Background: With the advent of HAART, natural history of HIV/AIDS has changed radically and PLHIV who are treated before significant immunosuppression can expect to have a longer life expectancy. Previous studies have suggested a rising rate of non-AIDS related events in HIV infected individuals of which cardiovascular diseases constitute significant morbidity and mortality. Identification of factors augmenting the cardiovascular risk is important in the management of HIV.
Methods: The present study was a cross-sectional, observational study carried out in a tertiary care centre from January 2021 to July 2022. 200 HIV positive individuals on HAART for atleast 2 years were included who underwent ECG, chest radiograph, 2D Echocardiography and CD4+ T cell count along with other routine investigations. Presence of cardiovascular involvement was determined on the basis of echocardiographic abnormalities and these findings were associated with demographic, haematological and biochemical profile of the patients. The echocardiographic abnormalities were correlated with CD4+ T cell count and clinical stage of the disease.
Results: 38.50% of the population had abnormal ECG findings of which sinus tachycardia was the most common (30.00%). The prevalence of echocardiographic abnormalities was 41.00%. The most common echocardiographic abnormality was diastolic dysfunction (32.00%) followed by valvular insufficiency (21.00%) and reduced LVEF (15.00%). The mean of CD4+ T cell count in those with abnormal echocardiographic findings was 194.43 ± 161.4 cells/mm3 which was significantly lower than those with normal echocardiographic findings (555.02 ± 393.36 cells/mm3). With decrease in the CD4+ T cell count and with increase in WHO stage, cardiac abnormalities increased significantly. Higher grades of diastolic dysfunction were seen with CD4+ T cell count < 350/mm3 and WHO stages 3 and 4.
Conclusion: In the present study, there was a high prevalence of cardiovascular abnormalities among PLHIV. Lower CD4+ T cell count was an independent risk factor for cardiovascular abnormalities and hence predicts the occurrence of cardiovascular disorders among PLHIV.
PLHIV, Cardiovascular, CD4 + T cell count, Diastolic dysfunction
AIDS: Acquired Immunodeficiency Syndrome; ALP: Alkaline phosphatase; ART: Antiretroviral Therapy; CVD: Cardiovascular Diseases; ESR: Erythrocyte Sedimentation Rate; HAART: Highly Active Antiretroviral Therapy; HIV: Human Immunodeficiency Virus; JVP: Jugular Venous Pressure; LVEF: LV ejection Fraction; NACO: National AIDS Control Organization; PLHIV: People Living with HIV/AIDS; UNAIDS: United Nations Programme on HIV/AIDS; VLDL: Very Low Density Lipoprotein
Human Immunodeficiency Virus (HIV) is an important infectious pathogen which has affected the entire world over past four decades with significant impact on the health of the population particularly in developing nations. As per the Joint United Nations Programme on HIV/AIDS (UNAIDS), around 38.4 million people globally were living with HIV in 2021. Around 1.5 million people became newly infected and approximately 650,000 people died from AIDS-related illnesses in 2021 [1]. Currently India has the third highest burden of HIV in the world after South-Africa and Nigeria. As per the National AIDS Control Organization (NACO) report 2021, the adult HIV prevalence is 0.21% and total number of people living with HIV/AIDS (PLHIV) was around 24.01 lakh by end of 2021. 41,970 patients died due to AIDS-related deaths in the year 2021 [2].
With the introduction of highly active antiretroviral therapy (HAART), there has been a substantial reduction in deaths related to AIDS while non-AIDS related deaths have increased due to greater longevity of PLHIV. Cardiovascular diseases (CVD) have emerged as an important cause of non-AIDS related morbidity and mortality among PLHIV. Large HIV cohort studies have convincingly demonstrated an increased cardiovascular risk in HIV-infected individuals compared with the general population in developing countries [3]. Global burden of CVDs on the background of HIV-infection has tripled over the last two decades and accounts for 2.6 million DALYs per annum with greatest impact in the sub-Saharan Africa and Asia Pacific regions. The crude rate of CVD was 61.8% (95%CI: 45.8-83.4) [4].
Combined burden of HIV and CVD has important implications with respect to regional and national health policies, guidelines and resource allocation. Traditional risk factors such as smoking, hypertension and dyslipidemia contribute substantially to increased risk of CVDs but there also is evidence that the HIV infection and to a lesser extent, its treatment are directly linked to atherogenesis, endothelial dysfunction, coagulation abnormalities, probably through inflammation and immune dysregulation. The degrees to which HIV itself, cardiovascular risk factors and HAART individually contribute to increased cardiovascular risk in PLHIV is unknown. Currently used 10 year risk calculators do not take in account these additional risk factors in HIV patients.
Due to a constellation of non-specific complaints, cardiovascular disorders are frequently missed and overlooked among PLHIV. Timely diagnosis and appropriate management is crucial for a holistic management of HIV/AIDS. The objective of the present study was to estimate the prevalence of CVD in PLHIV on HAART and risk factors associated with it.
This study was a single-centre, hospital based, observational study carried out at a tertiary care centre in Northern India from January 2021 to July 2022. 200 HIV positive individuals were enrolled in the study. All patients were explained about details of the study and informed written consent was obtained. The Institute Ethics committee approved the study.
Patients above 18 years of age who were seropositive for HIV confirmed by ELISA or western blot technique and receiving HAART for atleast 2 years were included in the study. Patients with any pre-existing cardiovascular disorders before the diagnosis of HIV, those suffering from chronic medical disorders other than HIV (diabetes mellitus, hypertension, asthma, chronic obstructive airway disease, hypothyroidism, chronic kidney disease) and those who did not consent for the study were excluded.
Detailed information of all enrolled patients was filled in a proforma designed for the said purpose. This included demographic variables, detailed clinical history, general physical examination and systemic examination. The WHO clinical stage of HIV was established using the WHO criterion [5]. CD4 + T cell count was performed using flow cytometry (BD FACS count system) by the kits provided by NACO to the ART centre. The study population was divided into five groups on the basis of CD4 + T cell count for further comparison and an easier interpretation. All patients underwent routine hematological, biochemical blood tests and appropriately screened for opportunistic infections.
A standard 12 lead ECG was obtained for all the patients in supine position after a 5 minutes rest using standardised procedures. Transthoracic echocardiography (TTE) was performed by experienced cardiologist using Philips Affinity 50 Ultrasound machine using 2.5 to 3.5 MHz transducer probe. Measurements were taken as per the American Society of Echocardiography guidelines [6]. Echocardiography was done by same observer to avoid intra-observer variation. Left ventricular volumes were measured and LV ejection fraction (LVEF) was calculated in apical four chamber (A4C) view and apical two chamber (A2C) views using modified Simpson biplane disc method. LVEF < 50% was considered abnormal and LV systolic dysfunction was graded as mild LV dysfunction (LVEF 40-49%), moderate LV dysfunction (LVEF 30-39%) and severe LV dysfunction (LVEF < 30%). Mitral E (early diastolic mitral flow velocity), mitral A (late diastolic mitral flow velocity) and deceleration time (DT) were measured in A4C view with mitral inflow pulse wave doppler examination at the tips of leaflets and LVDD was categorised into 3 grades- grade I/IV (E/A < 0.75), grade II/IV (E/A 0.75-1.5 with DT > 140 msec) and grade III/IV (E/A > 1.5 with DT < 140 msec).
Presence of cardiovascular involvement was determined on the basis of echocardiographic abnormalities and these findings were associated with demographic, haematological and biochemical profile of the patients. Subgroup analysis was done to look association of echocardiographic abnormalities with CD4 + T cell count and clinical stage of the disease.
Data for each patient was entered in Microsoft Excel spreadsheet and SPSS software, IBM manufacturer, Chicago, USA, version 25.0 was used for analysis of the data. Categorical variables were expressed in the form of number and percentage (%) and analyzed using Chi-Square test or Fisher's exact test. Quantitative data was expressed as the means ± standard deviation and analyzed using independent t test (for two groups) and ANOVA (for more than two groups). Point-Biserial correlation coefficient was used for correlation of cardiovascular abnormalities with CD4 + T cell count and WHO stage. Multivariate logistic regression was used to find out significant risk factors of cardiovascular abnormalities. Statistical significance was determined by p value of less than 0.05.
200 patients were enrolled in the study after taking informed and written consent. 67.00% patients were in the age group of 31-50 years. The mean age was 40.15 ± 10.2 years. 66.50% were male, 32.50% were female and 1.00% were transgender. 41.50% of the patients were smokers, 21.00% were alcoholics whereas 49.50% had no addictions. Only 6 out of 200 patients (3.00%) were substance abusers and 13.50% had more than one addiction.
Most of the patients were on Tenofovir + Lamivudine + Efavirenz (91.00%) followed by Zidovudine + Lamivudine + Nevirapine (4.50%) and Tenofovir + Lamivudine + Dolutegravir (2.50%). Mean duration of ART was 59.66 ± 30 months. More than half of the study population were asymptomatic. Among others, shortness of breath was the most common complaint as shown in Table 1. Majority (76.00%) had normal findings on physical examination (Table 2).
Table 1: Presenting complaints of the study population. View Table 1
Table 2: Physical examination findings of the study population. View Table 2
45.50% of the patients had opportunistic infections of which the most common was oral candidiasis (10.00%) followed by pulmonary tuberculosis (7.00%). Majority of the patients belonged to WHO clinical stage 1 (44.50%) followed by stage 3 (20.50%), stage 4 (18.50%) and stage 2 (16.50%). CD4 + T cell count ranged from 21 to 1496 cells/mm 3 with a mean of 408.36 ± 365.43 cells/mm 3 . Approximately a quarter (26.50%) patients had CD4 + T cell count greater than 500 cells/mm 3 (group A), 17.50% had between 350-499 cells/mm 3 (group B), 17.00% had between 200-349 cells/mm 3 (group C), 19.00% had between 100-199 cells/mm 3 (group D) and 20.00% had below 100 cells/mm 3 (group E).
The overall prevalence of ECG abnormalities was 38.50%, most common being sinus tachycardia (30.00%) followed by QRS abnormalities (9.50%), ischemic changes (8.50%) and poor progression of R wave (5.50%). Table 3 shows the ECG abnormalities in the study population.
Table 3: ECG findings of the study population. View Table 3
Chest radiograph was normal in majority (80.50%) of the population. Cardiomegaly was present in 9.00% which was associated with abnormal echocardiography such as cardiomyopathy, pericardial effusion, ischemic heart disease or pulmonary hypertension. Abnormal echocardiographic findings were seen in 41.00% of patients. Diastolic dysfunction was the most common echocardiographic abnormality (32.00%) followed by valvular insufficiency (21.00%) and reduced LVEF < 50% (15.00%) (Figure 1).
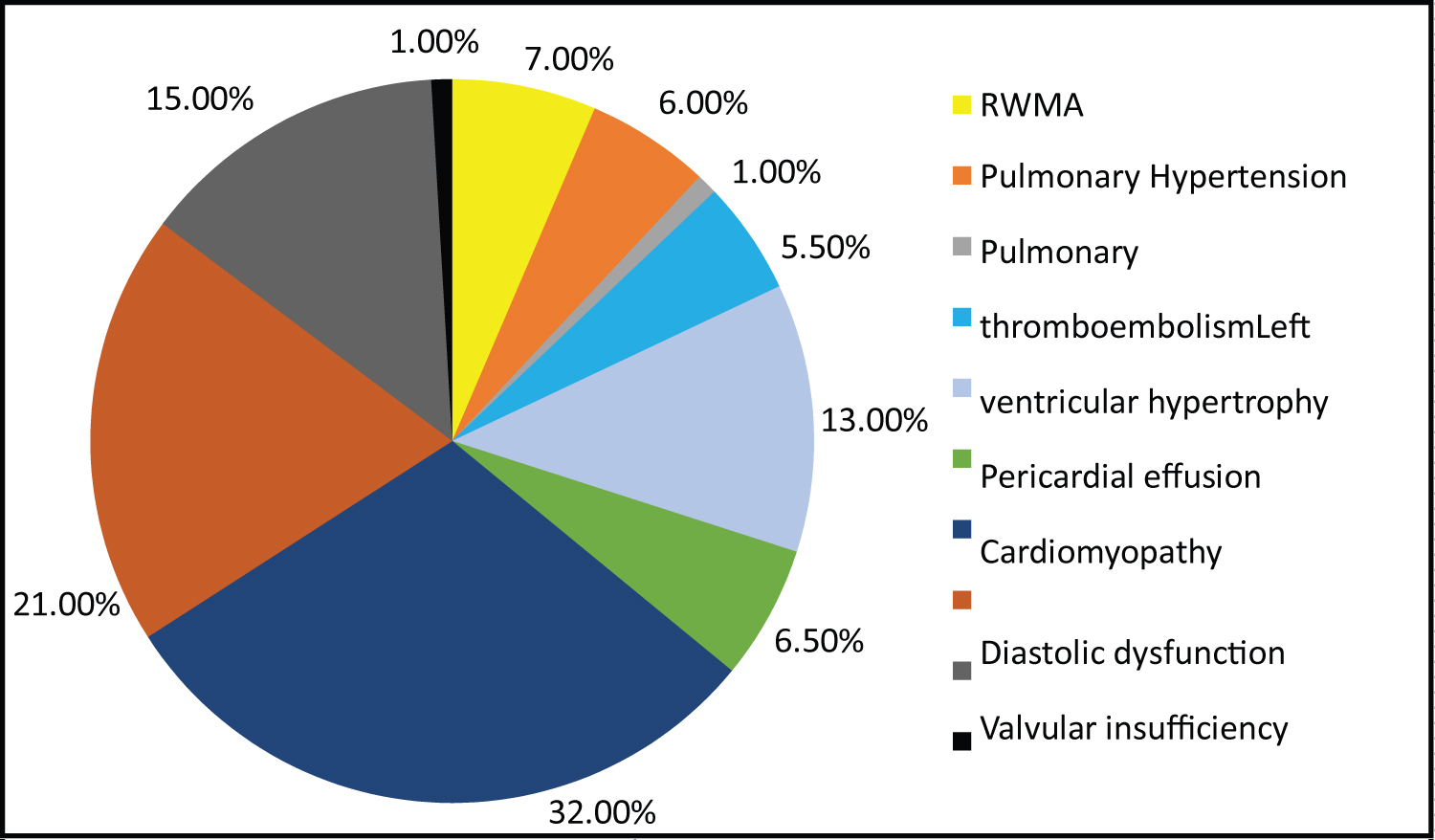 Figure 1: Echocardiographic abnormalities of the study population.
View Figure 1
Figure 1: Echocardiographic abnormalities of the study population.
View Figure 1
Mean age of subjects with abnormal echocardiographic findings was significantly higher as compared to those with normal echocardiography (38.49 ± 10.04 years) (p = 0.006) and 50% of the patients were smokers (p = 0.02). Significant association was seen between haemoglobin, serum calcium, alkaline phosphatase (ALP), serum triglycerides, VLDL and ESR with echocardiographic findings (p < 0.05).
Mean CD4 + T cell count (cells/mm 3 ) was significantly lower in patients with abnormal echocardiography (194.43 ± 161.4) (p < 0.0001). Positive correlation was also seen between echocardiographic abnormalities and WHO stage with correlation coefficient of 0.454. Opportunistic infections were associated with more echocardiographic abnormalities. No significant association was observed between duration or ART regimen with echocardiographic abnormalities.
Lower CD4 + T cell count (< 350 cells/mm 3 ) was associated with higher echocardiographic abnormalities (p < 0.0001) (Table 4). With decrease in CD4 + T cell count, the severity of diastolic dysfunction also increased (p = 0.03) (Figure 2). Higher grades of diastolic dysfunction were seen among patients with WHO stage 3 and 4.
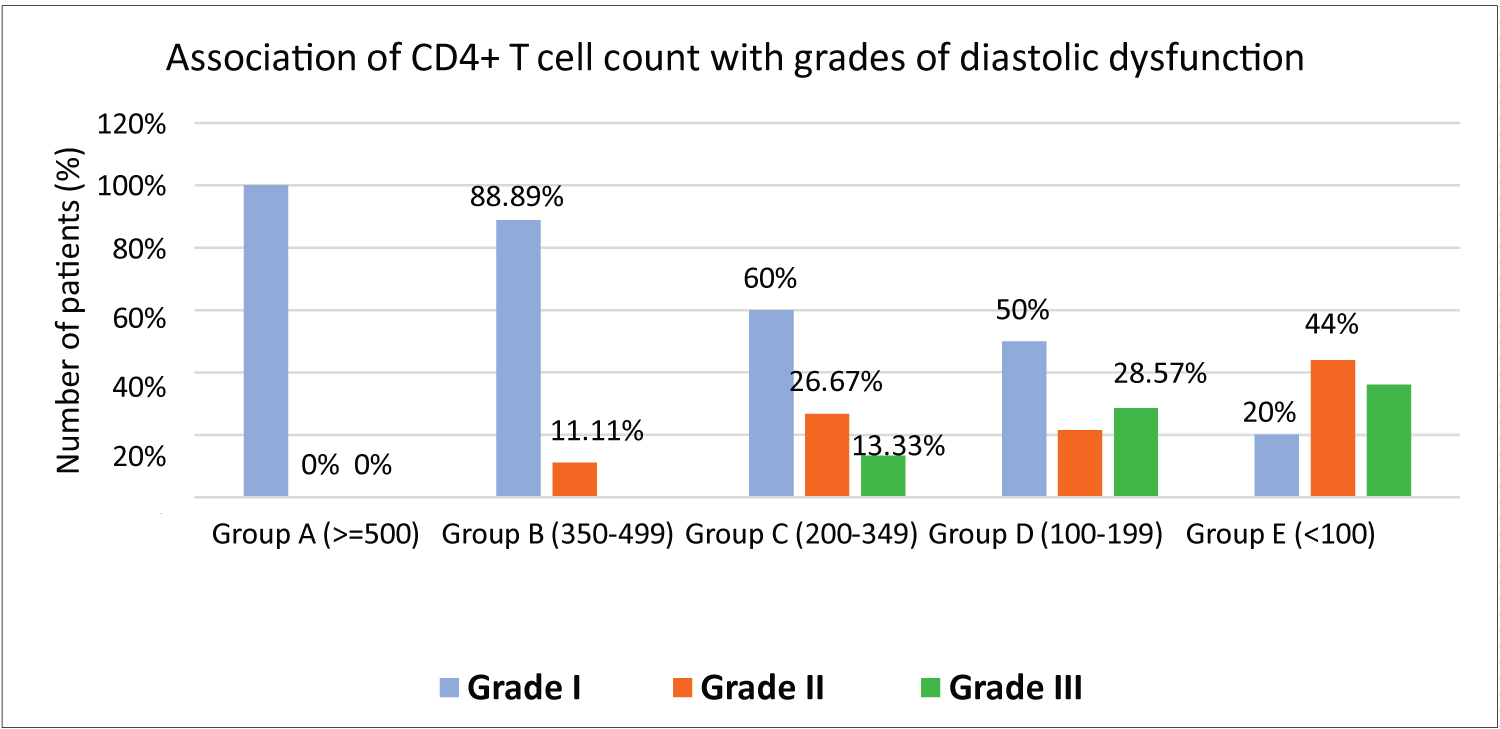 Figure 2: Association of CD4+ T cell count with diastolic dysfunction.
View Figure 2
Figure 2: Association of CD4+ T cell count with diastolic dysfunction.
View Figure 2
Table 4: Association of echocardiographic abnormalities with CD4+ T cell count. View Table 4
On performing multivariate regression, ALP, ESR, abnormal ECG finding and CD4 + T cell count were significant independent risk factors of abnormal echocardiographic findings after adjusting for confounding factors (Table 5).
Table 5: Multivariate logistic regression to find out significant risk factors of abnormal echocardiographic findings. View Table 5
HIV-infection is a global pandemic with cases reported from every corner of the world. It has led to significant morbidity and mortality among people of every age group posing a huge burden to the health system. In our study, most of the patients were in the age group of 31-50. A large proportion of HIV-infected population in our country belongs to the middle-aged reproductive age group. The frequency of males was almost twice that of females which could be attributed to gender bias in getting tested due to various socio-geographical, cultural and economic norms. Males have better access to healthcare facilities and at the same time more exposed to commercial sex workers due to migration.
Among the study population, majority were on Tenofovir + Lamivudine + Efavirenz regimen as this was the preferred regimen as per the NACO guidelines before 2020 and no significant association seen between ART regimen and duration of HAART with cardiovascular abnormalities. Other studies showed slightly different results such as study done by Reddy, et al. which found that longer the duration of disease, more were the echocardiographic abnormalities [7].
More than half of the study population were asymptomatic. Others largely had non-specific complaints and overlapping symptoms secondary to opportunistic infections. This was different from previous studies which aimed to look at cardiovascular involvement where shortness of breath, cough, fatigue, fever and chest pain were frequent presenting complaints [8-10]. Clinical examination of the study population revealed tachycardia as the most common finding (30.00%) followed by pallor (18.50%) and raised jugular venous pressure (7.50%). Marwadi, et al. demonstrated pallor as the most common finding (44.00%) followed by lymphadenopathy (38.00%), tachycardia (36.00%) and raised JVP (6.00%) [9].
40.00% of the study population had opportunistic infections, Tuberculosis being the most common. Tuberculosis enhances the progression of HIV infection by inducing immune activation which is also a proposed mechanism in the pathogenesis of CVD. Cardiac involvement in tuberculosis is frequent and can be fatal if not recognized in time. In tuberculosis, involvement of pericardium is relatively common, especially in AIDS, and tuberculosis remains the most common cause of constrictive pericarditis in endemic countries. Severe immunosuppression results in several opportunistic infections and the co-occurrence of these infections among PLHIV can involve the heart.
Among the ECG findings, sinus tachycardia was most common (30.00%). This could be secondary to anaemia which is commonly encountered among PLHIV. It can also be due to cardiovascular compromise such as ischemic heart disease, heart failure, myocarditis, cardiomyopathy, diastolic dysfunction, pulmonary thromboembolism etc.
Of total patients with abnormal ECG, 85.71% of patients had CD4 + T cell count < 350/mm 3 . Similar study by Tuteja, et al. observed a highly significant correlation between ECG abnormalities and CD4 + T cell count with a p < 0.0001 [11]. In our study, proportion of patients having abnormal ECG was significantly higher in WHO stage 3 and 4 (68.83%) as compared to stage 1 and 2 (31.16%) (p < 0.0001). Similar observations were made by Sunderrajan, et al. [12]. In our study, abnormal ECG was found to be a significant independent risk factor of abnormal echocardiography with an adjusted odds ratio of 5.07 (95%CI: 1.84-13.974). This suggests that ECG could provide a convenient screening tool for cardiovascular abnormalities in PLHIV. Soliman, et al. also found that the presence of ECG abnormalities can independently predict the incidence of CVD [13].
In our study, echocardiography was abnormal in 41.00% of the population and the most common echocardiographic abnormality in our study was diastolic dysfunction (32.00%). Jain, et al. also observed a high prevalence of diastolic dysfunction in their study (46.95%) and concluded that diastolic dysfunction has independent association with HIV infection without being affected by traditional risk factors such as hypertension, diabetes, smoking and age [14]. Most of the patients belonging to WHO stage 1 had grade I diastolic dysfunction (83.33%) while majority of the patients with grade II and III diastolic dysfunction belonged to WHO stage 3 and 4 (p < 0.0001). We observed that as the CD4 + T cell count decreased, the severity of diastolic dysfunction increased and was statistically significant (p = 0.0005). Indonesian study by Hidayat, et al. highlighted CD4 + T cell count < 200/mm 3 as an independent predictor of diastolic dysfunction [15]. Association of age with cardiovascular abnormalities was also statistically significant with a p = 0.05. Age is a traditional risk factor in the natural history of many cardiovascular disorders. CVD tends to occur approximately 10 years earlier in HIV infected individuals than in the general population [16]. With increasing age, due to decreased immunological tolerance, opportunistic infections increase in PLHIV and in turn can result in certain cardiovascular abnormalities.
Inflammatory marker like ESR was significantly higher in patients with abnormal echocardiography (p < 0.0001) and was also an independent risk factor for abnormal echocardiography (aOR = 1.021, 95%CI: 1.001-1.041). Inflammation could directly impair myocardial function, affect LV remodelling through local induction of matrix metalloproteinases, induce endothelial dysfunction in small vessels resulting in an impaired coronary flow reserve [17]. Also, higher ALP was significantly associated with cardiovascular involvement. ALP is an inflammatory biomarker and is also associated with vascular calcification and atherosclerosis. Raised ALP in HIV patients could also indicate accelerated atherosclerosis.
Serum triglycerides and VLDL were significantly higher among those with echocardiographic abnormalities. Studies have consistently shown a high prevalence of dyslipidemia among PLHIV both with and without ART which is a strong risk factor for CVD. It is not known how HIV itself causes dyslipidemia. One postulated mechanism is that HIV inhibits cholesterol efflux from human macrophages and accumulation of lipids within macrophages occurs via impairment of the ATP-binding cassette transporter A1 (ABCA1)-dependent cholesterol efflux [18]. Protease inhibitors and non-nucleoside reverse transcriptase inhibitors are notorious to caused derangement in lipid levels. But this association was not seen on multivariate analysis. Our findings agree with Riddler, et al. who opined that the use of ART results in a return to pre-HIV infection serum lipid levels [19]. Most of the patients in our study were on tenofovir and lamivudine-based regimen which has been associated with lesser adverse effect on lipid profile and even lipid lowering effect [20].
In our study, with decrease in the CD4 + T cell count, echocardiographic abnormalities increased significantly (p < 0.0001). Proportion of patients with abnormal echocardiography was significantly higher in WHO clinical stages 3 and 4 (p < 0.0001). It can hence be concluded that the degree of immunosuppression correlates significantly with the development of cardiovascular abnormalities. Our findings were consistent with the study of Mishra, et al. where echocardiographic abnormalities were strongly associated with CD4 + T cell count [10]. This appears accurate since CD4 + T cell cells play a central role in the activation of both humoral and cellular immune response and immunosuppression predisposes patients to a variety of opportunistic infections, increased oxidative stress and persistent immune activation. On multivariate logistic regression, CD4 + T cell count was significant independent risk factor for cardiovascular abnormalities after adjusting for confounding factors. Hence it can be inferred that early diagnosis and initiation of HAART in HIV can potentially prevent the occurrence and progression of cardiovascular abnormalities.
There were certain limitations in this study. As it was a cross sectional study, cause-effect relationships couldn’t be assessed. Also, should PLHIV with mild cardiovascular abnormalities be treated, are to be answered in the future through further studies with larger numbers of participants, which would also help in understanding the natural history of individual abnormalities with the course of disease. Since all the patients were on HAART, the effect of HIV alone on cardiovascular abnormalities couldn’t be assessed. Selection and comparison with HIV-negative controls would help to establish the contribution of HIV to cardiovascular abnormalities.
Since HIV virtually affects every organ of the body, incidence and prevalence of cardiovascular disorders have increased in the previous decades. Many of these disorders do not cause any symptom and on the background of a cluster of non-specific symptoms, cardiovascular abnormalities are often missed. The present study shows a high prevalence (41.00%) of cardiovascular abnormalities in adult population of PLHIV. Diastolic dysfunction, valvular insufficiency and reduced LVEF are common echocardiographic abnormalities encountered in this subset of population. This suggests that cardiovascular abnormalities still remain a challenge in patients taking HAART for more than two years.
ECG and importantly echocardiography are excellent non-invasive techniques for early recognition of cardiovascular abnormalities. The present study highlights that CD4 + T cell count can be a reliable predictor of cardiovascular abnormalities. Lower CD4 + T cell count and higher clinical stages are strongly associated with CVDs. Immunosuppressed individuals who continue to have lower CD4 + T cell count while on HAART should be adequately evaluated for cardiovascular disorders and practical efforts to optimise their immunological recovery should be stressed upon. Routine screening and assessment of cardiovascular abnormalities in PLHIV at periodic intervals should be done for a holistic management of HIV/AIDS.
This study was duly approved by the Pt. B.D. Sharma University of Health Sciences Ethics Committee (decision no: BREC/Th/20/Med./03) and the postgraduate board of studies of the institution.
Informed consent was taken from all participants.
No conflict of interest was declared by the authors.
The authors declared that this study received no financial & material support.
No assistance of medical writing experts taken.