Acute Respiratory Infections (ARIs) are classified according to the compromised anatomical site. The etiology can be associated with viruses, bacteria and fungi. The etiological agents that are responsible for the highest incidence in children, around 50 to 90% of occurrences, are viruses. This study aims to characterize the factors that contribute to acute respiratory infection in children from 0-6 years old and the main symptoms that are presented, thus classifying the etiological agents. This is a descriptive quantitative study carried out by the molecular virology laboratory of the Research Center in Tropical Medicine-CEPEM/RO and the Oswaldo Cruz Foundation Rondonia-FIOCRUZ/RO. Data collection was performed through a questionnaire with 660 patients of both sexes, with questions about socioeconomic data and clinical manifestations, from February to December 2013. Prevalent clinical manifestations were cough, coryza, pulmonary secretion, fever, nasal obstruction, otalgia, adventitious sounds, apnea, dyspnoea and ocular pruritus of the 113 children diagnosed with acute respiratory infection, the highest incidence was in males. The proposed pathogens in the study were rhinovirus, parainfluenza 1, 2, 3, and adenovirus. Parainfluenza 2 and rhinovirus are the most prevalent.
Acute respiratory infection, Etiological agent, Children
CEPEM/RO: Research Center in Tropical Medicine; FIOCRUZ/RO: Oswaldo Cruz Foundation Rondônia; ARIs: Respiratory Infections; URIs: Upper Respiratory Tract Infections; LRTIs: Lower Respiratory Tract Infections; UHS: Unified Health System (Sistema Único de SaÚde-SUS); REC: Research Ethics Committee (Comissão de Ética em Pesquisa-CEP); NRC: National Research Commission (Comissão Nacional em Pesquisa-CONEP); CPqRR: René Rachou Research Center; HRV: Human Rhinoviruses; PIV1: Parainfluenza Virus 1; PIV2: Parainfluenza Virus 2; PIV3: Parainfluenza Virus 3; NCBI: National Center for Biotechnology Information; BLAST: Basic Local Alignment Search Tool; ICF: Informed Consent Form; IBGE - Brazilian Institute of Geography and Statistics
Acute Respiratory Infections (ARIs) are classified according to the compromised anatomic site in Upper Respiratory Tract Infections (URIs), which include the most prevalent types of ARIs (rhinitis, rhinopharyngitis, sinusitis, pharyngotonsillitis, otitis, laryngitis and acute epiglottis) and acute Lower Respiratory Tract Infections (LRTIs), which have a greater potential to be severe (bronchitis, pneumonias, laryngotracheobronchitis, bronchopneumonia and acute bronchiolitis) [1,2].
All these dysfunctions can present symptoms such as fever (above 37.8 degrees Celsius), changes in pulmonary auscultation, production of purulent exudate, and pain [3-5]. However, the main symptoms of acute respiratory infections include fever, cough, difficulty breathing, coryza, nasal obstruction, sore throat and ear, oropharyngeal inflammation, otalgia, anorexia, sub and intercostal drainage, and cyanosis. However, these symptoms may vary depending on the site reached, etiologic agent, inflammatory process and accumulation of secretions in the respiratory tract. The etiology can be associated with viruses, bacteria and fungi [6,7].
Since the twentieth century, respiratory tract disorders have become the leading cause of infant mortality, affecting children under the age of five due to lack of knowledge during early symptoms, poor health and inadequate treatment [8]. Children are more susceptible to respiratory infections due to anatomical, physiological and immunological characteristics [9-11].
Risk factors for occurrence and hospitalization due to acute respiratory infections include environmental, socioeconomic, demographic, nutritional, and maternal factors along with morbid antecedents. The dry climate, high relative humidity, family agglomeration, low family income, male gender, age range of less than six-months-old, early weaning, smoking, low birth weight, low maternal education level, hospitalization history, inadequate housing conditions and antecedents of respiratory symptoms [12,13].
In 2010, infectious diseases accounted for 58% of global deaths among children under the age of 5, among which one-third of the deaths were caused by pneumonia, diarrhea and malaria in this age group. An estimated 70,000 children under the age of five die annually in the Americas from acute respiratory diseases. Approximately 15% of all child deaths occurring annually are due to acute respiratory diseases; however, their incidence varies from country to country. While in Canada and the United States of America they account for 2% of the deaths of children aged 0-5, the prevalence in Guatemala is around 21% and in Haiti, 39% [14]. Respiratory diseases were responsible for more than 80,000 child deaths per year in Latin America [15]. In Brazil, acute respiratory infections account for 30% to 60% of incidences mortality in children younger than 5-years-old [16]. In the years from 2008 to 2011, respiratory diseases in Brazil were responsible for 34.5% of the total number of hospital admissions by the Unified Health System (Sistema Único de Saúde-SUS) among those under the age of 1 and for 43.2% of children from 1-4 years old, being the first cause of hospitalizations in all regions of the country [13]. Thus, this study aims to evaluate the etiological agents of acute respiratory infections in children, as well as to analyze relationships between socioeconomic factors and climatic besides the severity of the clinical conditions.
This is a cross-sectional, quantitative and descriptive study, with prospective data collection, performed at Cosme and Damião Children's Hospital, located in the city of Porto Velho, Rondônia, Brazil. This hospital is a reference throughout the state, meeting not only the demand of the capital, but also that of patients from neighboring states such as Mato Grosso, Amazonas or even Bolivia. This study was submitted for approval by the Research Ethics Committee (Comissão de Ética em Pesquisa - CEP) accredited by the National Research Commission (Comissão Nacional em Pesquisa - CONEP) with approval number 17/11(31/08/2011) and CAAE 0007.046.000-11. The Informed Consent Form (ICF) was filled out by each child's guardian stating that they agreed to participate in the study.
The study population comprised children from 0-6 years of age, who were treated for free and had characteristic signs and symptoms suggestive of an acute respiratory infection, after being clinically evaluated by a health professional in the period from February 19, 2013 to December 8 of the same year. A sample of 660 children attended at the emergency room of the Cosmo and Damião Children's Hospital with a frequency of two visits per week during the study period was selected for collection of respiratory secretion. Inclusion criteria were children aged 0-6 years of both sexes, attended at the emergency room of Cosme and Damião Children's Hospital, with clinical symptoms suggestive of acute respiratory infection such as cough, fever, coryza, pulmonary secretion, wheezing, dyspnea, ocular pruritus and otalgia according to the completion of the epidemiological investigation sheet prepared by the study group. They were excluded from the study children younger than 6 years, indigenous, chronic diseases or guardians who did not accept the study. The data collection was composed of 660 questionnaires with closed questions, which was divided into categories such as sociodemographic data and clinical manifestations.
The biological material used for the test were nasopharyngeal and oropharyngeal secretions obtained using a swab, preferably collected prior to treatment using the combined swab technique (nasopharynx and oropharynx), where a rayon swab was used (provided in the collection kit). The sample was accessed from the biological bank of nasopharyngeal and oropharynx secretions of the Molecular Virology Laboratory of the Oswaldo Cruz Foundation, Rondônia.
Extraction of the genetic material was performed using the QIAamp DNA Mini Kit and QIAamp Viral RNA Mini Kit (Qiagen, Germany) using 200 µL of nasopharyngeal secretion according to the manufacturer's instructions. The precipitated material was resuspended in 200 µl of elution buffer and stored at -20 ℃ until it was used. To avoid false-positive results, stringent procedures for nucleic acid amplification were followed [17].
For the identification of viruses with genetic material composed of RNA, a reverse transcription process was performed. Thus, the extracted RNA was previously denatured at 95 ℃ for 5 minutes, and converted to cDNA from 7.5 µl of RNA from the 200 unit extract of the SuperScript® III First-Strand Synthesis System for the RT-PCR (Invitrogen®) enzyme according to the manufacturer's instructions.
The selection of primers was based on complete genome sequences of the following viruses: Parainfluenza, virus 1 (PIV1), Parainfluenza, virus 2 (PIV2), Parainfluenza, virus 3 (PIV3), Adenovirus and Rhinovirus, deposited in the American database GenBank through the National Center for Biotechnology Information - NCBI. Using MEGA 6 software, the nucleotide sequences were aligned and mapped, while the conserved regions of each virus were analyzed. For the analysis of the in silico specificity of the primers, the Basic Local Alignment Search Tool (BLAST) in NCBI was used. To measure the in vitro specificity of the primers, the melting curve was analyzed individually using the SYBR® Universal PCR Master Mix system (Applied Biosystems). Optimization of the primers was performed taking into account two distinct gradients, a temperature gradient and a concentration gradient, in which, for the concentration gradient, the variation from 100 nM to 900 nM of each primer was used and for the temperature gradient, for the Delineation of the gradient, the melting curve of each pair of primers was taken into account. The primers used in the study were described by Hammitt, et al. [18] (Table 1).
Table1: Sequences of oligonucleotides. View Table 1
The samples were amplified on the ABI 7500 platform (Applied Biosystems, Foster City, CA, USA) using the SYBR Green® PCR Master Mix system. For this, 5 µl of the nucleic acid extraction was used in reactions with an initial primer concentration of 400 nM, 12.5 µl of SYBR Green® PCR Master Mix, with a final volume of 25 µl. Subsequently, a concentration gradient from 100 nM to 900 nM was created with the positive controls to select the best primer concentration based on the amplification and dissociation curve, with all standardization reactions being performed in triplicate. In order to confirm the results, the positive samples were submitted to a standard PCR followed by 2% agarose gel electrophoresis for visualization and verification of the band size, later purified using the QIAquick Gel Extraction Kit (QIAGEN), according to the manufacturer's manual.
The purified PCR products were quantified by spectrophotometry on a NanoDrop 1000 (Thermo Scientific), diluted to a concentration of 5 µg/µl to 10 µg/µl and sequenced on the Sequencing platform at the René Rachou Research Center/CPqRR - Fiocruz in Minas Gerais, Brazil. The sequences generated in this process were analyzed and edited using the software BioEdit 7.2.5 (Ibis Biosciences) to remove non-specific regions, and later the Basic Local Alignment Search Tool (BLAST) was performed to verify its specificity.
Exact Fisher's Test, Pearson Trend Chi-square test and p-value were used. Both were run in software R 3.4.1 (R Development Core Team 2017, Vienna, Austria). They were considered significant alpha values equal to or lower than 5%.
Six hundred and sixty children between the ages of 0 months and 6-years-old with a suspected acute respiratory infection participated in the study, of which 329 (49.85%) were female and 331 (50.15%) were male. Regarding the sociodemographic data of the guardians of the children with acute respiratory infection, 302 had an educational level between 10th and 12th grade (45.76%); when the family income of those responsible for these children is evaluated, 350 (53.03%) receive less than $284,00 per month. Table 2 shows the socioeconomic data of the guardians and of the children with acute respiratory infection.
Table 2: Socioeconomic data from children with acute respiratory infection. View Table 2
Table 3 presents the main clinical manifestations of children with acute respiratory infection. In this study the seasonality distribution was observed between the months of January and December 2013, during the period of the study. The monthly average of the maximum Relative Humidity (RH) and monthly average of the minimum relative humidity and precipitation were also evaluated. Of the 660 children with acute respiratory infection, it is clear that in April and May the infection rates were (14.1%) and (14.9%) respectively, but in April the precipitation was (365.8) and in May (98); the average maximum relative humidity in the month of April was (97.53%) and in the month of May it was (97.58%); the average minimum relative humidity in April was (66.96%) and in May it was (65.93%). A decline is observed from May to June in the cases of acute respiratory infection, (7.0%), in relation to the precipitation (29.4), the average maximum relative humidity (98%), and the mean absolute minimum humidity (58.63%). According to the data below, Graph 1 shows the distributions of seasonality between the months of January and December 2013 of children with acute respiratory infection (Figure 1).
Table 3: Clinical characteristics of children with acute respiratory infection. View Table 3
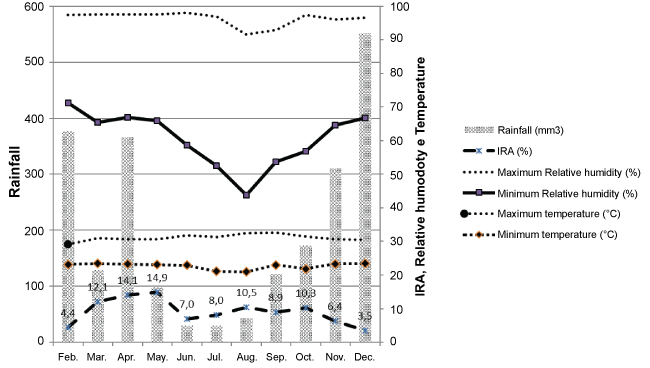 Figure 1: Distribution of seasonality in the months from January to December 2013. View Figure 1
Figure 1: Distribution of seasonality in the months from January to December 2013. View Figure 1
• For PIV1, PIV2 and PIV3 the concentrations of primers that showed the best results in standardization were 600 nM, 700 nM and 400 nM, respectively. For HRV the best concentration was 50 nM and for Adenovirus a concentration of 500 nM, with all reactions at a final volume of 25 µL. The dissociation temperature of the amplified samples was 83 ℃ for HRV, 73 ℃ for PIV1 and PIV3 and 76 ℃ for PIV2. Of the 660 samples, 161 were randomly selected and submitted to PCR. As a result, 113 were positive for at least one of the viruses proposed in the study and 48 were negative. Of the 113/161 cases confirmed for ARI, the most prevalent were males with 58 (52%) and 55 (48%) females. The following distribution was observed: 21.73% (35/161) were positive for Human Rhinoviruses, 14.28% (23/161) positive for Parainfluenza 1 and 44.72% (72/161) positive for Parainfluenza 2; Parainfluenza 3 was detected in 11.18% (18/161), Adenovirus in 10.55% (17/161) and 29.81% (48/161) were negative for all viruses analyzed (Table 4). Of the 113/161 positive patients for the viruses proposed in the study, 70 (62%) of the samples were classified as monoinfections. PIV2 had the highest incidence with 35/70 (50%) cases, followed by rhinovirus with 13/70 (18.57%), PIV1 08/70 (11.42%), PIV3 8/70 (11.42%) and adenovirus with 06/70 (8.57%) cases, according to distribution by gender in (Table 5).
Table 4: Result of the etiologic viral characterization. View Table 4
Table 5: Distribution of monoinfections by gender and etiologic agent. View Table 5
In the co-infection with 2 viruses, 34/113 (30%) patients were positive for the following samples: HRV/PIV1 03/34 (8.82%), HRV/PIV2 11/34 (32.35%), HRV/PIV3 01/34 (2.94%), PIV1/PIV2 10/34 (29.41%), PIV2/Adenovirus 03/34 (8.82%), PIV2/PIV3 04/34 (11.76%), PIV3/Adenovirus 01/34 (2.94), and HRV/Adenovirus 01/34 (2.94%). However, not all sexes had the same positive samples for the proposed viruses. On the other hand, HRV/PIV2 was the one with the highest incidence in both sexes, followed by PIV1/PIV2, according to gender specificity in (Table 6) below. As for a 3-virus co-infection, 9/113 (8%) patients were positive for the following samples: HRV/PIV2/PIV3 2/9 (22.22%), PIV1/PIV2/Adenovirus 2/9 (22.22%), PIV2/PIV3/Adenovirus 2/9 (22.22%), R/PIV2/Adenovirus 2/9 (22.22%), and HRV/PIV1/PIV2 1/9 (11.11%). However, not all sexes had positive samples for the proposed viruses, but all had the same incidence, except for HRV/PIV1/PIV2 with only 1 case, according to gender specificity in (Table 7).
Table 6: Distribution of coinfections by 2 viruses according to gender and pathogen. View Table 6
Table 7: Distribution of coinfections by 3 viruses according to gender and pathogen. View Table 7
Acute Respiratory Infections (ARI) are one of the main public health problems in children, mainly affecting children under the age of five, and one of the main causes of morbidity and mortality in this age group, especially in developing countries [5]. Six hundred and sixty-six children aged 0-6 years participated in the present study, where the highest frequency of ARIs occurred in male children 331 (50.15%). This fact may be related to the higher number of male children in the state of Rondônia, according to data from the Brazilian Institute of Geography and Statistics IBGE [19], which showed the total population of children aged 0-4 years old, with a total of 128,009 children, presenting 65,352 males and 62,657 females.
In a study conducted by Macedo, et al. [20], the prevalence in males was reported around 53.3%. The male gender, according to the literature, is most affected by the infections, which may be related to the lower caliber of the airways, which contributes to this phenomenon [10]. The cited studies are consistent with the current study in which there was a prevalence of males, but when the prevalence between ages is highlighted, the highest prevalence at the age of 1-3 years are of female children. A study by Silva, et al. [21] showed the relevance of children with ARI exposed to smoking at home. 21 mothers participated in the study; 8 of them declared to be smokers; in 3 cases the father was also a smoker, and in only 1 domicile was the father the only smoker. This result shows that secondhand smoke increases the incidence of ARIs in children. The amount of cigarettes smoked by the guardians of the children in this study was not observed, but it is important information that helps to know how much children are exposed to secondhand smoke. This study is consistent with data from the current study, since there was a prevalence of children exposed to secondhand smoke, but there is no statistical significance regarding the incidence of ARIs in relation to secondhand smoke.
It is reported that familial agglomeration related to dwellings and the amount of rooms is a risk factor for children acquiring acute respiratory diseases [22]. In the study conducted by Silva, et al. [21] with 21 children, 15 slept in the same bed as their parents. Children with a history of respiratory tract disease and submitted to poor housing conditions, with a greater number of people sleeping in the same room, have a higher incidence of ARI. In this study, the prevalence was observed in 2-4 (51.97%) children who sleep with their parents. This prevalence is significant in the age group of 5 years and under, mainly in children under 1-year-old, being around 55.08%. The environment in which the child lives is important, since it is a factor that contributes to preventing respiratory diseases, such as: controlling the humidity inside the house, allowing ventilation and sunshine, and avoiding collections of water, thus preventing children from being affected by respiratory disease [3]. Compared with the data from this study, most of the households had between 2-4 windows (53.91%) showing similarity with the study performed by Silva, et al. [21] in which most of the mothers had ventilated houses.
The family income of children under the age of five is related to the occurrence of ARIs, since better living conditions for children depend on the good economic conditions of their parents. There are reports that children of families that receive less than three minimum wage salaries have greater risks for these diseases than families receiving their income from more than five minimum wage salaries [23,24]. According to a study carried out by Oliveira, et al. [25], the family income of children with ARIs was between 1 and 2 minimum wage salaries (58.09%). Based on data collected in this study, the prevalence of family income was lower than one minimum wage salary (53.03%), being significant among all age groups, more prevalent among children under the age of 1 (58.98%). Maternal education level is another risk factor for children to develop ARIs. Based on the data presented, maternal education levels below 10th and 12th grade were observed more frequently in children the age of 1-3 years old (47.77%). This factor is probably due to the fact that the mother cares for the child in a way that may be incorrect or even because of a lack of knowledge about the proper way to care for the child [24,26]. Children under one-year-old presented a prevalence of cough compared to all symptoms and ages, with 167 (67.23%). Children from 1-3 years old presented most of all symptoms in relation to the other ages. The study conducted by Thomazelli, et al. [27] was not different from this study; children in the age range between 18 days and 4-years-old had a cough as their most common respiratory symptom, around 86%. Bonfim, et al. [10] also observed the predominance of symptoms which were 93.2% coryza and 58.2% cough, in children in the age group between < 24 months and > 24 months, who demonstrated an absence of severe symptoms due to ARI. This study showed cough as the prevalent symptom in all age groups.
A study conducted by Souza; Cardoso; San`T Anna [28], with children under the age of 5 who were hospitalized for respiratory diseases, showed a prevalence in the under-1 age group for hospitalization, with a cough being the most prevalent symptom (80.8%), followed by fever (66.2%) and dyspnea (65.4%). This study was similar to the current study, since cough was prevalent in (75.4%), followed by fever (60.40%) and dyspnea (46.31%) in 1-year-old children with respiratory infection. Among several factors contributing to ARIs, environmental factors such as air pollution and climatic variables also proved to be important factors for increasing the number of cases and their severity in children under the age of five [29]. In Porto Velho the predominant climate is tropical and humid, characterized by warm weather and a high level of relative humidity [6].
The cases of infection prevalence in Porto Velho - RO in the year 2013 from February to December were in the month of May (14.9%), showing a sharp fall in June (7.0%) which may be related to the maximum relative humidity and precipitation, since when there is rainy weather people gather in their homes which is a factor contributing to infection; however, the maximum relative humidity remains high in the months of May and June (97.58%) and (98%), respectively, and precipitation is not high compared to the month of June (29.4), but in May the precipitation was (98); that is, when precipitation decreased, people stopped staying in their homes, thereby reducing cases of transmission, causing a decrease in cases of acute respiratory infection. In the study conducted by Dal Moro, et al. [30] in Montes Claros - MG, the incidence of ARI is influenced by climatic conditions, with variation according to the climate. In the dry season, there were 10% more hospitalizations due to ARI than in the rainy season. Viral pathogens are responsible for 30-40% of ARI cases, most frequently HRSV, HPIV 1, 2, 3, HRV, Adenovirus [10]. Of the 660 children who participated in this study, 161 were classified with suspicion of acute respiratory infection, with 113/161 (70%) cases confirmed and 48/161 (30%) discarded. Of the 113/161 cases confirmed for ARI, 58 (52%) patients were male and 55 (48%) were female, making males the most prevalent, coinciding with data in the literature.
The age range of the children present in the study goes from 0-6 years old, with the highest incidence in the range of 0 to 11 months, with 47 (41.60%) cases, followed by 1-year-old children with 27 (24%), 2-year-olds with 22 (19.40%), 3-year-olds with 9 (8%), 4-year-olds with 3 (2.65%), 5-year-olds with 3 (2.65%), and 6-year-olds with 2 (1.7%) cases. Children aged 3-6 years old were less affected by ARI-causing viruses. These data coincide with those published in the literature, with a variation in the peak age of occurrence depending on the agent involved and the local epidemiological conditions. All 161/660 patients with suspected ARI were tested for the 5 respiratory viruses proposed in the study in the period from February to December 2013. The 113/161 ARI-positive patients provided a total of 165 positive samples for the following pathogens, including monoinfection and coinfection by 2 and 3 viruses: 21.21% (35/165) HRV, 13.93% (23/165) PIV1, 43.63% (72/165) PIV2, 10.90% (18/165) PIV3, and adenovirus at 10.30% (17/165). PIV2 was the most prevalent virus, followed by HRV. During the period from July 2003 to September 2005, BONFIM, et al. 2011 [10] conducted a case study of ARI in children at a day care center in the city of São José do Rio Preto - SP, with 176 children aged 4-72 months, constituting 100 boys and 76 girls. A total of 782 samples were tested, of which 294 (37.6%) were positive for: PIV1 9 (3.1%), PIV2 and PIV3 10 (3.4%), and HRV 111 (37.7%). The highest incidence occurred in males with 70 (56.4%) cases and 54 (43.5%) female cases. The most prevalent virus in both sexes was the HRV. HRV was detected during all the seasons of the year, but more frequently in the fall, and with low frequency in the summer. It has been observed that in the summer for many years, cases of ARIs have decreased. Viral pathogens account for 30-40% of ARI cases.
The types of infections were classified as: monoinfection with 70 (62%) cases, coinfection with 2 viruses 34 (30%) and coinfection with 3 viruses 09 (8%). The monoinfection had PIV2 as its prevalent pathogen with 35/70 (50%) of the cases, followed by the other viruses proposed in the study, as specified in (Table 1). Coinfection by 2 viruses, had a higher incidence with Rhino/PIV2 11/34 (32.35%), followed by the other viruses proposed in the research. However, not all the sexes had positive samples for the proposed viruses, as specified in (Table 2). In coinfections with 3 viruses, not all sexes had positive samples for the proposed viruses, but all had the same incidence, with the exception of R/PIV1/PIV2 with only 1 case, as specified in (Table 7).
Males had PIV2 as the most prevalent virus, followed by rhinovirus, PIV1, PIV3 and adenovirus. PIV2 was most prevalent in the age group from 1 month to 11-months-old, reaching all ranges in a different incidence, with the exception of the 6-year-old group (with no cases). Rhinovirus (11 cases), PIV1 (07 cases), and PIV3 (05 cases) were also prevalent in the group of 1 month to 11-months-old. Adenovirus, on the other hand, had its highest incidence in 2-year-old children with 03 cases. In the 4-6 year old age group, there was only one episode at the age of 4-years-old with R/PIV2, 5-years-old with R/PIV2/A and 6-years-old with PIV3. Coinfection with 2 viruses was prevalent around 2 months to 1-year-old. Coinfection with 3 viruses was prevalent in children aged 1 to 2-years-old. In females, PIV2 was also the most prevalent virus, followed by rhinovirus, PIV1, adenovirus and PIV3. PIV2 was more prevalent in the age group of 1 month to 1-year-old with 24 cases. Rhinovirus (07 cases) and PIV3 (04 cases) were prevalent in the age range from 1 to 11-months-old, with the highest incidence of these viruses in this age group. PIV1 was prevalent in 3-year-old children, with 4 cases. Adenovirus was prevalent in 2-year-olds with 03 cases. PIV2 affected all age groups, but with a different prevalence, with a lower incidence among children aged 3-6 years old. HRV did not affect the age range of 5-6 years old. PIV3 did not affect the age range of 3-6 years old. PIV1 did not affect the 4-year-old age group. Adenovirus did not affect the 3-year-olds. Coinfection with 2 viruses was prevalent in children aged 1-2 years old. Coinfection with 3 viruses was prevalent in children around 2-years-old, with R/PIV2 (03 cases).
This study concludes that some of sociodemographic factors and climate could be risk factors for acute respiratory infection, especially in children under three-years-old. It seems to be that agglomeration, low salary, and low education level of the guardian are factors associated with presence of acute respiratory infections in this population.
Males were most affected, with high number of incidence in range from 0-3 years old, but it is impossible to assert that these data are statistically significant. The city of Porto Velho had the highest number of cases, especially in the months of May and June, which are the hot months in the region. PIV2 and Rhinovirus were the main etiological agents causing ARI, classified as mono infection and co infection with 2 and 3 viruses, coinciding with data from the literature.