The preference for empiric antibiotic therapy with carbapenems over non-carbapenems in all extended-spectrum β-lactamase-producing Enterobacteriaceae (ESBL-E) colonized patients with sepsis warrants further investigation from an ecologic perspective; even more so in the elderly (≥ 65 years) because the proportion of ESBL-E in adults increases with patients' age. In this retrospective observational study, enrolling 547 ESBL-E colonized elderly patients with community-onset sepsis, hospitalized at a single medical centre from 2011 to 2015, the positive predictive value of ESBL-E faecal colonization for ESBL-E aetiology of sepsis was significantly higher (66.1%) when sepsis originated from a urinary tract infection than from a respiratory tract infection (26.1%), other known origins (31.6%), or an unidentified origin (13.0%). Carbapenems were prescribed empirically in 145 patients (26.5%), and 402 received non-carbapenem antibiotics. Univariate analysis suggested a higher 30-day mortality in the non-carbapenem vs. carbapenem group. However, the estimated association was smaller and not significant (OR = 1.1, 95% CI, 0.6-1.9, P = 0.62) in the multiple regression analysis adjusted for age, sex, Charlson comorbidity index score, and severity, origin or aetiology of sepsis. Therefore, carbapenem-sparing empiric therapy seems appropriate for non-critically ill elderly ESBL-E carriers with community-onset sepsis, even more so when sepsis originates outside urinary tract.
ESBL colonization, Elderly, Empiric therapy
The proportion of extended-spectrum β-lactamase-producing Enterobacteriaceae (ESBL-E) increases in hospital-acquired and in community-onset infections worldwide [1-3] but also with patients' age [4,5]. Guidelines and treatment recommendations endorse empiric anti-ESBL-E antibiotic therapy, preferably with carbapenems, in patients colonized with ESBL-E, regardless of patients' age or severity of illness [6-8]. However, estimates of a risk represented by ESBL-E colonization for subsequent ESBL-E infection are ranging from 7.4% to 25% [9-11]. Such divergent results originate from countries with marked differences in the prevalence of ESBL producers among Enterobacteriaceae [3].
Estimates of a risk represented by inappropriate empirical therapy for unfavorable outcome in patients with ESBL-E infections are not unequivocal [12]. Palacios-Baena, et al. were unable to show that empiric treatment with non-carbapenem antibiotics was associated with a worse outcome compared with carbapenems in patients with bloodstream infection due to ESBL-E [13]. On the other hand, the results of the MERINO Trial suggest that piperacillin-tazobactam as definitive therapy for bloodstream infections caused by ESBL-producing Escherichia coli or Klebsiella pneumonia was inferior to meropenem and should be avoided in this context [14]. Broadness of empiric antibiotic therapy raises not purely clinical, but also ethical dilemmas. Argument that patients with moderate to severe infections should be given empiric antibiotic treatment, not covering all potential pathogens, in order to reduce the rise in resistance and protect future patients in this way has been advocated reasonably [15].
In this study, we focused on community-onset sepsis in elderly ESBL-E carriers with the aim to assess the positive predictive value of ESBL-E faecal colonization for infection with these organisms according to the origin of sepsis, and to evaluate empiric treatment outcome in non-carbapenem versus carbapenem prescribed patients.
A total of 547 patients aged ≥ 65 years were eligible during the 5-year study period (Figure 1). Sepsis originated from RTI in 253 patients, UTI in 195, and other identified origins in 50 (skin and soft tissue and/or bone in 32, abdomen in nine, heart or central nervous system in four, and infection at two sites in five); the origin of sepsis was unidentified in 49 patients. ESBL-E colonization was known on admission to hospital in 219 patients, and it was confirmed in 328 (60%) patients when results of rectal swab cultures, taken on admission, were available.
 Figure 1: Flow diagram.
Figure 1: Flow diagram.
ID: infectious diseases; ESBL-E: extended-spectrum β-lactamase-producing Enterobacteriaceae; CRB: carbapenem. View Figure 1
The aetiology of infection was microbiologically identified in 62.2% (340/547) patients (Table 1). The PPV of ESBL-E faecal colonization for ESBL-E aetiology of sepsis was 45.6% (95% CI, 40.2-51.0%). The highest PPV was found when sepsis originated from UTI (66.1%; 95% CI, 58.4-73.2%), but it was much lower for RTI (26.1%; 95% CI, 18.2-35.3%), for other identified origins (31.6%; 95% CI, 17.5-48.7%), and for sepsis of unidentified origin (13.0%; 95% CI, 2.8-33.6%). The PPV of ESBL-E colonization for ESBL-E bacteraemia was only 4.4% (95% CI, 2.8-6.5%).
Table 1: Characteristics of patients according to empiric therapy: Patients who received Carbapenem (CRB) vs. patients treated with Non-CRB. View Table 1
BLI were prescribed empirically in 266 patients, 145 patients were treated empirically with CRB, and 136 patients were given empirically other anti-Gram-negative antibiotics, mostly fluoroquinolones (Table 1). The most common dosing regimens were: amoxicillin-clavulanate 1200 mg/8 h; piperacillin-tazobactam 4500 mg/8 h; ertapenem 1 g/24 h; imipenem 500 mg/6 h; and meropenem 1 g/8 h, or adjusted equivalent in the case of renal failure. Patients who received BLI or other anti-Gram-negative antibiotics were combined in the non-CRB group for further analysis.
On admission to hospital, patients not known to be colonized with ESBL-E were less often treated empirically with CRB than patients whose colonization with ESBL-E was already documented (26/328, 7.9% vs. 119/219, 54.3%; P < 0.001) and had worse 90-day survival (Figure 2A; P = 0.03 from log-rank test).
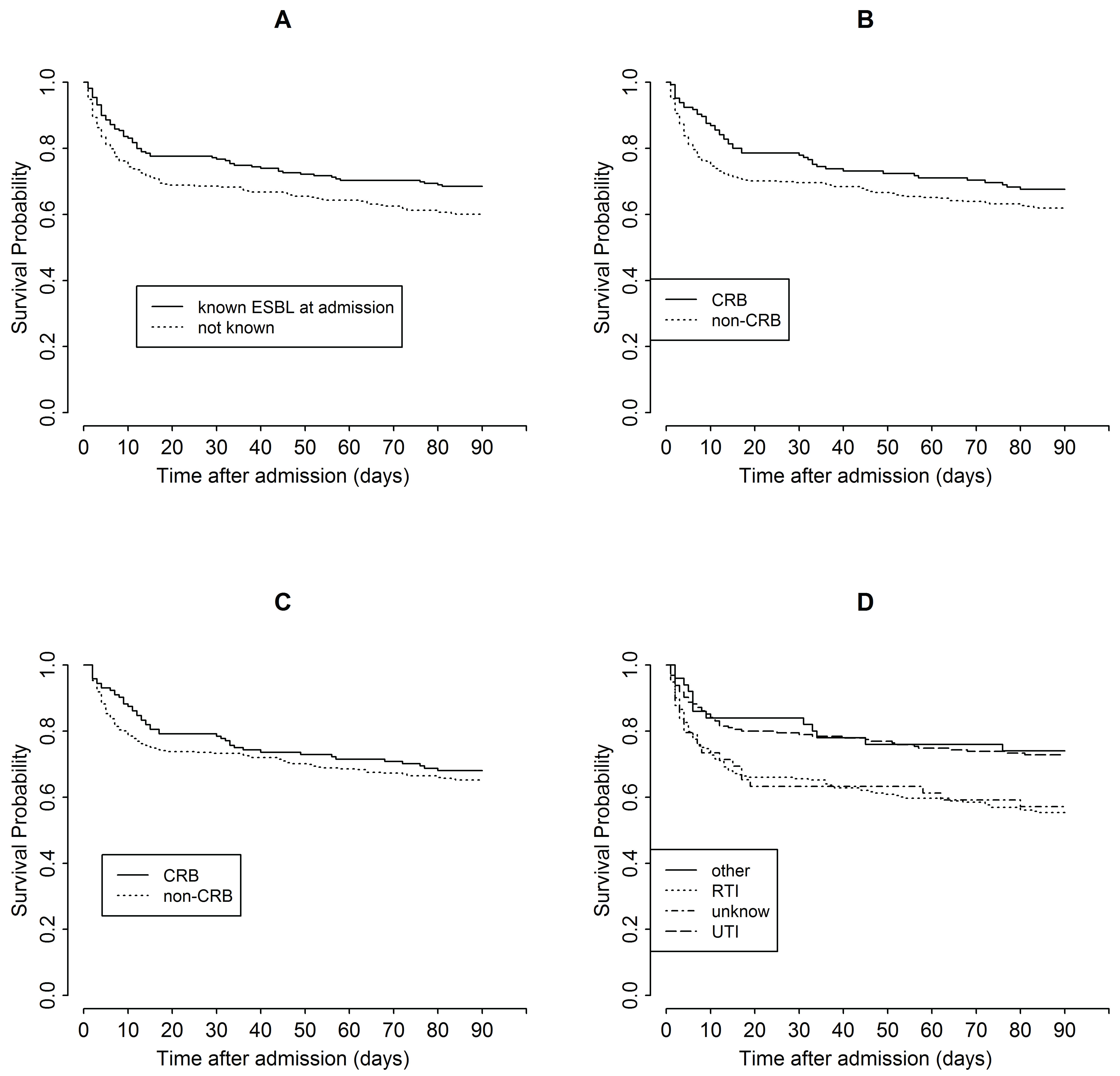 Figure 2: Estimated probability of survival in the first 90 days after admission for (A) patients known vs. not known to be already colonized with ESBL-E on admission, (B,C) patients empirically treated with carbapenem (CRB) vs. non-CRB for all patients (B) and for those who survived > 24 h (C), and (D) patients with different origins of sepsis. RTI: respiratory tract infection; UTI: urinary tract infection. View Figure 2
Figure 2: Estimated probability of survival in the first 90 days after admission for (A) patients known vs. not known to be already colonized with ESBL-E on admission, (B,C) patients empirically treated with carbapenem (CRB) vs. non-CRB for all patients (B) and for those who survived > 24 h (C), and (D) patients with different origins of sepsis. RTI: respiratory tract infection; UTI: urinary tract infection. View Figure 2
The 30-day mortality was higher in patients empirically prescribed non-CRB than in those who were given CRB, but the difference was not statistically significant (22.1% vs. 30.3%; difference, 8.2%; 95% CI, 0.98-2.4; P = 0.06, OR = 1.5; Table 2). The association between treatment regimen and 30-day mortality was even weaker when the analysis was adjusted for other covariates, listed in Table 2, in multivariable regression analysis (OR = 1.1; 95% CI, 0.64-1.90, P = 0.71; Table 2). Only age, and severity and origin of sepsis, but not the empiric antibiotic regimen (non-CRB versus CRB), were significantly associated with 30-day mortality in the multivariable analysis. In comparison with the CRB group, patients who received non-CRB were older, had different origins of sepsis, and less often had an identified microbiologic aetiology of infection (Table 1).
Table 2: Univariate analysis and multiple regression analysis of associations between selected variables and 30-day mortality. View Table 2
The observed overall survival in the first 90 days after admission was significantly better for patients who were known to be ESBL-E colonized at admission (Figure 2A) yet non significantly better for those treated with CRB (Figure 2B), but the differences were small for both comparisons (P = 0.03 and P = 0.13 from log-rank test, respectively). Notably, the greatest difference was observed in the first day after admission, when only one death out of 21 was among CRB-treated patients. When patients who did not survive past day 1 were removed from the analysis, the differences between the treatment groups were smaller (Figure 2C; P = 0.42 from log-rank test).
Cox regression analysis also indicated that CRB-treated patients had a higher probability of survival than patients treated with non-CRB (HR = 1.29, 95% CI: 0.93 to 1.78, P = 0.13), however when the analysis was adjusted for other covariates, listed in Table 2, in the multivariable model, the difference of the estimate was substantially reduced (HR = 1.05, 95% CI: 0.71 to 1.56, P = 0.80). In addition, when HR was estimated as a time-dependent coefficient, the better survival of patients treated with CRB was confirmed to be mostly due to the differences observed within the first few days after diagnosis (Figure 3).
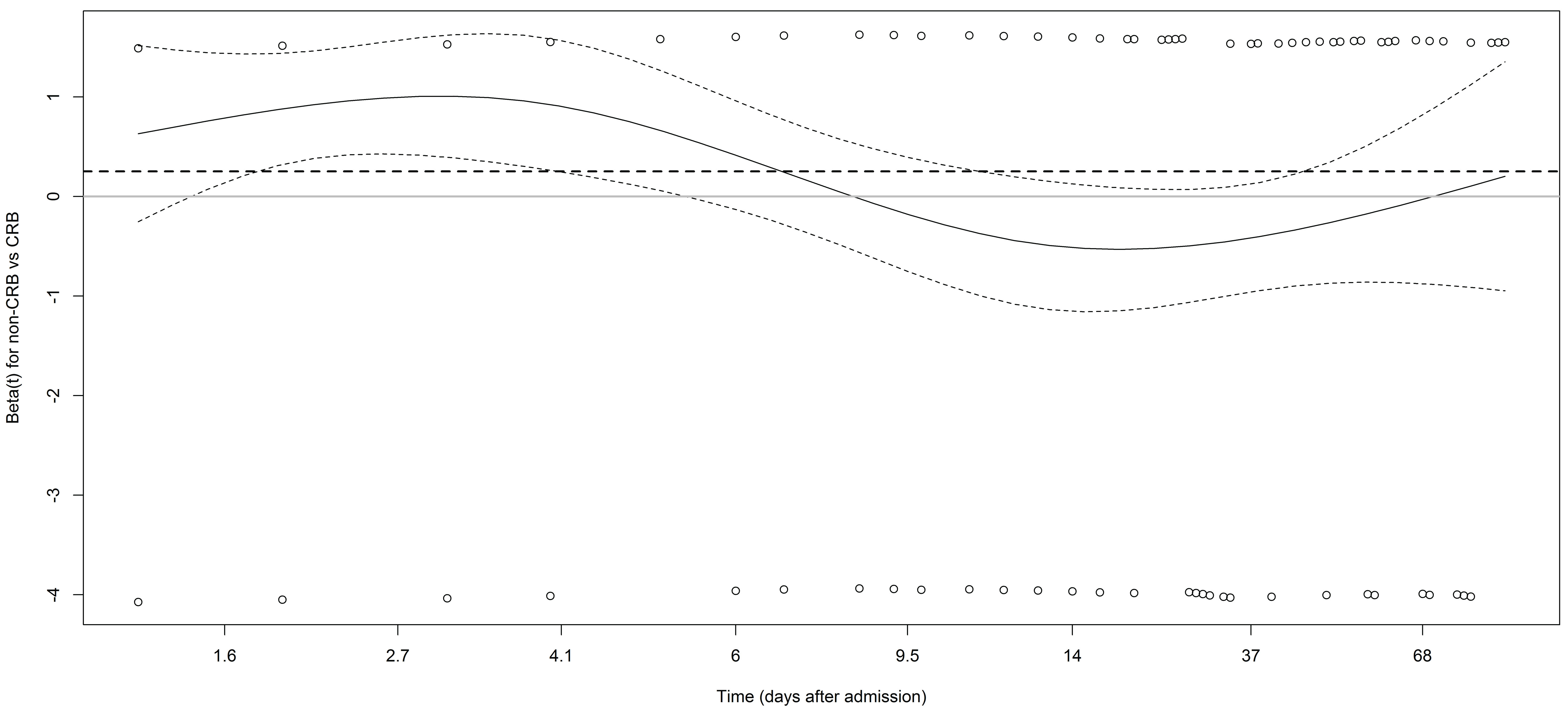 Figure 3: Plot of the estimate of β(t) (log of HR(t)) and its pointwise 95% confidence intervals used to diagnose and understand non-proportional hazards (values above 0 indicate that patients with non-carbapenem (non-CRB) have worse survival than patients treated with CRB). The dashed line represents the estimate obtained using the Cox model that assumes the proportional hazards (β = 0.25, HR = 1.29).
Figure 3: Plot of the estimate of β(t) (log of HR(t)) and its pointwise 95% confidence intervals used to diagnose and understand non-proportional hazards (values above 0 indicate that patients with non-carbapenem (non-CRB) have worse survival than patients treated with CRB). The dashed line represents the estimate obtained using the Cox model that assumes the proportional hazards (β = 0.25, HR = 1.29).
t: time; HR: hazard ratio. View Figure 3
Patients with a UTI were much more likely to receive CRB empirically than patients with RTI (96/228, 42.1% vs. 45/302, 14.9%; P < 0.001), and more often had ESBL-E identified as the causative bacteria (124/228, 54.4% vs. 34/302, 11.3%; P < 0.001). Different anatomic origins of sepsis were associated with distinct treatment outcomes as measured by 30-day mortality (Table 2 and Table 3) and overall survival time (Figure 2D, P < 0.001 from log-rank test); patients with RTI or sepsis of unknown origin had worse outcomes. However, the interaction between sepsis origin and empiric antibiotic treatment was not associated with 30-day mortality in the model that included the two covariates and their interaction (P = 0.48) or in the model that additionally included all the covariates shown in Table 2 (P = 0.16).
Table 3: Association between origin or etiology of sepsis, empiric therapy and 30-day mortality. View Table 3
The difference in mortality between the CRB and non-CRB cohorts was more pronounced in patients with an identified aetiology of sepsis, even more so if ESBL-E were the causative bacteria. The difference was almost non-existent for sepsis of unidentified aetiology (Table 3). The interaction between sepsis aetiology and empiric antibiotic treatment was not associated with 30-day mortality in the model that included the two covariates and their interaction (P = 0.58) or in the model that included all the covariates shown in Table 2 (P = 0.76).
Proportion of ESBL-E clinical isolates is rising globally [1-3], but also with patients' age [4]. The number of elderly patients is growing in most countries as life expectancy is increasing [16]. Age showed to be an independent factor for sepsis mortality [17], leading to suggestions that broader empiric therapy for elderly may be required [4]. Therefore, in this study we focused on elderly ESBL-E-colonized patients with community-onset sepsis to further investigate whether such an approach is appropriate.
This was a retrospective study and a decision on empiric therapy was left to discretion of the attending physicians. Because there is some ambiguity regarding sepsis definition, we decided to apply the SIRS criteria, which for decades had played a fundamental screening role for sepsis [18]. Since patients with confirmed and those with suspected colonization did not differ in their actual colonization status on admission, we were able to observe retrospectively a large enough population of colonized elderly patients who were prescribed non-CRB empirically.
A recent meta-analysis of randomized controlled trials showed no advantage of empiric treatment with CRB over BLI in patients with Gram-negative sepsis, when all-cause mortality was used as the primary outcome [19].
Our study, which focused on community-onset sepsis in elderly patients colonized with ESBL-E, showed similar results. Although our results suggest higher 30-day mortality in non-CRB (predominantly BLI) compared to CRB treatment group, the difference was particularly pronounced during the first 24 h of hospitalization. This time is too short to allow reliable evaluation of an antibiotic effect. Indeed, patients who died within the first 24 h after admission were more severely ill on admission than those who survived > 24 h. Multivariable regression analysis showed that age, severity of sepsis, and origin of sepsis, but not empiric therapy were associated with 30-day mortality (Table 2). Our results accord with findings showing no association between empiric BLI vs. CRB and 30-day mortality in patients with ESBL-E. coli bacteraemia [1] or ESBL-E. coli acute pyelonephritis [20], and comparable efficacy of empiric BLI (if active in vitro) and CRB for ESBL-E bacteraemia regardless of specific species or the origin of infection [21]. Although median age of the patient population analysed in these reports ranged from 67 to 74 years, in none of them the analysis focused exclusively on elderly population. Meini, et al. concentrated their analysis on elderly patients with ESBL-E bacteraemia (40/42 patients were ≥ 65-years-old) and did not find beneficial effect of empiric CRB over BLI. They advocate non-CRB empiric therapy if the likely origin of infection is UTI but assume that in those patients whose bacteraemia originates in RTI, CRB-sparing empiric regimen should not be promoted because of higher risk of death in these patients [22]. Inversely, Leclercq, et al. advise that with the exception of UTI, the use BLI in infections caused by ESBL producers remains controversial and should be approached with caution [23]. In the present study, we confirmed higher mortality rate if sepsis originated from RTI compared to UTI. However, when considering the interaction of sepsis origin and empiric therapy, we could not establish association between this interaction and 30-day mortality. Additionally, since the probability of ESBL-E aetiology was lower when sepsis originated from RTI (26.1%) than from UTI (66.1%), empiric therapy with non-CRB appeared particularly substantiated in patients with RTI as opposed to UTI.
When aetiology of sepsis is identified, empiric antimicrobial treatment can be tailored according to the susceptibility results. In a substantial proportion of patients in the present study (37.8%) the aetiology of sepsis remained unidentified, which is not surprising [24]. According to our results, elderly patients colonized with ESBL-E with sepsis of unidentified aetiology did not benefit from empiric CRB in comparison with non-CRB therapy. On the contrary, empiric CRB treatment may have disadvantages such as promoting resistance, constraints regarding further treatment options, and may represent overtreatment for a proportion of patients in whom non-CRB therapy might have been successful.
Several limitations should be considered when interpreting our results. First, the study was not randomized, but retrospective and observational in design, and was limited to a single centre, which may preclude generalization of the results to other hospitals. Second, not all elderly patients colonized with ESBL-E with community-onset infection were included in this analysis because the screening strategy with a single admission swab as used in the present study is not 100% sensitive. Third, a comparative group of non-colonized patients was not included in the study. Therefore, we can only speculate that an absence of any benefit of empiric CRB would be even more pronounced in a subgroup of patients without ESBL-E colonization, because of a lower probability of ESBL-E infection in these patients. Fourth, confounding due to unmeasured variables may have occurred. Fifth, some of the microbiologically unidentified cases with SIRS could not have had sepsis, therefore sepsis could have been over diagnosed. Nevertheless, we were able to assess a relatively large case series of ESBL-E colonized patients composed exclusively of elderly and to identify the major factors such as age, and severity and origin of sepsis, that potentially affect mortality in these patient population.
In conclusion, in a population of elderly patients colonized with ESBL-E who have community-onset sepsis, our data suggest that non-CRB empiric treatment does not correlate with worse outcome. In order to reduce the rise in resistance, it may be reasonable to treat these patients with an alternative to CRBs, particularly if sepsis originates from a site other than a UTI, since in UTI, Enterobacteriaceae are the predominant aetiology. Consideration of future resistance may be taken into less account for critically ill patients.
A retrospective, observational cohort study was conducted in a single tertiary care university hospital in Ljubljana, Slovenia in the period from January 2011 to December 2015. Consecutive elderly (≥ 65 years) patients admitted for suspected community-onset Gram-negative sepsis, who had an ESBL-E-positive rectal swab obtained on admission as identified from the microbiology laboratory databases, qualified for the study. The study was approved by the Ethics Committee of the Republic of Slovenia (No. 84/10/14). All methods were performed in accordance with the relevant guidelines and regulations. Written consent was not required because of the retrospective observational nature of the study. The data had been published previously for other purposes [25]. Only the first episode per patient during the study period was included. Clinical information was retrieved from medical records and reviewed by two authors. Patients were excluded if ESBL-E fecal colonization was found later during hospitalization and/or they had a hospital-acquired infection (infection occurring > 48 h after hospital admission.
Clinical samples were analyzed using standard methodology. ESBL surveillance samples were inoculated onto chromogenic selective agar (chromID™ ESBL from bioMérieux, Marcy l'Etoile, France). Colonies that were consistent with ESBL-producing E. coli and K. pneumoniae were identified using BBL Crystal Enteric/Nonfermenter kits (BD, Sparks, MD) until 2012, when MALDI-TOF mass spectrometry using Biotyper Microflex LT (Bruker Daltonics, Bremen, Germany) was introduced into routine laboratory practice. The standard disk diffusion method or the Vitek® 2 system (bio-Mérieux) was used for antimicrobial susceptibility testing. Susceptibility of ESBL-producing E. coli and K. pneumoniae to β-lactam antibiotics (penicillin without inhibitors, third and fourth generation cephalosporins, carbapenems) and non-β-lactam antibiotics (amikacin, gentamicin, ciprofloxacin) was routinely determined. Results were interpreted in accordance with Clinical and Laboratory Standards Institute (CLSI) guidelines until 2014 when European Committee on Antimicrobial Susceptibility Testing (EUCAST) guidelines were introduced [26]. Production of ESBL was confirmed phenotypically in a combination disk diffusion test (cefotaxime, ceftazidime alone and in combination with clavulanic acid); the test was considered positive if the diameter of the inhibition zone ≥ 5 mm larger with clavulanic acid than without [26,27].
The data collected included survival time collected from the National Population Registry for those who lived past day-30.
A Charlson comorbidity index score was calculated for each participant [28]. Sepsis was defined as the presence of at least two defining parameters for systemic inflammatory response syndrome (SIRS) in a patient with suspected infection [29]. Gram-negative sepsis was defined as sepsis in a patient in whom blood samples were obtained for culture and antibiotic treatment which included anti-Gram-negative coverage started on admission to hospital. Severity of sepsis at presentation was graded by the Pitt bacteraemia score [30]. Origin of sepsis was defined as: urinary tract infection (UTI), lower respiratory tract infection (RTI), other identified origins, and sepsis of unidentified origin. The standard definitions of the Centers for Disease Control and Prevention were used to define UTI and RTI [31].
Sepsis was declared as community-onset when infection occurred among non-hospitalized patients or < 48 h after admission. Community-onset sepsis was considered as community-acquired if none of the following criteria for healthcare-associated infection was fulfilled: the patient had received i.v. therapy, wound care, or specialized nursing care at home or in a day hospital in the preceding 30 days; the patient had attended a haemodialysis clinic or resided in a nursing home during the preceding year [32].
On admission to hospital, stool samples were obtained by rectal/anal swabs from patients with known ESBL-E colonization or infection sometime in the past, from patients with a healthcare-associated infection and from those with a community-acquired infection who had received aminopenicillins, cephalosporins, or fluoroquinolones or who had travelled from an area highly-endemic for ESBL-E in the previous 30 days. Blood samples for culture were drawn from all patients. Respiratory specimens were collected from patients with suspected RTI, and urine was sent for culture in patients with suspected UTI.
The selection of antibiotic therapy was left to the discretion of the attending physicians. Antimicrobial therapy administered on admission (before susceptibility results were available) was considered empiric. Microbiologically identified etiology was defined as clinically relevant bacteria isolated from a primarily sterile site, and/or from urine in the case of UTI and/or a respiratory specimen in the case of RTI.
Data were summarized with medians (interquartile ranges) or frequencies (percentages). Patients treated with (non-CRB vs. CRB) empiric therapies were compared according to characteristics listed in Table 1 using univariable logistic regression. The association between 30-day mortality (binary endpoint) and various covariates (age, sex, Charlson index score, Pitt severity score, sepsis origin, sepsis aetiology (identified ESBL-E vs. identified not ESBL-E vs. unidentified), empiric therapy (non-CRB vs. CRB), ESBL-E bacteraemia) was estimated using univariable and multivariable logistic regression. Age, Charlson index score and the Pitt severity score were included as continuous covariates in the model. The association between the same covariates and time to death was estimated using a Cox regression model, censoring survival times longer than 90 days. To account for the time-dependent effect of the empiric therapy, an additional Cox regression with a time-dependent regression coefficient for empiric therapy was fitted. To understand the behaviour of the time dependent coefficient, the estimate was presented graphically using the cox.zph() function in R. Survival curves were estimated using the Kaplan–Meier method and their univariate comparisons were based on the log-rank test. Results from logistic regression models were expressed as odds ratios (OR) with their 95% confidence intervals (CI) and Wald P values, and results from Cox models as hazard ratios (HR); 95% CI for proportions were based on binomial distribution. The PPV of ESBL-E colonization for ESBL-E etiology of sepsis was estimated on the basis that culture results were the gold standard. The R statistical language was used for all analyses [33].
We determined the PPV of ESBL-E faecal colonization for sepsis caused by these bacteria and assessed the impact of empiric therapy (non-CRB vs. CRB) on treatment outcome. The primary treatment outcome was 30-day all-cause mortality; the secondary treatment outcome was a 90-day survival time.
The data analysed during this study are included in Supplementary Data, online.
Concept and design: MNK and DS. Acquisition of data: MNK, ST, MP, MMP. Analysis and interpretation of data: LL, DS. Preparation of manuscript: MNK, DS.
The authors declare no competing interests.