Herein is a case of systemic lupus erythematosus (SLE) who developed myasthenia gravis (MG) and presented with proximal muscle weakness and easy fatiguability. Although inflammatory polymyositis is a common cause of muscle weakness in SLE, MG is not frequently reported in SLE. The diagnosis was established by positive Antibodies to acetylcholine receptors, Electroneuromyography (ENMG). Her computed tomography (CT) scan of the chest showed nodular soft tissue in the anterior mediastinum. She was successfully treated with intravenous immunoglobulin (IVIG). A thymectomy was performed.
Systemic lupus erythematosus, Myasthenia gravis, Autoimmunity
Systemic lupus erythematosus (SLE) is a chronic multi-systemic autoimmune disease that may affect any organ of the body. Neurological manifestations are common and the American College of Rheumatology (ACR) has defined 19 neuropsychiatric syndromes that may occur in patients with SLE [1].
Myasthenia gravis (MG) is an autoimmune neuromuscular disease due to a defect of neurotransmission induced by autoantibodies mainly directed against acetylcholine receptor (AChR) or more rarely against another post synaptic molecule, MUSK (muscle specific kinase) [2]. These two diseases show similarities in that they occur mainly in young women, are characterized by periods of exacerbation and remission, and by production of autoantibodies [3].
Although both MG and SLE are relatively common autoimmune disorders, their coexistence is rarely reported, and most of published studies are case reports.
In this article we describe a patient with systemic lupus erythematosus who developed myasthenia gravis 10 years after her SLE diagnosis.
A 30-year-old female with past medical history of SLE presented to the hospital with the chief complaint of shortness of breath, intermittent fatigability, and muscle weakness, mainly in her lower limbs, for two months duration.
She was diagnosed with SLE 10 years before, her initial clinical findings were photosensitivity, malar rash, arthritis, pericarditis, and pleurisy. Her laboratory investigations at the time of diagnosis showed positive anti- nuclear antibody (ANA), positive anti-double stranded DNA (anti-ds DNA), and positive anti-Smith antibody. Rheumatoid factor (RF) was negative. Her initial complement levels showed C3, 73 mg/dL (reference value: 88-201 mg/dL), C4 11 mg/dL (reference value: 14-39 mg/dL). Complete blood count (CBC) showed mild leucopenia, and lymphopenia, erythrocyte sedimentation rate (ESR) was highly elevated (122 mm/hr). Her 24 hours urine protein (573 mg/24h).
She was treated initially with prednisone (40 mg/day), and hydroxychloroquine (400 mg/day) with improvement. During her follow-up, the main symptoms were joints pain, and back pain. Her musculoskeletal examination and radiologic evaluation showed non-erosive inflammatory arthritis, she also had fibromyalgia picture with widespread pain and positive tender points. Magnetic resonance imaging (MRI) of her lumbar spine showed congenital spine stenosis with no cord compression or significant neural foramina narrowing. Methotrexate (15 mg/week) was added for her joint's symptoms, but she developed leucopenia so it was discontinued and replaced with azathioprine (100 mg/day), her prednisone dose was tapered down to (7.5 mg/day), she had intermittent flares in her joints symptoms that were treated with temporary increase in her prednisone. Her fibromyalgia symptoms were treated with pregabalin (150 mg twice/day), and duloxetine (60 mg/day). For her initial proteinuria she was placed on lisinopril (5 mg/day), her urinary protein decreased, and kidney biopsy was not done.
Her physical examination on admission showed proximal muscle weakness around her shoulders, and pelvic girdle, walking difficulty, and bilateral incomplete eyelid ptosis. Her oxygen saturation on room air was low (87%), her arterial blood gas (ABG) showed pH (7.35), PCO2 (60.7 mmHg), PO2 (63.8 mmHg), HCO3 (33.4 mEq/L). Her CK levels were normal. Chest x-ray showed low lung volume, mild bilateral pleural effusion, computed tomography angiography (CTA) of the chest was done to exclude pulmonary embolism and it was negative, computed tomography (CT) scan of the chest showed nodular soft tissue in the anterior mediastinum, small bilateral pleural effusion, and small pericardial effusion (Figure 1).
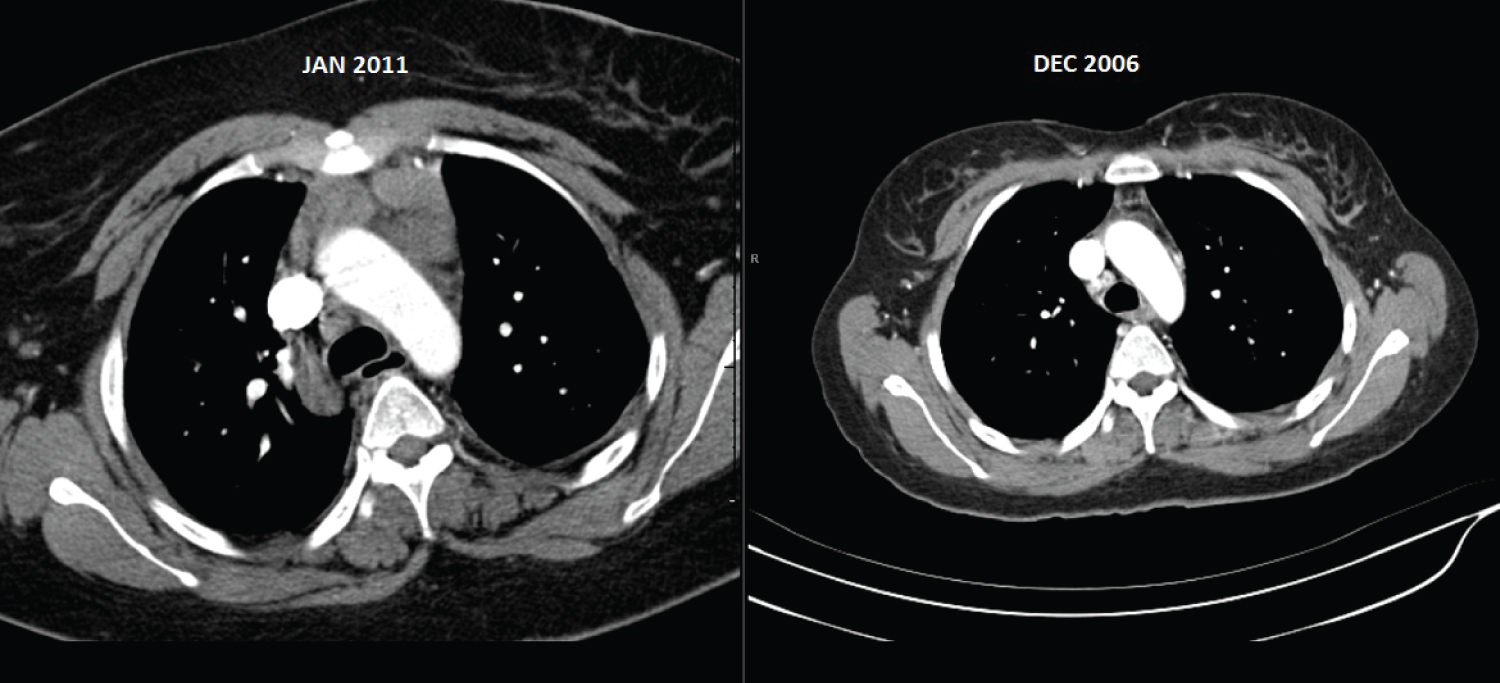 Figure 1: CT chest in 2011 with nodular soft tissue in the anterior mediastinum, CT chest in 2006 with no changes in the anterior mediastinum.
View Figure 1
Figure 1: CT chest in 2011 with nodular soft tissue in the anterior mediastinum, CT chest in 2006 with no changes in the anterior mediastinum.
View Figure 1
Electroneuromyography (ENMG) with repetitive nerve stimulation showed post synaptic neuro muscular defect with decremental response. Antibodies to acetylcholine receptors (AChR) was positive, > 80 nmol/L (reference value positive > 0.40 nmol/L). These findings confirmed the diagnosis of MG, as a cause of her new onset muscle weakness and respiratory symptoms.
She was treated with intravenous immunoglobulin (IVIG) 500 mg/kg per day for 4 days, and pyridostigmine 90 mg every 6 hours. Her muscle weakness and respiratory symptoms, rapidly improved. She was maintained on her previous medications, prednisone (10 mg/day), azathioprine (100 mg/day), and hydroxychloroquine (400 mg/day) in addition to pyridostigmine. A thymectomy was performed and follicular hyperplasia was found in histology.
She was followed in the rheumatology clinic for 5 years after the diagnosis of MG, with no change in the clinical manifestations of her SLE.
The association of MG and SLE was previously reported, however there is limited number of reported cases. Jallouli, et al. [4] reported a series of 17 patients with SLE and MG among a cohort of approximately 1,300 SLE patients. The prevalence of MG in their SLE population was estimated as 1.3%, which is higher than the 0.02% prevalence of MG in general population. In their research they found 53 similar cases in the literature for a total of 70 cases.
In most patients (62%), MG appeared first, and it was hypothesized that thymectomy in MG patients could be a precipitating factor for the development of SLE due to the loss of central tolerance and the overproduction of antibodies. Given that our patient was diagnosed with SLE 10 years before the diagnosis of MG, the role of thymectomy was not a factor in our patient. Our patient had ac CT chest done 4 years before the diagnosis of MG for evaluation of chest pain that did not show the nodular soft tissue in her mediastinum that was shown later when she was diagnosed with MG, which suggest that MG was not evolving at the time of her SLE diagnosis.
The main clinical manifestations of SLE in our patient was inflammatory arthritis that was adequately controlled with hydroxychloroquine, azathioprine and low dose prednisone and this clinical picture did not change after the diagnosis of MG and thymectomy. It was suggested that the association between SLE and MG may be due to common genetic predisposition and immunological abnormalities [5] including dysfunction of regulatory CD4+ CD25+ T lymphocytes [6], high B-cell chemoattractant of CXC chemokine (CXCL13) production [7].
In the study of Jallouli, et al. patients with MG and SLE were predominantly female (93%), and this was true for patients diagnosed with MG after the age of 40 years, contrasting with what is usually observed among unselected MG patients. Indeed, it has been reported that MG is nearly three times more frequent in women during early adulthood, whereas no sex predominance is observed after the age of 40 years [8]. They found that 84% of patients were less than 40-years-old at MG onset, compared with 68 and 54% in large series of 756 and 1,298 unselected MG patients, respectively [8,9]. The clinical presentation of MG, the percentage, and the histological results of thymectomy did not differ than from large cohort of unselected MG patients [8]. The prevalence of AChR antibodies (94%) was higher than the one reported in unselected MG patients, which is around 85% [10].
In SLE patients who developed MG the most common manifestations were arthritis (88%), and skin rash (23%). Positive antinuclear antibodies were reported in all patients, anti dsDNA antibodies in 82%, anti-SSA in 35%, anti-Sm in 23%, anti-SSB in 12%, and anti-RNP in 12% [4].
A Swedish population-based study found an increased frequency amongst patients with MG of other autoimmune disease, in particular those conditions regulated by HLA-B8-DR3 [11]. In a study conducted in western Denmark during the period 1975-1989, 14% of prevalent MG cases had another autoimmune disease, with thyroid disorders and rheumatic arthritis being most common [12].
The clinical picture of weakness due to muscle disease in SLE patients is usually attributed to etiologies other than MG. Mainly inflammatory polymyositis, steroid induced myopathy, and antimalarial drugs induced neuromyotoxicity. These disorders usually lack the classic ocular findings of MG, and its characteristic electromyogram pattern.
In our patient the treatment with hydroxychloroquine was continued after the diagnosis of MG without worsening of her MG, her routine eye monitor did not show signs of retinopathy.
This case emphasis that SLE patients with proximal muscle weakness should be referred for an EMG study and determination of acetylcholine receptor antibody to confirm or exclude MG.
The increased prevalence of autoimmune disease in MG patient, suggest that they should be watched for the possible development of autoimmune disease including SLE.
The author declares that he has no conflict of interest.
A written consent for publication was obtained from the patient.