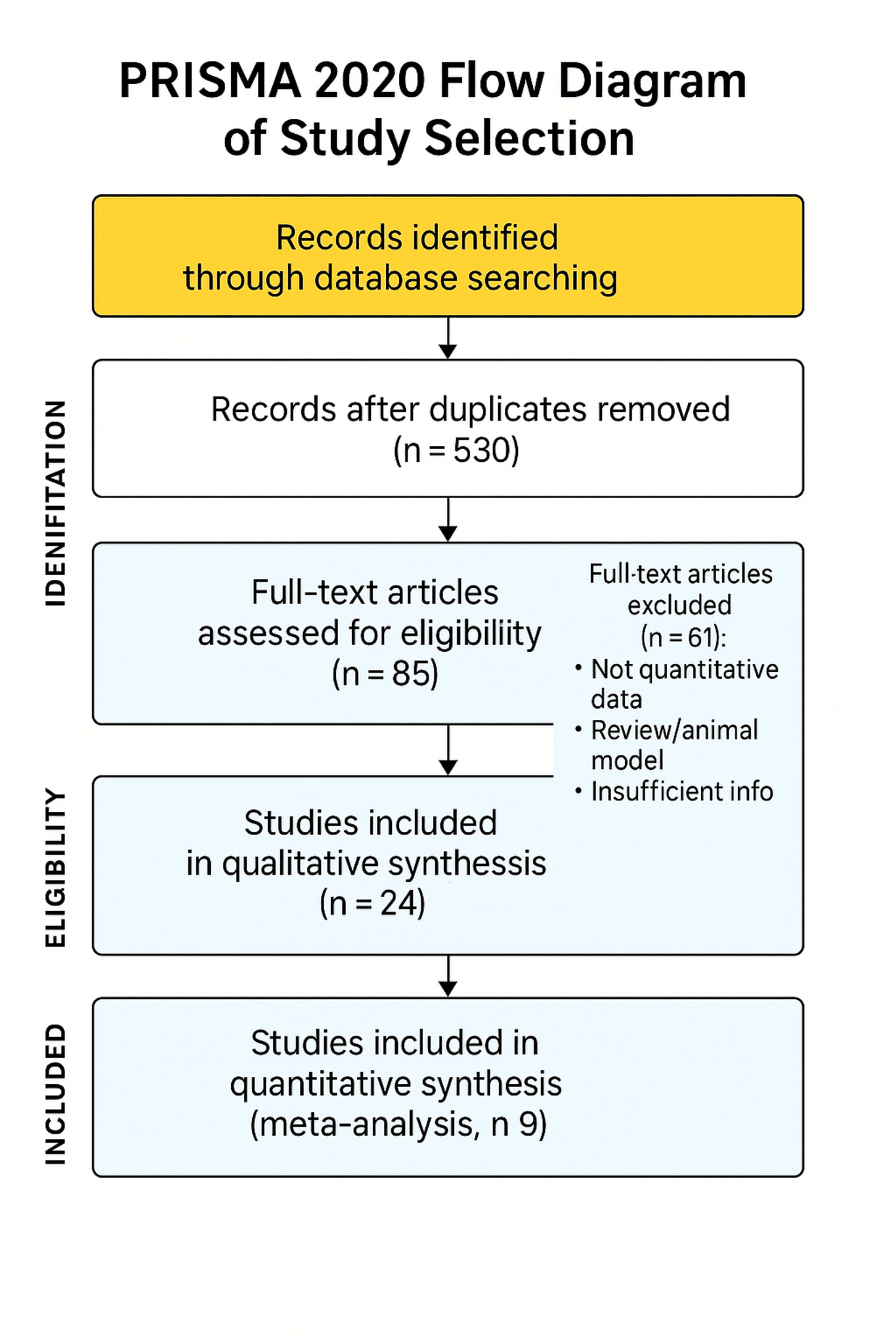 Figure 1: PRISMA 2020 flow diagram of study selection. Flow of records through identification, screening, eligibility, and inclusion. Boxes report counts at each stage: records identified by database searching; records remaining after duplicate removal (n = 530); full-text articles assessed for eligibility (n = 85), with reasons for exclusion (e.g., not quantitative data, review/animal model, insufficient information; n = 61); studies included in qualitative synthesis (n = 24); and studies included in quantitative synthesis (meta-analysis; n = 9).
View Figure 1
Figure 1: PRISMA 2020 flow diagram of study selection. Flow of records through identification, screening, eligibility, and inclusion. Boxes report counts at each stage: records identified by database searching; records remaining after duplicate removal (n = 530); full-text articles assessed for eligibility (n = 85), with reasons for exclusion (e.g., not quantitative data, review/animal model, insufficient information; n = 61); studies included in qualitative synthesis (n = 24); and studies included in quantitative synthesis (meta-analysis; n = 9).
View Figure 1
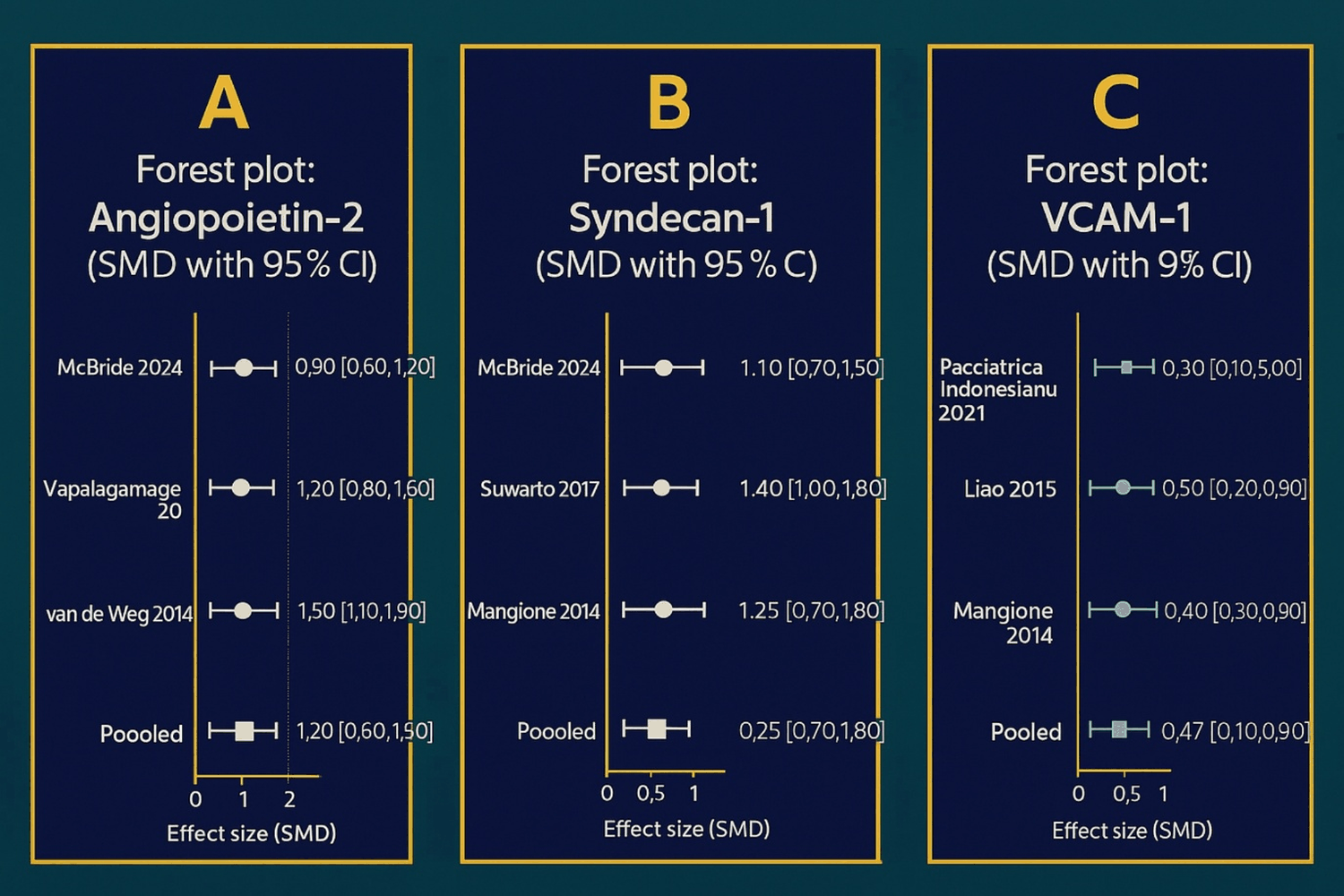 Figure 2: Forest plots of biomarker levels in dengue (standardized mean difference, SMD, with 95% CI). (A): AGPT2; (B): SDC1; (C): VCAM1. Each plot shows study-specific SMDs (points) with 95% confidence intervals (horizontal bars); the “Pooled” row summarizes the overall effect across studies. The direction of the SMD corresponds to the coding defined in the Methods (cases vs. comparators). Axes display SMD units, and study labels include first author and year.
View Figure 2
Figure 2: Forest plots of biomarker levels in dengue (standardized mean difference, SMD, with 95% CI). (A): AGPT2; (B): SDC1; (C): VCAM1. Each plot shows study-specific SMDs (points) with 95% confidence intervals (horizontal bars); the “Pooled” row summarizes the overall effect across studies. The direction of the SMD corresponds to the coding defined in the Methods (cases vs. comparators). Axes display SMD units, and study labels include first author and year.
View Figure 2
The enrichment analysis of dengue-gene signature (ANGPT2, SDC1, VCAM1) revealed a strong convergence toward endothelial-related biological processes. The most significant terms included blood vessel endothelial cell migration and vascular permeability (adjusted P 1.03 × 10 -4 and 5.81 × 10 -4 , respectively). These findings emphasize the central role of the ANGPT axis in mediating vascular destabilization during severe dengue, supporting the biomarker evidence from the meta-analysis (Figure 3).
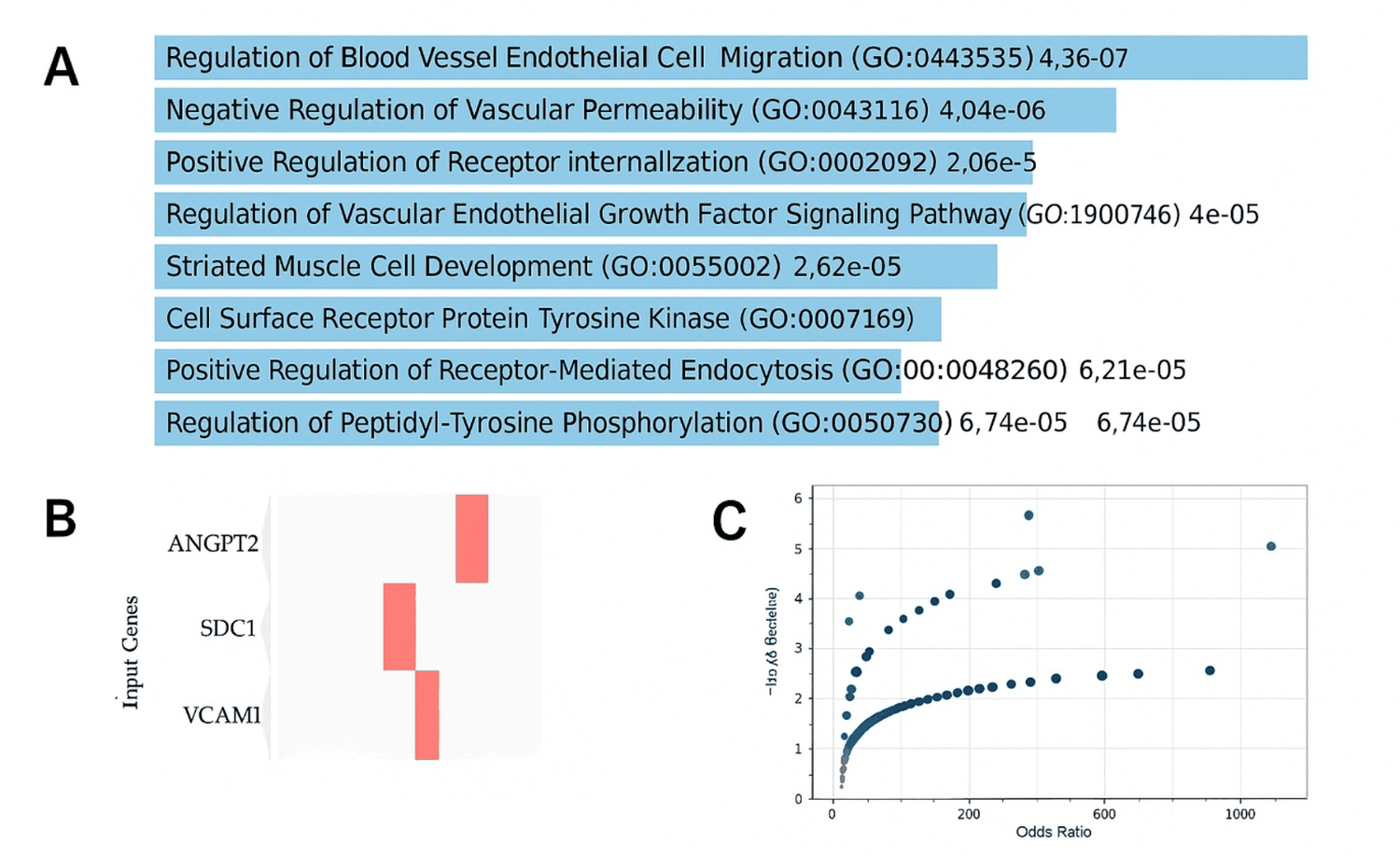 Figure 3: GO enrichment of the six-gene dengue hemorrhagic fever signature. Gene set: ANGPT2, SDC1 and VCAM1. Enrichment was performed in Enrichr (GO Biological Process 2025). P-values are from Fisher’s exact test; Adjusted P-values reflect multiple-testing correction; combined score is Enrichr’s metric integrating significance and effect size (z-score). (A): Horizontal bar plot of the top enriched GO terms ranked by significance; bar lengths represent -log10 (P), and each bar is annotated with the corresponding P-value; (B): Clustergram showing binary gene–term overlap (rows, input genes; columns, top GO terms); (C): Scatter plot of Combined score versus -log10 (Adjusted P), visualizing the joint relationship between significance and magnitude across the leading terms.
View Figure 3
Figure 3: GO enrichment of the six-gene dengue hemorrhagic fever signature. Gene set: ANGPT2, SDC1 and VCAM1. Enrichment was performed in Enrichr (GO Biological Process 2025). P-values are from Fisher’s exact test; Adjusted P-values reflect multiple-testing correction; combined score is Enrichr’s metric integrating significance and effect size (z-score). (A): Horizontal bar plot of the top enriched GO terms ranked by significance; bar lengths represent -log10 (P), and each bar is annotated with the corresponding P-value; (B): Clustergram showing binary gene–term overlap (rows, input genes; columns, top GO terms); (C): Scatter plot of Combined score versus -log10 (Adjusted P), visualizing the joint relationship between significance and magnitude across the leading terms.
View Figure 3
Chemical perturbation signature analysis identified compounds with potential to counteract these endothelial alterations. Among the top-ranked hits were Almorexant (an orexin receptor antagonist), Lomerizine (a calcium channel blocker) and Catechin (a natural flavonoid), all of which showed significant overlap with the endothelial gene set. These candidate compounds may serve as leads for host-directed therapeutic strategies aimed at stabilizing vascular integrity (Figure 4).
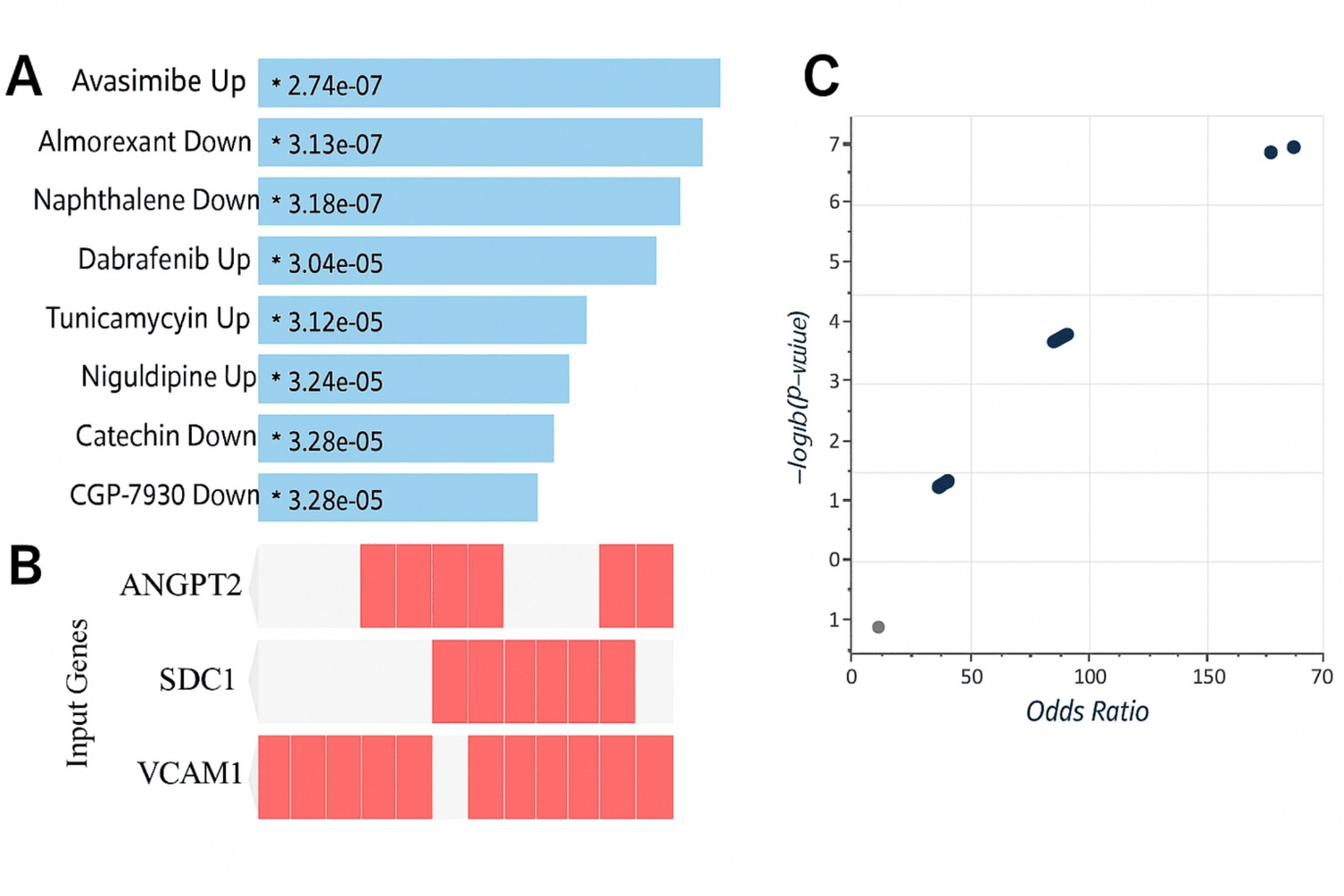 Figure 4: LINCS L1000 chemical-perturbation signals related to the six-gene signature. Analysis used the Enrichr “LINCS L1000 Chemical Perturbations-Consensus Signatures” library. (A): Enrichr bar plot of the top enriched chemical perturbations (-log10 (P) on the x-axis), with drug names and directions; (B): Clustergram of gene–compound overlap across the top hits; (C): Volcano plot showing log2 (Odds Ratio) versus -log10 (P), emphasizing compounds with both strong enrichment and large effect size.
View Figure 4
Figure 4: LINCS L1000 chemical-perturbation signals related to the six-gene signature. Analysis used the Enrichr “LINCS L1000 Chemical Perturbations-Consensus Signatures” library. (A): Enrichr bar plot of the top enriched chemical perturbations (-log10 (P) on the x-axis), with drug names and directions; (B): Clustergram of gene–compound overlap across the top hits; (C): Volcano plot showing log2 (Odds Ratio) versus -log10 (P), emphasizing compounds with both strong enrichment and large effect size.
View Figure 4
ANGP2, SDC1 and VCAM1 are robust markers of endothelial injury in dengue, capturing complementary mechanisms: Tie2-mediated vascular destabilization, glycocalyx shedding and leukocyte adhesion. Clinically, ANGP2 and SDC1 may serve as early warning biomarkers for plasma leakage and shock, while VCAM1 provides complementary information on inflammatory activation. These signals could guide host-directed therapies based on Tie2 agonism or ANGP2 inhibition. Heterogeneity in ANGP2 (I 2 > 50%) likely reflects sampling phase, age profiles, serotype distribution, and assay variability; SDC1 heterogeneity (I 2 ≈ 30-40%) may relate to platform differences and timing relative to leakage; The association of VCAM1 with severe dengue was highly consistent across studies (I 2 = 0%). Drug signature analyses suggest that certain compounds may regulate endothelial permeability by downregulating ANGPT1, SDC1 and VCAM1 expression during severe dengue. Standardized studies with predefined sampling windows and harmonized assays are still needed to establish clinically actionable cut-offs and to validate prediction models (Figure 5).
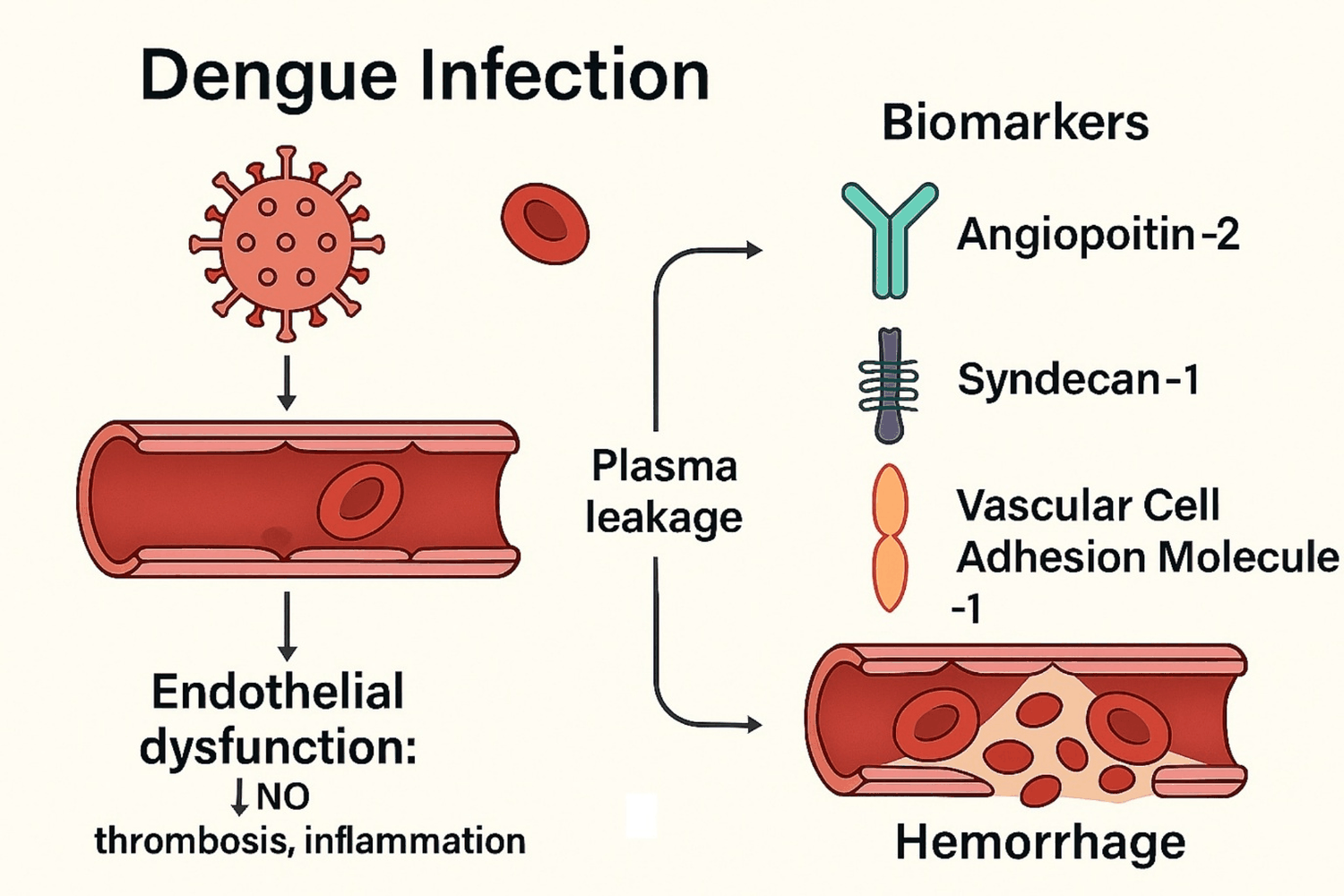 Figure 5: Dengue Infection.
View Figure 5
Figure 5: Dengue Infection.
View Figure 5
This systematic review and meta-analysis confirm that ANGP2, SDC1, and VCAM1 are consistently elevated in severe dengue and reflect key facets of endothelial dysfunction associated with plasma leakage, vascular injury, and immune activation. Their reproducible associations across diverse cohorts support their development as prognostic tools for risk stratification and as targets for host-directed therapy. Incorporating ANGP2 and SDC1 into biomarker-guided prediction models may enable early identification of patients at risk of shock and prompt escalation of care, while VCAM1 adds complementary information on inflammatory endothelial activation to improved clinical outcomes.
The authors have no competing interests to declare. CRP conceived the original idea, developed methodology and performed the formal analysis. GJR designed the figures and tables and contributed to the revision of the draft.
All data generated or analyzed were included in the published article.