Concerning two cases of atypical toxoplasmosis and progressive multifocal leukoencephalopathy coinfection in HIV-positive patients and due to the clinical and prognostic relevance of both entities, we decided to make a review of space-occupying lesions in the CNS in HIV patients.
Both patients presented neurological symptoms and atypical intracranial lesions on imaging tests. On admission they were diagnosed with advanced stage HIV, so we made the differential diagnosis with the most frequent opportunistic diseases. To do this, we use a systematic review of the literature in PubMed. The pathologies reviewed include: Cerebral toxoplasmosis, PML, primary CNS lymphoma, histoplasmosis, tuberculosis or cryptococcal infection.
Neurological manifestations in HIV patients can appear at any stage of the disease. We find several possible etiologies within the space-occupying lesions of the CNS: Infectious, inflammatory or tumor. The immunological status of the patient, the presence of other concomitant diseases and the epidemiological environment, mainly the place of origin and travel, will be essential to approach the etiology [1]. Since the introduction of highly active antiretroviral therapy (HAART), this type of injury has reduced its incidence [2]. However, today they still represent a pathology that carries significant morbidity and mortality, so it is essential to carry out an adequate differential diagnosis and an early diagnosis and treatment to improve the prognosis [3].
Among the most frequent etiologies of space-occupying lesions are toxoplasmosis, primary CNS lymphoma, progressive multifocal leukoencephalopathy (PML), HIV encephalopathy and CMV encephalitis, without forgetting other relatively frequent causes such as tuberculosis or cryptococcal infection. The differential diagnosis of space-occupying lesions in HIV patients is often a challenge for the clinician, since many of them can present in a similar way, both in clinical and radiological manifestations. Due to their similarity, those that most frequently cause difficulty in diagnosis are: Toxoplasmosis and primary CNS lymphoma [4,5].
In this article we are going to present two unknown HIV infected patients with atypical cerebral toxoplasmosis at presentation that entailed a diagnostic challenge for the medical team.
A 65-year-old man, Spanish, smoker and occasional cocaine user, with a history of bladder cancer cured for more than 15 years and without regular treatment who comes to the emergency department brought by his family due to confusion and behavioral disturbances. Clinical case began about 3 weeks before, with progressive deterioration. According to his family, from the beginning of the condition, the patient presented generalized weakness, confusion, sleep disturbance and progressive clumsiness that prevented him from carrying out his usual tasks, he also associated bradypsychia, inappropriate language, strange behavior and gait alteration with lateralization to the right side. He had not presented fever at any time or other symptoms except for loss of 4 kg of weight in 2 months. On examination, the patient presented oral candidiasis and ataxia in the right hand with dysdiadochokinesia.
A cranial CT scan did not show significant lesions, an urgent EEG observed signs of focal involvement in the temporal region of the right hemisphere, and with this results he was admitted into the hospital to complete the study. Once admitted, serologic tests were requested with a positive result for HIV infection (viral load log 6.4, TCD4 cell count 95 (15%), and CD4/CD8 0.28. Other relevant results were: Positive CMV IgG and positive IgG Toxoplasma with negative IgM. Other serologic tests including antibodies for HCV, anti-HBs and anti-HBc, were negative. The study for tuberculosis was negative as well as for other opportunistic infections.
Due to the neurological symptoms and the diagnosis of HIV, an MRI and a lumbar puncture were performed. Magnetic resonance imaging revealed hyperintense lesions in the subcortical white matter in T2 and in diffusion-weighted sequences with two different patterns, on the one hand a patchy lesion in the subcortical white matter in the left frontal region and on the other hand a pattern punctate located in the left frontal and bilateral parietotemporal subcortical white matter, which are reinforced with contrast, being compatible with PML or lymphoma. In the lumbar puncture, a clear fluid was obtained with mild proteinorrachia and positive results for EBV PCR (1480 IU/ml) and JC virus PCR (89 copies/ml). ART with abacavir/lamivudine plus dolutegravir QD was initiated as PML and HIV treatment.
Due to neurological worsening of the patient with the appearance of right hemineglect, dysarthria, motor aphasia, as well as worsening gait, and due to a positive IgG result for toxoplasma in an HIV patient with CD4 < 200, despite CSF with negative PCR, we decided to start treatment for cerebral toxoplasmosis with atypical presentation with Sulfadiazine 1500 mg every 6 hours associated with Pyrimethamine 75 mg every 24 hours (and folinic acid) (Figure 1).
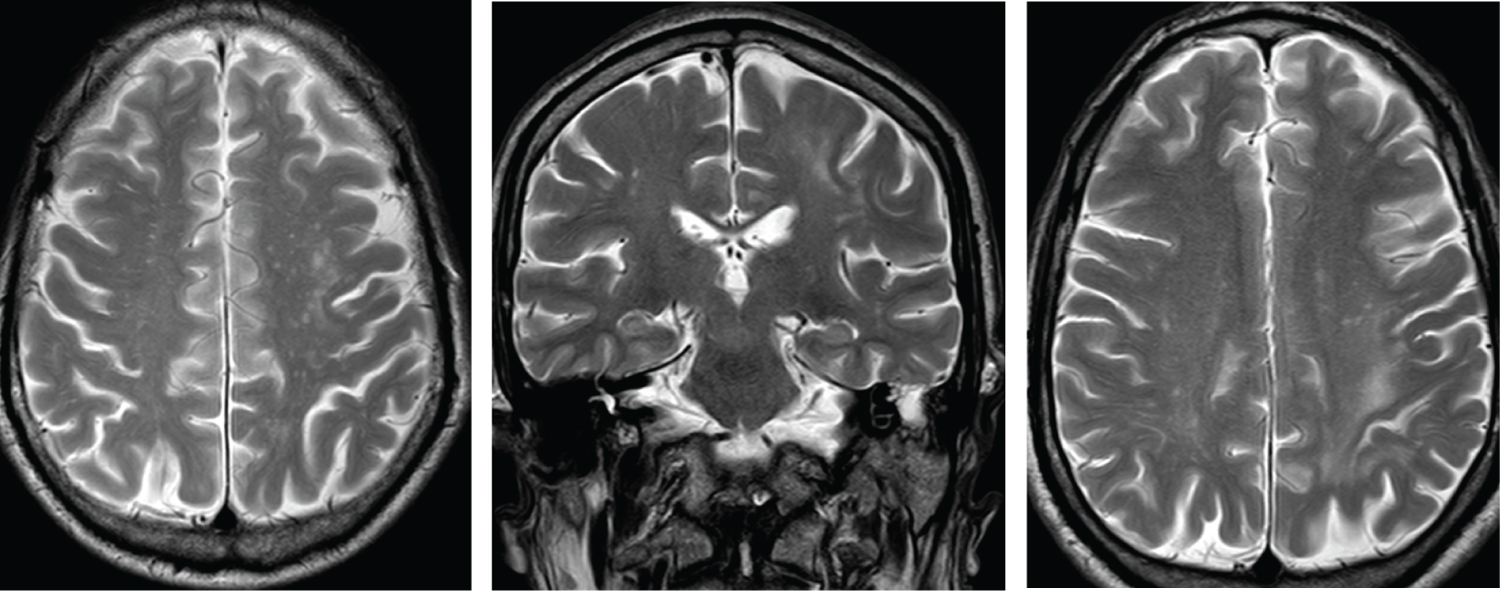 Figure 1: MRI images with hyperintense lesions in the subcortical white matter in T2 with two different patterns: patchy lesion and a pattern punctate located.
View Figure 1
Figure 1: MRI images with hyperintense lesions in the subcortical white matter in T2 with two different patterns: patchy lesion and a pattern punctate located.
View Figure 1
Meanwhile, we expanded both radiological and cerebrospinal fluid studies to try to reach the diagnosis. We performed a CSF cytology with no malignant cellularity and a CSF flow cytometry that did not detect pathological lymphocytes, it only detected cells compatible with a reactive process. We performed a Thallium scintigraphy which did not show uptake (lymphoma was not compatible).
From day 11 after starting toxoplasmosis treatment, the patient presented spectacular clinical improvement with practical resolution of all neurological signs and symptoms. Subsequent brain MRIs show remarkable radiological improvement. Four months after admission, the patient is fully recovered.
A 53-year-old man, Spanish, smoker, with anxiety disorder treated with antidepressants and a history of primary syphilis treated with Penicillin in 2009, came to the emergency department due to language impairment, clumsiness in the right hand and gait alteration with lateralization to both sides that had started two weeks before. In addition, he associated weight loss of 15 Kgs in the last year. On examination, we appreciated a moderate dysarthria, mild facio-brachio-crural hypoesthesia and dysmetria of the right arm, as well as subtle gait disturbance.
A head CT scan was performed, which did not show intracranial lesions. Upon admission, serologic tests were requested with a positive result for HIV infection (viral load of 122,000 copies/ml), TCD4 cell count 128 (13.6%), and CD4/CD8 0.28. Other relevant results were: CMV IgG positive with the rest of serologic tests, including toxoplasma serology, negative. As in the previous case, the tuberculosis study combined with PPD skin test and QuantiFERON was negative. Subsequently, an MRI was performed where a focal lesion hyperintense in T2-Flair without enhancement after administering gadolinium was observed in the right cerebellar subcortical white matter that was considered PML as the first possibility, a diagnosis that was supported by the images obtained with a thallium scintigraphy. In addition, a lumbar puncture was performed to complete the study. The biochemical analysis did not show significant alterations, the cultures and microbiology samples, including EBV and toxoplasma PCR, were negative, except for JC virus PCR (669 copies/ml) (Figure 2).
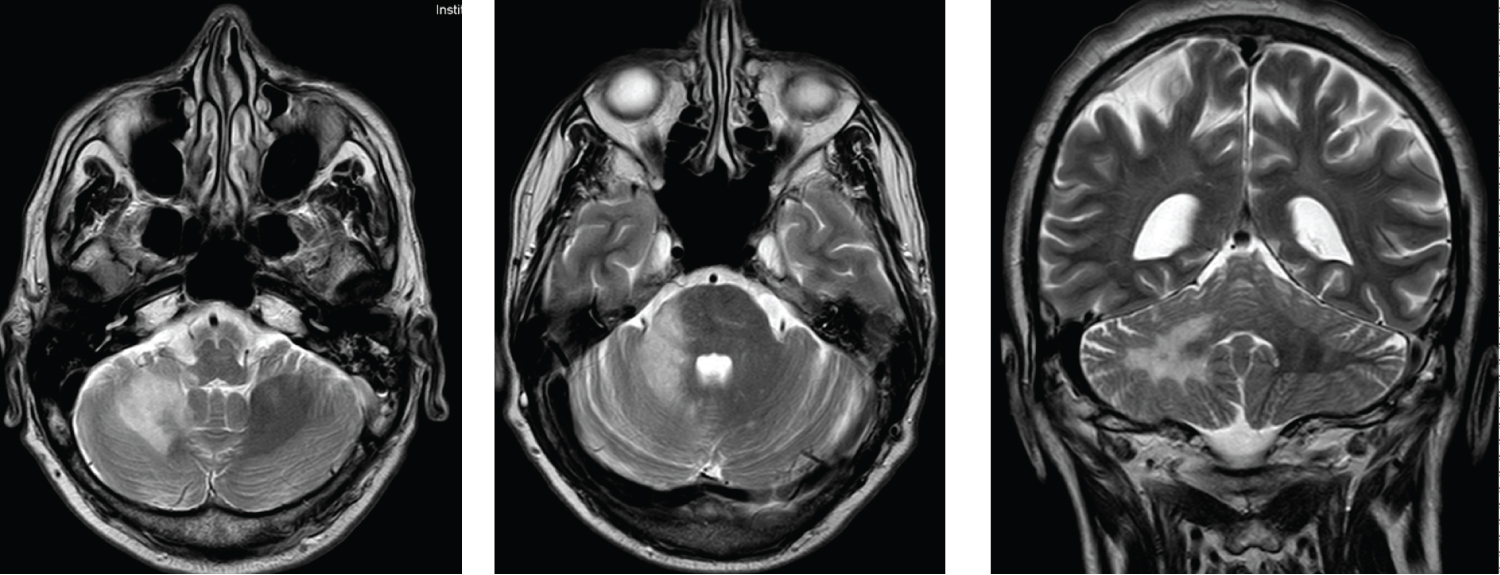 Figure 2: MRI images with hyperintense lesions on T2 in right cerebellum hemisphere.
View Figure 2
Figure 2: MRI images with hyperintense lesions on T2 in right cerebellum hemisphere.
View Figure 2
The patient was discharged from the hospital with a presumptive diagnosis of PML in a patient with late HIV infection and he started antiretroviral treatment (TAF/FTC/BIC) without presenting neurological improvement.
Six weeks later, the patient returned to the hospital with neurological deterioration. He presented inability to walk, intelligible speech due to severe dysarthria and right hemineglect. At this time, 6 weeks after starting HAART, the patient presented an improvement in the parameters of HIV infection with a viral load of 36 copies/ml, CD4 264 (11.5%), and CD4/CD8 0.22. The brain resonance was repeated and showed radiological worsening of the intracranial lesions with greater number and extension of them. With the improvement at the immunological level with HAART, we repeated all the serologic tests with an actual positive result for toxoplasma IgG, the rest remained negative. Likewise, we did a new lumbar puncture with PCR for toxoplasma and JC that persisted negative and we extended the study of the primary lymphoma with cytology and flow cytometry with negative results.
Upon admission, and while we waited microbiological results, corticosteroid treatment (dexamethasone) was started for possible IRIS in relation to the initiation of ART that could justify the clinical worsening. With this treatment, the patient presented minimal improvement in speech.
Due to this neurological deterioration and the result of positive IgG for toxoplasma in an HIV patient with CD4 < 200, we decided to start treatment for cerebral toxoplasmosis with Sulfadiazine 1500 mg every 6 hours associated with pyrimethamine 75 mg every 24 hours and folinic acid. At treatment day 12, the patient presents a spectacular clinical improvement with practical resolution of neurological symptoms at discharge, being able to walk without help and with normalized speech. Currently, he only presented slight dysmetria in the right arm with the rest of the symptoms resolved and he is awaiting a new evaluation MRI at outpatient consultation.
We present two cases of atypical toxoplasmosis in patients with HIV infection in an advanced stage of immunosuppression in which a widespread differential diagnosis had to be made between the most frequently CNS space-occupying lesions, mainly, cerebral toxoplasmosis, primary brain lymphoma and progressive multifocal leukoencephalopathy.
Toxoplasmosis is the most common infection of the central nervous system (CNS) in patients with HIV/AIDS who do not receive adequate prophylaxis and continues to be the most important cause of space-occupying lesions of the CNS in HIV patients [3,6,7]. It is caused by a worldwide intracellular protozoan parasite called Toxoplasma. In immunocompetent individuals, the primary infection is usually asymptomatic and latent infection may persist in the host for life. The reactivation of the parasite that causes the disease usually occurs in patients with cellular immunosuppression, especially HIV/AIDS patients, and more frequently in those with advanced immunosuppressive states [8]. This disease has a high morbidity and mortality and it continues to have a poor prognosis for patients with HIV/AIDS [7].
Cerebral toxoplasmosis usually occurs in subacute evolution, although it can sometimes appear rapidly and progressively. The clinic will depend essentially on the location of the lesion and the number of them. The most common symptoms are headache, focal neurology, confusion, behavioral disturbances and epileptic seizures. In the absence of treatment, the disease progresses leading to death [4,7].
The gold standard imaging test for toxoplasmosis is magnetic resonance, preferred over CT scan because of its higher sensitivity. Usually, cerebral toxoplasmosis presents as multiple brain lesions with ring-enhancing, occasionally they are single lesions, frequently associated with cerebral edema. There is a predilection for involvement of the basal ganglia, frontal lobe and temporal lobe, although they can appear in any region of the brain [4,6,7].
Concerning serological tests, the majority of patients with cerebral toxoplasmosis are positive for antibodies anti-toxoplasma IgG. However, the absence of IgG does not rule out the diagnosis, since a small percentage of patients with cerebral toxoplasmosis are IgG negative, as happened in one of our patients that despite the negative serology the disease was not ruled out as a possible diagnosis, although in a repeated analysis, the patient later presented positive IgG. Possible reasons for presenting a negative serology include a primary infection or severe immunosuppression that leads to the inability to produce antibodies [1,8]. This second assumption is the one that we consider presented our patient, in whom after recovering part of his immunological status with the start of HAART, he presented positive IgG for toxoplasma in the repeated analysis.
Habitually, to reach the diagnosis it's necessary to perform a lumbar puncture in which we don't find relevant biochemical alterations, but PCR is able to detect the presence of Toxoplasma DNA that will support the diagnosis, although as it happened with serological tests, a negative result does not exclude the diagnosis (low specificity around 50-60%) [1,4,6,7].
According to recent literature, the definitive diagnosis of cerebral toxoplasmosis in HIV patients requires: a compatible clinical picture, one or more compatible brain lesions and histological confirmation that allows visualization of the parasite. In clinical practice, a brain biopsy is not performed to reach the diagnosis due the risk of the test and we count on other criteria to get to a presumptive diagnosis. Generally, the diagnostic approach can be made based on several aspects [4,6-8]:
A) Compatible clinical syndrome.
B) Presence of one or more space-occupying lesions in the neuroimaging compatible with Toxoplasma.
C) Evidence of parasite DNA in CSF.
D) Presence of positive IgG antibodies for Toxoplasma gondii.
E) Clinical-radiological improvement 10-14 days after starting treatment.
F) Immunosuppression status based on CD4 numbers < 200 cells/mm3 (especially in those with numbers < 100 cells/mm3) without prophylaxis.
G) Absence of another more probable diagnosis.
In a practical way, an HIV positive patient with severe immunosuppression (CD4 < 200 cells/mm3) without effective prophylaxis, compatible clinical syndrome and the presence of imaging lesions suggestive of toxoplasmosis, should receive treatment against toxoplasma, especially those with positive anti-toxoplasma IgG [6-8]. Therefore, following this premise, we stared treatment for cerebral toxoplasmosis. If no clinical improvement appears within 14 days, other alternatives should be search [6-9].
Within the differential diagnosis of space-occupying lesions in HIV patients with advanced immunosuppression, in addition to cerebral toxoplasmosis, we can also find the primary cerebral lymphoma and progressive multifocal leukoencephalopathy, as previously mentioned. In addition, other entities to take into account are tuberculosis and cryptococcosis. Also, depending on the epidemiological environment of the patient, other infections such as histoplasmosis and other diseases related to the epidemiology of the country of origin, such as Trypanosome cruzi, should be considered [7-9].
Primary CNS lymphoma is often the main differential diagnosis. It is a non-Hodgkin lymphoma associated with an infection by the Epstein-Barr virus (EBV). It generally appears in highly immunosuppressed patients with CD4+ lymphocytes less than 50 cells per mm3 [9]. It usually presents as a subacute onset with focal neurological deficits depending on the location of the intracranial lesion, headache and cognitive alterations [1]. Primary lymphoma is usually accompanied by positive EBV PCR in CSF in 60-80% of cases with HIV [10,11].
The different imaging tests (brain MRI, PET or SPECT) can help distinguish both entities, but in some cases a brain biopsy may be necessary to reach the diagnosis, especially in cases of not presenting a good response to empirical treatment of toxoplasmosis [9]. On MRI, lymphoma presents as single or multiple lesions, rarely more than 4 lesions, isointense or hypointense, and occasionally hyperintense on T2/FLAIR sequences. It is characteristically localized in supratentorial white matter that extends along the ependymal surfaces or crosses the corpus callosum. Other locations include the basal ganglia, the corticomedullary junction, the pons and the midbrain-pons. In these cases, enhancement is usually more irregular and less homogeneous, contrary to what occurs in toxoplasmosis [1,9]. They are generally lesions that cause a mass effect on the adjacent brain parenchyma, as occurs in toxoplasmosis and not in the case of PML, as we will discuss later. The rapid progression of the cerebral lesion in the imaging studies performed repeatedly over time favors the diagnosis of lymphoma [9].
Despite being the most widely used imaging technique in these patients, MRI may sometimes not be a reliable test to establish the differential diagnosis between lymphoma and toxoplasmosis [1]. A test that can help in the differential diagnosis of these lesions is Thallium 201 SPECT/scintigraphy. This technique shows the uptake of thallium by lymphoma tumor cells, while other lesions do not, which makes it a very useful tool to differentiate CNS lesions [9].
In the absence of contraindication, lumbar puncture to detect tumor cells, present in up to 15% of patients, and EBV-positive PCR in CSF, can be very useful to reach the diagnosis [9]. The combination of a positive SPECT for lymphoma and a positive Epstein-Barr virus (EBV) PCR in CSF has been shown to obtain a sensitivity and a negative predictive value of 100% [1].
Another clinical entity that is part of the differential diagnosis of space-occupying lesions in HIV-patients is progressive multifocal leukoencephalopathy (PML), named for its pathological characteristics. It is an opportunistic viral infection of the CNS that progressively affects the white matter caused by the JC polyomavirus. The fundamental requirement to develop a PML is to present a profound immunosuppression of cellular immunity like is seen in HIV/AIDS patients. Diagnosis is based on the neurological symptoms, characteristic lesions on magnetic resonance imaging (MRI) and the presence of JC virus DNA in the CSF. Currently, achieving reconstitution of the immune system is the best prognosis for this condition [12].
PML clinic is characterized by a subacute and insidious onset of progressive neurological deficits and the symptoms will depend on the location of the lesions. They can present a multitude of symptoms, but the most frequent in HIV patients is an alteration of the motor system, cognitive deficit and language disorders. Although rare, brainstem involvement causes the most severe symptoms. It is a progressive disease and symptoms get worse over time [1,12].
Typical features in PML imaging include lesions with a predilection for subcortical areas of the frontal, parieto-occipital and cerebellar regions, although involvement of other areas has also been described. In T2, we can observe multifocal hyperintense demyelinating lesions of asymmetric distribution. Typically they are lesions that do not have a mass effect, although there are few cases described in the literature with mass effect. In most cases, there is no gadolinium enhancement, although a small percentage of patients may show weak areas of enhancement, but not a ring-enhancing pattern [1,12].
For the diagnosis, the detection with PCR of JC virus DNA in CSF is very useful. With the newest ultrasensitive PCR techniques, the sensitivity is more than 95% and the specificity is more than 97%. Despite its high sensitivity, a negative result does not rule out the diagnosis, because viral loads can be very low and may not be detected by laboratory tests [1,12]. The evolution of these patients, despite improve their immune status with antiretroviral treatment, is usually unfavorable [12].
We would like to make a mention of a rare form of disease associated with JC virus infection known as granule cell neuronopathy (GCN). GNC is characterized by lytic infection of cerebellar granule cell neurons and clinically it manifests with cerebellar symptoms such as dysarthria or ataxia. Its etiopathogenic is not exactly known but it seems to be caused by a mutation in the VP1 gene of the JC virus that modifies the cellular tropism of the virus and affects cerebellar neurons. MRI findings are characterized by cerebellar atrophy with or without cerebellar white matter lesions [13-15].
Although they are less frequent, other differential diagnoses that must be taken account are: tuberculosis, histoplasmosis and cryptococcosis. Next, we describe briefly the main characteristics of each one.
CNS tuberculosis can manifest as meningitis or a tuberculoma. Tuberculous meningitis is becoming more and more uncommon [1,16]. Tuberculomas can be asymptomatic or present with headache, seizures, neurological focality and/or signs of elevated intracranial pressure. It's not necessary a very advanced state of immunosuppression to present CNS tuberculosis. Radiologically, tuberculomas are single or multiple lesions, hypointense on T1 and hyperintense on T2, with homogeneously ring-enhancing and surrounded by perilesional edema. The diagnosis should be suspected in patients with a brain lesion with the characteristics previously described and with relevant epidemiological factors (history of previous TB infection or disease, known or possible exposure to TB and/or past or present residence in or travel to an area where TB is present). A presumptive diagnosis can be made in the context of relevant clinical and epidemiological factors and typical radiographic findings, especially if a definitive diagnosis of TB has been established at the extraneural level [1,16].
Cryptococoma is a parenchymal lesion that appears as a result of Cryptococcus infection. It typically affects patients with severe immunosuppression with CD < 100 and presents as subacute fever, headache and general malaise in addition it might associate symptoms of intracranial hypertension [9,17]. On MRI, it presents as single or multiple lesions of variable size with hypointensity in T1 and hyperintensity in T2 with variable peripheral edema and ring-enhancing. Cystic lesions close to the perivascular spaces, in the ganglia of the base, thalamus and cerebellum are also frequent and very characteristic. Other alteration found in a non-negligible percentage of patients is hydrocephalus, which sometimes even requires the placement of a ventriculo-peritoneal drain [1,17-19]. To reach the diagnosis of cryptococcosis are useful: the positivity of cryptococcal antigens in serum and CSF, culture and the Chinese ink technique [17,20,21].
Finally, Histoplasmosis is included in the differential diagnosis of space-occupying lesions in HIV patients, acquires great relevance in patients from Latin America [22]. Histoplasmosis in HIV patients generally presents as a disseminated infection, which differentiates it from toxoplasmosis, although in some cases it can present with involvement only of the CNS [21]. MRI often shows single or multiple lesions with ring-enhancing, as occurs in toxoplasmosis, without other characteristic features [23]. For the diagnosis of histoplasmosis, several techniques are used in an associated manner, among which are: direct microscopy, cultures, including bone marrow and affected tissues, detection of antibodies and PCR in CSF and detection of Histoplasma antigen in urine, blood, CSF or bronchoalveolar lavage according to symptoms and affected organs; it is also essential to know the epidemiology of the patient to help in diagnosis [21-24].
In our cases, the patients presented to the emergency department with symptoms of neurological deficits without other symptoms in the anamnesis that suggested affectation of other organs. During the initial study, both patients were diagnosed an HIV infection in advanced stages, which opened a range of diagnostic possibilities focused mainly on the differential diagnosis of space-occupying lesions in the CNS. Both were Spanish and did not travel abroad, which allowed us to consider histoplasmosis-type diseases to be of low or null probability, as well as they did not present respiratory symptoms or other clinical symptoms that could suggest this diagnosis. In both of them, all the tests of Mycobacterium tuberculosis were negative, as well as they did not present respiratory symptoms or symptoms at other level or typical lesions in the imaging tests, it seemed equally unlikely the diagnosis of tuberculomas.
Based on the clinical features, only with neurological symptoms, the main suspicions diagnosis were: cerebral toxoplasmosis, progressive multifocal leukoencephalopathy, primary CNS lymphoma and cryptococcosis.
MRI images showed hyperintense lesions on T2, most of them punctate, without ring-enhancement with the administration of contrast and with affectation of multiple areas of the CNS (lobar, corpus callosum, cerebellar, brain stem ...). Due to the distribution of the lesions, any diagnostic option was possible. It is true that typical cystic lesions of cryptococcus were not seen, which raised doubts about its diagnosis. However, it was very remarkable that cerebral lesions did not present contrast enhancement, a typical characteristic of toxoplasmosis and not of PML and lymphoma as previously described. Likewise, some lesions presented associated vasogenic edema and others did not, which did not help to define a specific entity.
One of the patients had positive toxoplasma serology while the second patient was negative in a first determination; as we have indicated previously, highly immunosuppressed patients may not present positive serology due to lack of immunological response; on the second admission, the same patient had a positive IgG anti-Toxoplasma and we consider that the initial test was a false negative.
In both cases, SPECT and CSF lymphocyte analysis were very useful. In both patients, the thallium scintigraphy did not show uptake consistent with lymphoma and the CSF analysis did not show tumor cells or atypical lymphocytes, so the diagnosis of primary lymphoma was ruled out despite the fact that one of the patients had a positive EBV PCR which was finally considered a virological escape with no real clinical relevance.
We detected replication of JC virus in CSF of both patients so PML was an added diagnosis of co-infection. GNC was ruled out, because of the absence of cerebellar atrophy in MRI, which is the typical finding of this disease.
Serological and laboratory tests for the diagnosis of cryptococcus were negative, therefore, with all the data together, this diagnosis option was ruled out.
Therefore, we were faced with two patients with advanced HIV immunosuppression without any prophylactic treatment, with compatible neurological symptoms, positive serology tests for Toxoplasmosis and with images of brain unusual of toxoplasmosis. Consequently, following the criteria outlined above and given the severity of both conditions, we opted to start treatment for cerebral toxoplasmosis with a spectacular response in both cases and with practical resolution of symptoms 10-14 days after the start of treatment with the exception of mild residual focal deficits.
Thus, due to negative results from the rest of the studies and given the evolution and response to treatment for cerebral toxoplasmosis, both patients were diagnosed with atypical cerebral toxoplasmosis and PML co-infection, in both cases with atypical presentations due to the characteristics of the lesions in the imaging tests.
With these cases we want to highlight the importance of making an adequate differential diagnosis, especially important in immunosuppressed patients, in which cases the presentations may be atypical, and to consider different coexisting opportunistic infections.
Written informed consent was obtained from the patient for publication of this case report and accompanying images. A copy of the written consent is available for review by the Editor-in-Chief of this journal on request.
No funding or sponsorship was received for publication of this article.
All named authors take responsibility for the integrity of the work as a whole, and have given their approval for this version to be published.
Conceptualization, design and methodology: AGV, ADS; Attentions to the patients: All authors; Writing first draft: AGV; Critical revision for important intellectual content: AGV, ADS; Final approval: All authors.
This manuscript has not been previously published in other journal.