Alopecia areata (AA) is a potentially reversible auto-immune non-scarring baldness on the scalp, which can be extended to the entire body. There are many scientific evidence as regards the impact of diet on scalp diseases related to hair growth. Diet is also able to strongly influence gut microbiome. On the contrary, few evidence reports as regards the link between microbiome, especially scalp microbiome and hair diseases. Here we reported a two case-reports study on patients affected by AA, with and without lactose intolerance, respectively, with the aim to underline how diet could emphasize microbiome changing related to scalp disease. Subjects were asked to fill out a 7-day dietary survey and scalp and oral swabs were collected. Data from the dietary survey, qRT-PCR on main bacterial strains inhabiting the scalp and 16S sequencing of the scalp and oral microbiome were matched and compared each other and with healthy and general AA population. Beyond diet well-known impact on general human health, our results highlighted the role of diet in modifying oral and scalp microbiome, which in turn seems to have an impact on AA evolution. The findings of the present works suggested a kind of intercorrelation between microbial dysbiosis on the scalp of patients with AA and dietary habits.
Alopecia areata, Hair disorders, Dietary therapy, Microbiome, Dysbiosis
Hair follicle is a dynamic mini-organ [1] with an high cellular turnover. As a consequence, hair follicle is characterized by a very active metabolism requiring a good nutrient's intake. Even though the role of macro and micronutrients in normal hair follicle development has not been completely clarified [2], the impact of diet and nutritional deficiencies on hair growth diseases is well documented [3,4].
Diet is also reported to have ability of shaping the gut microbiome [5,6] but also skin microbiome in relations to some dermatological conditions [7,8] especially acne and psoriasis. Poor knowledge is currently available as regards dermatological conditions affecting the scalp and hair growth.
Among hair growth disorders, Alopecia areata (AA) is reported as the second most common disorder affecting the scalp [9]. AA is a type of non-scarring baldness affecting the scalp and, eventually the entire body [10] which causes have been strictly associated with immunity and inflammation [9,11,12].
In the past two years researchers focused their attention also on the role of microbial community inhabiting the scalp and hair growth disorders, including AA [13-17]. More recently Nair and collaborator reported evidence as regards the role for the gut microbiome in the pathogenesis of AA [18]. Taking together these findings pose the need of a more deeply investigation as regards the link between diet, microbiome and hair growth disorders.
In the present work, we reported a two case-reports study on patients affected by Alopecia areata, with and without lactose intolerance, respectively, with the aim to underline how diet could emphasize microbiome changing related to scalp disease (Figure 1).
 Figure 1: Diet importance in microbiome changing related to scalp disease. View Figure 1
Figure 1: Diet importance in microbiome changing related to scalp disease. View Figure 1
A 17-year-old male (Milan, Italy), affected by Alopecia universalis (Figure 2). The patient presented to the dermatology clinic with a history of rapidly progressing total body hair loss. There was no history of similar illness in family members and also no history of drug intake and trauma. Previously treatment includes stem cell therapy. No therapy for at least 3 months at the time of inclusion. The clinical evaluation reported 100% hair loss of the scalp based on the Severity of Alopecia Tool (SALT) Score [19] with no signs of erythema or scaling. Eyebrows, eyelashes, and body hair were also completely absent. The patient demographics include being Caucasian, weight 72 kg, height 1.80 m, and BMI 22.2 kg/m2.
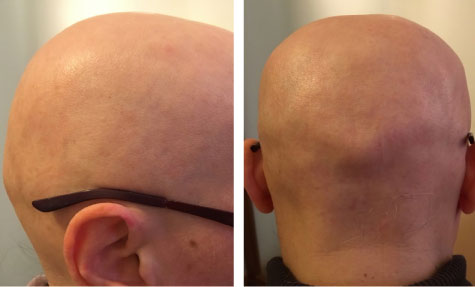 Figure 2: Alopecia universalis affected male. View Figure 2
Figure 2: Alopecia universalis affected male. View Figure 2
A 36-year-old female (Figure 3) came to the clinic reporting a history of strong hair loss since one month. Other reported symptoms are severe itching, psoriasis on the scalp, birch and pauliary allergy, insomnia and high sensation of fatigue. The patient is also intolerant to lactose. Clinical evaluation showed strong hair loss but no signal of miniaturization. Alopecia areata was confirmed by histological examination. No therapy for at least 3 months at the time of inclusion. The patient demographics include being Caucasian, weight 48 kg, height 1.62 m, and BMI 18.3 kg/m2.
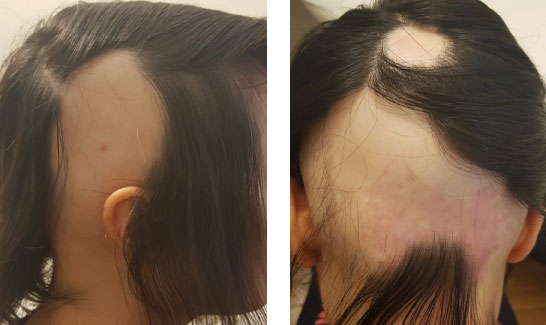 Figure 3: Women with initial stages of Alopecia areata. View Figure 3
Figure 3: Women with initial stages of Alopecia areata. View Figure 3
Both subjects were asked to fill out a 7-day dietary survey at the time of enrollment, following being instructed by a dietician on how to record the food and beverages consumed. The Winfood software (Winfood 2.7 Medimatica Srl, Colonnella, Italy) was used for analysis of surveys by estimating the energy intake and the percentage of macronutrients and micronutrients. Data collected were compared to the tables of food consumption and recommended dietary intakes of the Italian National Institute of Nutrition and Food Composition Database in Italy.
Before sampling patients had to avoid the use of antibiotics in the last 30 days no probiotics, the use of probiotics in the last 15 days, to perform the last shampoo 48 h before sampling. They also did not have to undergo to anti-tumor, immunosuppressant or radiation therapy in the last 3 months and also topical or hormonal therapy on the scalp. The study was under the approval of the Ethical Independent Committee for Clinical, not pharmacological investigation in Genoa (Italy) and in accordance with the ethical standards of the 1964 Declaration of Helsinki. All of the volunteers signed the informed consent.
Microbiome samples were collected from the scalp (minimum area sampled of 16 cm2) and oral mucosa with a sterile cotton swab, previously soaked in ST solution (NaCl 0.15 M and 0.1% Tween 20) for at least 30s [20,21]. Samples from the same subjects were collected together and stored at 4℃ until DNA extraction. Sterile cotton swabs placed in ST solution have been used as negative controls.
Genomic DNA from scalp swabs was extracted by mean of QIAamp UCP Pathogen Mini Kit (Qiagen) according to manufacturer protocol, with minor modifications [22]. After extraction, bacterial DNA was suspended in DNAse free water and quantified by the QIAexpert system (Qiagen) before sequencing and qRT-PCR.
For sequencing, variable region V3-V4 was amplified by mean of the following universal primers: 341 F CTGNCAGCMGCCGCGGTAA [23,24] and 806bR GGACTACNVGGGTWTCTAAT [25-27]. Library preparation and Illumina MiSeq V3-V4 sequencing were carried out at StarSEQ GmbH, Mainz, Germany, according to Caporaso, et al. [28] and Kozich, et al. [29] methods, with minor modifications. Real-Time Analysis software (RTA) v. 1.16.18 and 1.17.22, MiSeq Control Software (MCS) v. 2.0.5 and 2.1.13 were using.
Main bacterial species (Propionibacterium acnes, Staphylococcus epidermidis and Staphylococcus aureus) on the scalp were quantified by real-time quantitative PCR (RT qPCR), using Microbial PCR assay kit (Qiagen). Samples were mixed with 12.5 μL of Microbial qPCR Mastermix, 1 μL of Microbial DNA qPCR Assay, 5 ng of genomic DNA sample and Microbial-DNA-free water up to a final volume of 25 μL. Positive PCR Control, No Template Control, and Microbial DNA Positive Control were also included. Pan-bacteria assays are also included as positive controls for the presence of bacterial DNA, as human GAPDH and HBB1 for the determination of proper sample collection. Following thermal cycling conditions were used: 95℃ for 10 min, 40 cycles of 95 ℃ for 15 sec, 60 ℃ for 2 min. Each PCR reaction was performed in duplicate using an MX3000p PCR machine (Stratagene, La Jolla, CA). Relative abundance in the expression of each strain was calculated using the ΔΔCt method [30], normalizing fold-change against Pan Bacteria, using MX3000p software (v.3; Stratagene).
Data are expressed as Relative abundance % ± SEM for qRT-PCR analysis. Results were checked for normal distribution using the D'Agostino & Pearson normality test before further analyses. Statistically significant differences in the bacterial community were determined by Student's t with Welch's correction. Analyses were performed with GraphPad Prism 7.0 (GraphPad Software, Inc., San Diego, CA). P-values equal to or less than 0.05 were considered significant.
In the present work, we investigated diet impact on microbial dysbiosis caused by the presence of AA which in turn could impact on disorder evolution and manifestations.
The preliminary analysis of macronutrients % intake (data not shown) suggested a Mediterranean diet framework [31] for both case reports included in the present study. Case report 1 diet included processed food, red meat and low intake of fruits and vegetables. Case report 2 diet included vegetables, fruit, peas, and beans (legumes) and grains. When analyzed more deeply as regards the type of proteins, lipids and carbohydrates ingested, diet from case-report 1 is better classified as High fat-diet (more processed food, sugars, and few fibers). The second case reports can be considered, instead, as following the Mediterranean like diet (lower fruits intake compared to normal Mediterranean regimen).
Tables 1 and Table 2 show the intake of macronutrients and micronutrients in both case reports, compared to Recommended (LARN) values in Italy.
The daily amount of total calories was significantly different among case reports (p < 0.01) (1,331.58 ± 189.61 and 3.09 ± 143.48, respectively) (Table 1). Food diary from both subjects also reported a very small intake of fiber (16.59 ± 13.23 and 7.44 ± 1.33, respectively) and polyunsaturated fatty acids (6.45 ± 1.31 and 6.36 ± 1.82, respectively). Therefore, a lower percentage of saturated fatty acid intake was reported for case report 2 (3.12 ± 1.00).
Table 1: Daily reported energy and nutrient intake of studied case reports, assessed by a 7-day weighed food record. LARN: Nutrition and Energy Reference Assuming Levels. a-cValues with different superscript letters, in the same row, differ significantly (P < 0.05). View Table 1
As regards micronutrients intake (Table 2) we noticed significant lower intake of iron (5.50 ± 1.66 and 4.26 ± 1.91 vs. recommended) (p < 0.001) and folic acid (149.25 ± 43.89 and 67.33 ± 24.08 vs. recommended) (p < 0.01), riboflavin (0.45 ± 0.11 and 0.46 ± 0.25 vs. recommended) (p < 0.01) and vitamin D (0.78 ± 1.48 and 4.58 ± 6.55 vs. recommended) (p < 0.01) for case report 1 and case report 2, respectively. Case report 1 also has a significant (p < 0.01) lower intake of niacin (7.79 ± 3.45) and vitamin E (3.21 ± 2.57) and higher (p < 0.001) intake of vitamin A.
Table 2: Daily reported micronutrient intake of studied case reports, assessed by a 7-day weighed food record. LARN: Nutrition and Energy Reference Assuming Levels. a-cValues with different superscript letters, in the same row, differ significantly (P < 0.05). View Table 2
Unbalancing in nutrients intakes is reported to have an influence both on hair follicle structure and hair growth diseases such as telogen effluvium, androgenetic alopecia, AA and cicatricial alopecia [4,32-35].
Indeed, many of the above cited micronutrients are reported to affect the hair follicle as regards restoration of hair growth, cell division, cycling [3]. Therefore, Singh and collaborators [5] also highlighted diet effect on gut microbiome. The same effect has been reported on oral microbiome [36].
Evidence on the influence of diet and microbiome dysbiosis on skin disorders are mainly linked to topic dermatitis [37], acne vulgaris [8,38-40], and psoriasis [41]. In our previous works [14,15] on scalp microbial community in hair growth disorders we showed, for the very first time, the presence of scalp bacterial shift in such kind of disorders.
Figure 4 reports the % of the distribution of main bacterial strains in case report 1 and case report 2 compared with data from 15 healthy subjects and 15 AA subjects in our database.
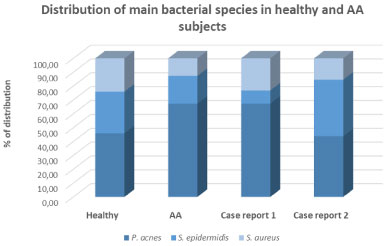 Figure 4: Distribution of main bacterial strains in healthy subjects and AA subjects. View Figure 4
Figure 4: Distribution of main bacterial strains in healthy subjects and AA subjects. View Figure 4
Data from case report 1 showed an increase in P. acnes population and parallel decrease both of S. epidermidis and S. aureus species. These data are in line with data from the panel of AA subjects and clearly evidence the presence of bacterial dysbiosis compared to healthy control.
On the contrary, the percentage of distribution of main bacterial species in case report 2 resulted more similar to the healthy population (Figure 4). Even if an interindividual difference has to be considered, the analysis of food diary of the panel of fifteen AA subjects and case report 1 and 2 strongly suggested the impact of diet in shaping scalp microbiome.
Data from oral bacterial DNA sequencing corroborated these findings (Figure 5). Also, in this case, data from case report 1 and 2 were compared to data from our internal database of healthy and AA subjects, previously collected. The analysis of sequence at the phylum level highlighted a slow decrease of Firmicutes both for case report 1 and 1 (Figure 6) and these results are in line with our previous findings in AA subjects and results found in another autoimmune disease [42]. In both case reports, an increase of Proteobacteria (33.28% and 27.71%, respectively) has been reported. Most interesting, analysis of sequences from case report 1 bacterial oral DNA showed a decrease in Bifidobacteria. A link between a high-fat diet and this phylum decrease was previously reported [43] thus confirming the role of diet in influencing oral bacterial composition. A significant reduction of Bacteroidetes has been found in case report 2 compared to healthy control subjects. Since the high intake of n-6 PUFA by case report 2, a link between diet and this microbial unbalance could be hypothesized as also suggested by some authors [44].
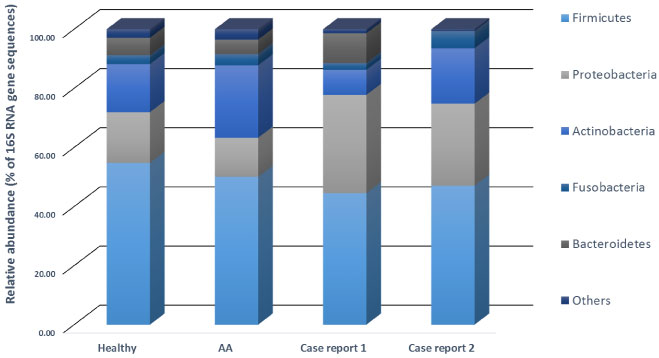 Figure 5: Data from oral bacterial DNA sequencing corroborated these findings. View Figure 5
Figure 5: Data from oral bacterial DNA sequencing corroborated these findings. View Figure 5
Results on bacterial composition of scalp microbiome confirm our previous findings on microbial shift on the scalp in patients affected by AA [14,15]. In the present work, we investigated if different dietary habits can re-modulate this microbial dysbiosis with the aim to highlight the strict intercorrelation between diet and oral but especially scalp microbiome of these subjects. The case reports of the present work represent just an example of a larger clinical investigation we are leading on this topic. For example, our clinical observations suggest that, in some patients affected by non-celiac gluten sensitivity (NCGS), AA manifestations systematically recurred following a non-gluten free diet. An explicative photographic example was reported in Figure 6. Most interesting this modulation reflects also in the microbial composition of scalp microbiome (data not shown) enhancing the existence of a link between diet and skin bacterial communities scalp microbiome.
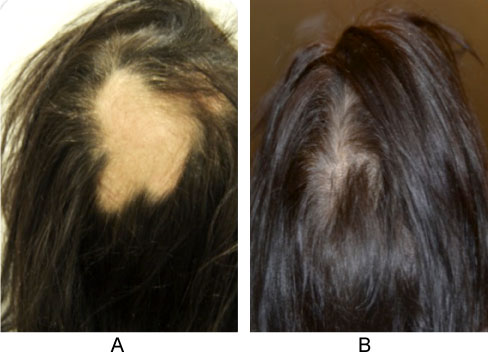 Figure 6: A slow decrease of Firmicutes. View Figure 6
Figure 6: A slow decrease of Firmicutes. View Figure 6
Data from the present study add to knowledge to this evidence also highlighting that not only gut but also oral and scalp microbiome could be modulated by dietary habits.
Nowadays, the study of human microbiome represents a novel diagnostic and therapeutic approach to treat many human conditions, also including that strictly related to skin and scalp. Beyond diet well-known impact on general human health, our results highlighted the role of diet in modifying oral and scalp microbiome, which in turn seems to have an impact on Alopecia areata evolution.
According to our findings and previous reported evidence cited above, the modulation of the gut microbiome by mean of diet could represents a valid approach in the managing of hair growth disorders, especially AA, in which also the permeability of the gut can be compromised [44].
This study was supported by Giuliani SpA.
R.F. and S.E. serve as a consultant for Giuliani S.p.A. P.D. is employed by Giuliani S.p.A.