Human papilloma virus (HPV) infection is the major etiologic agent of cervical cancer. Both human papillomavirus (HPV) and human immunodeficiency virus (HIV) are classified as carcinogens. We describe the prevalence and distribution of HPV genotypes among HIV-positive and -negative women aged 20-37 years-old in Côte d'Ivoire.
Women were recruited among those who come to the gynecology department of the Military Hospital, Abidjan with a positive acetic acid test. HIV testing was performed on the basic of the Retrovirus Côte d'Ivoire testing algorithm. HPV DNA was detected with the Linnear Array HPV Genotyping Test (Roche Diagnostics, Indianapolis, IN, USA).
A total of 105 women were recruited. DNA testing was done on 87 sample specimens who had a positive beta globin. Fifty-five women (55) were positive for HPV, giving an overall prevalence of 63.2%. The HPV rates was higher among HIV-positive women (39.1%) than HIV-negative women (24.1%). The most prevalent HPV were 16 (20%), 18 (12.7%). Infections with multiple HPV types were more common in HIV+ (45.5%) than HIV- (12.7%). HPV high risk (HR) accounted for 72.7% of the genotypes detected.
Prevalence of HPV and HR types were high in young women. Screening strategies incorporating HPV genotyping and vaccination should be effective in preventing cervical cancer in both HIV-positive and -negative women living in Côte d'Ivoire.
Cervical cancer, HPV, HIV, Genotype
Papillomavirus (PV) are members of the distinct virus family, the Papillomaviridae, divided into 39 genera [1,2], based on L1 sequence identity of 60% or greater, with each genus designated by a letter of the Greek alphabet. More than 300 papillomaviruses have been identified and completely sequenced, including over 200 human papillomaviruses (HPV) [3]. PVs within a genus that share 60-70% L1 sequence identity are termed a species [1,2].
Although the vast majority of HPV, some genotypes can lead to the development of cervical cancer (CC) [4]. There is a classification according the carcinogenic risk. So, high-risk (HR), probably carcinogenic or probably high-risk (pHR) and low-risk (LR) types are described [5].
Routine screening is critical for the early detection of HPV and prevention of associated diseases and cancer. There are 3 different types of screening tests that are currently recommended by WHO: HPV DNA testing for high-risk types, visual inspection with Acetic Acid (VIA) or conventional (Pap) test and liquid-based cytology (LBC) [6].
Nevertheless low-income countries do not have access to routine screening as recommended by WHO. Therefore, incidence and mortality countries is very high in these [7].
Risk factors for cervical carcinoma include tobacco use, young age of sexual debut, multiple sexual partners, high-risk sexual partners, history of other Sexual Transmitted Infections (STIs), and a history of genital carcinoma and human immunodeficiency virus (HIV) and immunosuppression [8,9].
In Côte d'Ivoire, CC is the second most common cancer among women [10]. In 2020, it was estimated a total of 2067 new cases and 1417 deaths, with an age-standardized incidence and mortality rates of 31.2 and 22.8 per 100,000 women, respectively [10]. This led the government to put in place a policy to combat CC and other HPV-related infections, including early detection and vaccination.
Recent data on the circulation of HPV genotypes in women date from 2012 and the study population was women of 25-65 years-old. A high prevalence of oncogenic HPV was found in women with no cervical neoplastic lesions, especially in HIV-positive women, 59.1% [11]. Because HPV infection cause CC incidence to increase among younger women [12], we focused our attention on young women aged 20 to 37-years-old. The aim of the present study was firsly to document the prevalence of HPV in and secondly to identify genotypes in this specific population to reduce the burden of this infection by raising awareness of early detection and vaccination.
Women aged 20 to 37 years who come to the gynecology department of Military Hospital (Abidjan) are recruited from April 2017 to May 2018. All women underwent pre- and post-test counselling and signed informed consent form prior to participation in the study. This study was approved by the national ethics committee for medical research in Côte d'Ivoire.
After obtaining written informed consent from the women and collecting participant's history, gynecological examination and visual inspection with Acetic Acid (VIA) were conducted as previously described [13].
HIV serology was performed on the basic of the Retrovirus Côte d'Ivoire (RETROCI) HIV testing algorithm using the three rapid assay confirmatory strategies for the detection of antibodies to HIV as previously described [14].
The MagNAPure LC 2.0 system (Roche Diagnostics GmbH, Mannheim, Germany) was used for DNA isolation from 200 μl of cervical samples with MagNAPure LC Total Nucleic Acid Isolation kit (Roche Diagnostics GmbH), following the instructions of the manufacturer. Nucleic acids were eluted in 100 μl of elution buffer and 50 μl were used for PCR. The integrity of the DNA samples was ascertained by positive amplification of human DNA β-globin control. Human Papillomavirus amplification, hybridisation and detection were performed with the Linear Array HPV Genotyping Test following Roche instructions (Roche Diagnostics GmbH). HPV DNA was detected with the LINNEAR ARRAY Human Papilloma Virus GENOTYPING Test of Roche (www.roche.pl), which enables a simultaneous amplification of the target HPV DNA and of beta-globin DNA- the latter providing an internal cell control. The test employs pairs of primers, which detect nucleotide sequences within L1 polymorphic region of HPV genome. A pool of HPV primers, included in the test, encompasses 37 HPV genotypes, including 15 high-risk genotypes: 6, 11, 16, 18, 26, 31, 33, 35, 39, 40, 42, 45, 51, 52, 53, 54, 55, 56, 58, 59, 61, 62, 64, 66, 67, 68, 69, 70, 71, 72, 73, 81, 82, 83, and 84.
The statistical analysis was performed with IBM SPSS Statistics for Mac, Version 24.0 (Armonk, NY: IBM Corp) using Chi-Square (χ2) to compare the categorical variables with a 5% significance level.
A total of 175 femmes were recruited, 105 had a positive acetic acid test and DNA testing was done on 87 sample specimens who had a positive beta-globin. Fifty-five women (55) were positive for HPV, giving an overall prevalence of 63.2% (Table 1). HPV infection rates were 53.3%, 48.7% and 44.4% for the age groups [20-25], [25-30] and [30-37] respectively. According HIV statute 39 were HIV+ and 48 HIV- giving 87. The HPV infection rate was 39.1% in HIV+ and 24.1% in HIV- women (Table 1).
Table 1: HPV prevalence according age and HIV statute. View Table 1
Of the 37 targeted genotypes, 20 were identified. The six most prevalent HPV were16 (20%), 18 (12.7%), 33 (5.4%), 35 (5.4%), 52 (5.4%), 68 (5.4%). These six types constituted about 64.3% of the total. Their distribution is given in Figure 1. HPV+ women (55) included HIV+ (36) and HIV- (19). In HIV+, all genotypes were found excepted 70, 16 (10.9%), 18 (10.9%), 33, 35, 58, 68, 73 with 3.6% each and the the others less than 2% each (Figure 1). In HIV- women, genotypes 56, 59, 68 and 73 were not identified, 16 (9.9%), 52, 70 with 3.6% each and the others less than 2% each (Figure 1).
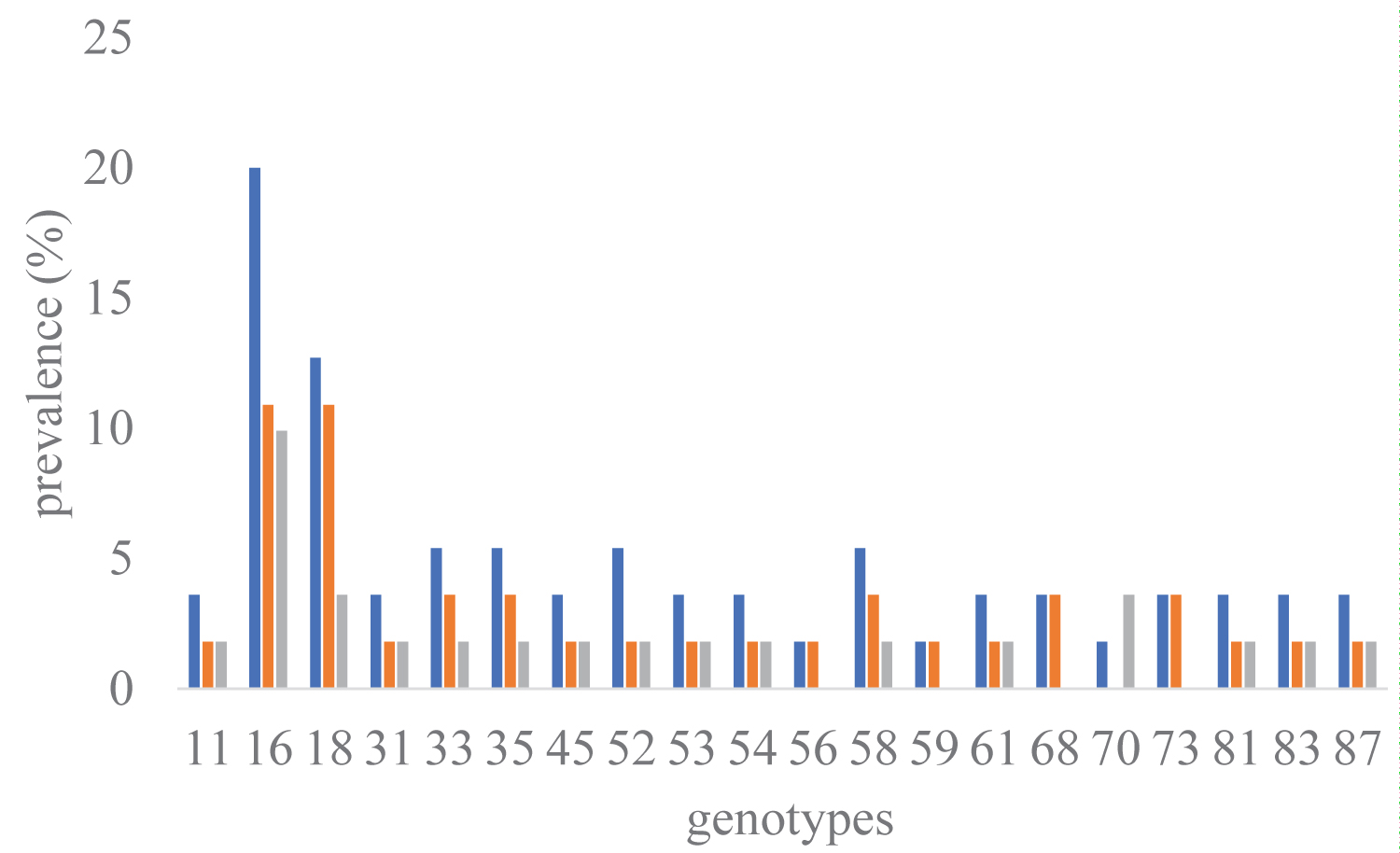 Figure 1: Prevalence and distribution of HPV genotypes according HIV statute.
Figure 1: Prevalence and distribution of HPV genotypes according HIV statute.
Prevalence of each subgroup was calculated from 55 sample specimens who had a positive HPV DNA test. For each genotype, the prevalence of the HIV+ (red bars) and HIV- (green bars) subgroups are calculated and then summed to give the total (blue bars).
View Figure 1
Single HPV type was calculated 41.8% (23 cases), multiple HPV type was calculated 58.2% (32 cases). Multiple types were detected in HIV+ and HIV- women respectively 45.5% and 12.7% of case (Table 2). Of the 15 targeted high-risk (HR), 12 genotypes were found consisted in 72.7% (40) cases of all genotypes detected, 49% (27) for HIV+ and 23.6% (13) for HIV- (Table 2). Genotypes 16 and 18 were calculated 47.5% (19) of HR types.
Table 2: HPV prevalence in women with HIV or without according the types of infection and genotypes. View Table 2
The study of the prevalence allows to assess the burden of an infection in a population and to appreciate the effectiveness of prevention policies. The prevalence of HPV obtained was 63.2% and is close to that reported, 63.0% in developing countries [15] and among women in Abidjan, Côte d'Ivoire [16].
Higher rates have been reported by several authors. Forman, et al. [17] on his global based review reported close to 90% prevalence of HPV in women with different cervical abnormalities including neoplasia. Besides, a review by Peng, et al. [18] conducted to demonstrate the HPV genotype among Asian women reported HPV prevalence at 84.8% from different kinds of advanced cervical lesions. Furthermore, other review studies from different corner of the globe reported the following ranges of HPV prevalence from different grades of cervical dysplasia; 87.2% (Burkina Faso) [19], between 70 and 80% (North America) [20], 78.1% (Central and Eastern Europe) [21] and between 64.8 and 90.1% (Thai) [22]. The prevalence of HPV from Ethiopian woman with different kinds of cervical dysplasia was between 67.8 and 93% [23]. In Côte d'Ivoire, the frequency of infection with HPV was 72.4% [11]. In contrast, the Nigerian authors reported lower rates, 17.3% [24]. The difference regarding the report from countries could be explained by a population difference in which these studies might be included woman with cervical abnormalities [25].
The data collected showed that Côte d'Ivoire is in a zone of high endemicity. This is a good opportunity to reinforce the prevention policies through information, education, communication and vaccination to reduce HPV burden [26].
The test used in the study allowed the identification 37 HPV genotypes, including 15 high-risk genotypes (HPV-16, 18, 31, 33, 35, 39, 45, 51, 52, 56, 58, 59, 68, 73, and 82). Several global meta-analyses on the distribution of HPV genotypes have confirmed the five most prevalent strains in women with normal cytology [27] and cervical neoplastic diseases [28,29] to be HPV-16, 18, 31, 52, and 58. HPV16 and 18 were identified as the predominant oncogenic genotypes, causing approximately 70% of global cervical cancer cases [8,28]. Regarding the HPV genotypes, we found that the most prevalent were HPV-16 (20%), HPV-18 (12.8%). Data are similar than which reported in a systematic review and meta-analysis of Molecular epidemiology of human papillomavirus among HIV infected women in developing countries [15]. In Africa, as in other parts of the world, the dominant HPV type associated with invasive cervical cancers is HPV-16 [30]. In 515 cancers from Ghana, Nigeria, and South Africa, the six most prevalent HPV types were HPV-16 (51.2%), HPV-18 (17.2%), HPV-35 (8.7%), HPV-45 (7.4%), HPV-33 (4.0%) and HPV-52 (2.2%) [31]. Some factors that could be responsible for variations in the distribution of HPV types across the world include: The type of assay used, multiple HPV infections, differences in the study population, and varying exposures of individuals to different risk factors in different geographical regions [32].
The review conducted to estimate prevalence of HPV genotype among African women, revealed that HPV 16, 52, 35, 18, 58, 51, 45, 31, 53, and 56 were the ten most common genotypes in the normal cervical cytology while HPV 16, 18, 35, and 52 were the four common types [33]. Another review from Ethiopian women depicted that HPV 16, 52, 18, 58, 45 were top five genotypes with the proportion of 45.3%, 9.4%, 8.2%, 6.9%, 5.2%, respectively [23]. The data obtained are distinct from those reported by Nejo, et al. [24], HPV-31 as the most predominant (32.8%), followed by HPV-35 (17.2%) and HPV-16 (15.5%). Ivoirian authors reported in HIV-positive women, HPV 35 (15.7%), 16 (14.2%), 18 (11.4%) and 58 (11.4%) were the four most common HPV types identified [11].
Genotypes 16 and 18 are high risk-HPV and commonly implicated in cervical malignancies [8]. Genotype 16 was isolated alone in 6/55 (10.9%) or associated with genotypes 18 in 3/55 (5.4%). Genotype 18 was isolated alone in 3/55 (5.45%) or associated with genotype 16 in 3/55 (%) of cases. Higher rates have been reported by several authors. In Ethiopia, the combined prevalence of HPV 16/18 was at 53.6% [23]. A review by Ogembo, et al. [33] showed HPV 16/18 at 45.1% from high-grade cervical lesions and 67.7% of invasive cervical cancer among African women. Among HPV positive cases, the co-prevalence of HPV 16/18 was reported 60% (in Israel) on both pre-neoplastic lesions and cervical cancer [34,35], 87.5% (in Central and Eastern Europe), [21], 80% (India) among high grade cervical lesions [36].
HPV 16 is clearly the most potent, followed by HPV 18. This knowledge is consistent with HPV vaccine formulations and has led to approval of screening tests that include at least HPV 16/18 genotyping to improve risk stratification among HPV-positive women in Côte d'Ivoire.
HIV and HPVs are both sexually transmitted and are also the cause of significant public health problems in Africa. In the genital tract the interaction between these two viruses is complex, infection with multiple HR-HPV types reported to make women more susceptible to HIV-1 infection [37], and HIV-infected individuals are more likely to have HPV infection [38]. In West Africa, Cote d'Ivoire has the highest HIV prevalence among women, 4.1% [39]. The data obtained showed that HIV+ women were 1.6 times more infected with HPV than HIV-negative women (p < 0.01). Our results are consistent with the findings from other studies that showed women with HIV infection were more likely [8,30,40].
The high prevalence of high-risk type is a cause for concern because persistent infections with these types have been recognised as a necessary cause for cervical cancer [8]. The prevalence of high-risk HPV (HR-HPV) type was higher than the low-risk type, 40/55 (72.7%) vs 10/55 (27.3%). Similar results of higher prevalence of HR-HPV have been previously reported in Nigeria [24,41]. Related findings have also been documented in some other African countries [42-45].
On the other hand, there is no significant difference between the different subgroups according to HIV statute and high or low risk HPV. This finding is not consistent with results from other studies from Africa and elsewhere [46-49].
The high prevalence of high-risk HPV types detected in this study even in multiple infections reveals the burden of HPV infection in Côte d'Ivoire, particularly in HIV infected women. Multiple HPV types detected with HPV 16 and 18 dominating, indicated that HPV-16 and 18 might be the major circulating types in Côte d'Ivoire and the available vaccines in the country may be effective in controlling the infection. These data will persuade policy-makers to promote strategies such as cervical screening and HPV vaccination to reduce the incidence of cervical cancer in this population.
We would like to acknowledge the Ministry of Defense and the authorities of Military Hospital, Abidjan for all authorisations and facilitations, the National Public Health Laboratory for its technical support.
Authors reports no conflict of interest related to this work.
The information presented in this paper represent the views and opinions of the authors and must not be taken as representing the opinions or views of any organizations that they work for or represent in other ways.
No funding support for this research.