The aim of this study was to evaluate the patterns of recurrence and metastatic spread of breast cancers in relation to subtypes and elapsed time since original cancer, to guide follow up imaging.
Between June 2011 and December 2016, all patients with local breast recurrence or distant recurrence after a previous breast cancer had Computed tomography of chest, abdomen and pelvis (CT TAP). All locally advanced primary breast cancers (LABC) also had CT TAP as part of staging investigations. Time to recurrence was stratified in three time frames- ≤ 5 yrs; 6-10 yrs; > 10 yrs. Review of recurrence data, both local and systemic, along with metastasis to organ-specific sites was undertaken.
There were 201 patients with CT TAP in the study group, of which 79 (39.3%) patients had distant metastases. The most significant association was between HER2-positivity and the liver metastases (p = 0.02). Fifty patients (24.7%) had recurrence of cancer. Of these, 24/50 (48%) had local recurrence only, 16 (32%) had distant relapse only and 10/26 (20%) had both local and distant relapse. Time line of recurrence at ≤ 5 yrs; 6-10 yrs; > 10 yrs for local and distant relapse was 13:4:7 and 16:6:4 respectively. 80.4% of local recurrences were symptomatic at the time of presentation.
This study questions the role of routine 'mammogram only' follow in all types of breast cancer. Certain subtypes like HER2/TN could benefit from systemic imaging, along with faster referral after symptoms.
Breast cancer is the most common cancer in women, accounting for approximately 15% of all cancers [1]. Over 85% of these people survive beyond 5 years after diagnosis, due to continual progressions in early detection and treatment quality [1]. Despite advances in detection and treatment, 20-30% of patients develop local relapse or distant metastasis [2]. Molecular subtyping of breast cancers based on receptor expression has been introduced in clinical practice and has developed into an important tool in predicting prognosis and constructing personalised management plans for breast cancer sufferers [3].
Receptor status with respect to oestrogen (ER), progesterone (PR) and Human epidermal growth receptor 2 (HER2) can be determined through immunohistochemical staining and treatment can be targeted to treat the specific subtype of cancer. The four major molecular types of breast cancer differentiated by sub-typing are ER positive/HER2 negative, ER/HER2 positive, ER negative/HER2 positive and ER negative/HER2 negative (Triple negative -TN) which itself may be further subdivided into basal-like and TN non-basal [4]. This histoclinical classification system is the most commonly used system in routine clinical practice. Studies have shown that ER-positive cancers are the type associated with the lowest risk and TN cancers are far more aggressive than other types of cancer, with respect to local or distant relapse [5,6]. However, many earlier studies lack data on HER 2 positive cancers, which is crucial due to recent advancements in HER2 cancer treatment. In addition, to the best of our knowledge, there is limited data depicting clinical symptoms at the time of relapse and time interval of local and distant recurrences with respect to different cancer subtypes.
Imaging plays a critical role in management of breast cancer patients, not just at the time of diagnosis but also during follow up. Routine annual mammograms are offered to women after breast cancer treatment to detect local and contralateral recurrence. There is currently no universally agreed imaging monitoring guideline to detect systemic relapse for breast cancer survivors. Knowledge of pattern and timing of relapse could guide the radiologist to deliver individualised care based on the subtype of cancer. This knowledge can also be used to direct personalised monitoring with imaging to ensure further improvements in outcomes for breast cancer patients.
This study involved the retrospective evaluation of data between June 2011 and December 2016 of all patients with local breast recurrence or possible distant recurrence after a previous breast cancer. All had Computed tomography of chest, abdomen and pelvis (CT TAP). All locally advanced primary breast cancers (LABC) also had CT TAP as part of staging investigations. LABC was defined as clinical N2, N3 or T4 stage, according to the staging system of the American Joint Committee on Cancer [7]. The need for obtaining informed consent or ethical approval was waived due the retrospective nature of the study, as per local policy. For the purpose of this study, a biopsy proven second breast cancer (ipsilateral or contralateral to site of previous breast cancer), irrespective of stage and time of primary cancer or recurrence was classified as recurrent cancer and eligible for CT TAP. All study patients were discussed in Breast Multi disciplinary team meeting (MDTM). Exclusion criteria included all patients with missing clinical details or incomplete data on receptors or incomplete work up of primary or recurrent cancer. Patients were grouped based on the indication for CT as primary breast cancer staging, local or distant recurrence.
Metastatic disease was defined as breast cancer beyond the confines of the ipsilateral breast, regional lymph nodes and chest wall. Metastatic sites were classified as bone, liver, lung, brain, pleura/peritoneum, extra axillary lymph nodes and others. Patients with both local and distant recurrence were noted and patients with multiple sites of metastases were also recorded. In cases of local recurrences, age of the patients, type and grade of primary cancer, presence of DCIS, lympho-vascular invasion in primary cancer, nodal staging and symptoms at the time of relapse were also recorded. Treatment given to the primary cancer was also recorded. Time interval since the primary diagnosis and receptor status of cancers was recorded. Time interval from the primary cancer was stratified in three time frames; 5 or less years, 6-10 years and more than 10 years (≤ 5 yrs; 6-10 yrs; > 10 yrs).
The immuno-histochemistry result for all cancers was recorded. ER positivity was defined as nuclear staining in 10% or more of tumour cells. Scoring of HER 2 was performed with immunohistochemistry or by Flourescent-in-situ hybridization (FISH) in borderline cases.
SPSS version 21.0 (IBM Corp, Chicago, USA) statistical software was utilised to statistically analyse data. Association between the molecular subtype and metastatic behaviour was assessed using either Chi square and Fischer exact test. All statistical tests were two sided and p < 0.05 was taken as statistically significant.
Between 2011 and 2016, there was an active pool of greater than 1500 patients for the study with a median follow up of 8 years. Out of these, 248 patients were eligible for CT based on either biopsy proven local recurrence, symptoms raising the suspicion of distant metastases or locally advanced primary breast cancer. From the total patient group (n = 248), patients with missing data were excluded from the analysis. Following these exclusions, there were 201 patients with adequate data for analysis. The median age of the patients in the study group was 60 yrs (range 24-96 yrs). Out of 201 eligible patients, 111 (55.2%) were oestrogen receptor positive, 25 (12.4%) were both oestrogen and HER2 receptor (ER/HER) positive, 20 (10%) were HER2 enriched (HER2+) and 45 (22.4%) were ER-/HER2-, classed as triple negative (TN). The majority (70.1%) of breast cancers were of Ductal type. The most common grades of cancers found were grades 2 and 3, with percentages of 50.8% and 43.7% respectively.
Out of all patients (201), 79 (39.3%) were found to have metastases on CT TAP; of which 53 (67%) were detected at the time of the initial staging investigations and 26 (32.9%) were detected in patients with a previous primary cancer, and were therefore treated as relapse. Patients with systemic relapse presented with vague symptoms of back or chest pain and CT TAP was ordered on the suspicion of distant metastases by the surgeon and agreed in the MDTM. Patients had equivocal imaging findings on CT TAP in 29 (14.4%) of cases, which were all later found to be benign on follow up imaging, most of which included indeterminate lung nodules and liver cysts. TN cancers had the highest proportion of metastases and the lowest mean age at presentation compared to other subtypes (Table 1). Bone was found to be the most common site of metastasis from a primary breast tumour (46%), followed by lung (40.3%) and then liver (31.6%) (Table 2). ER positive breast cancers showed increased bony metastasis but this was not statistically significant (p = 0.44) (Figure 1). HER2 status positivity was significantly associated with liver metastasis (p = 0.02) (Figure 2). There was an association between TN cancers and brain metastases (p = 0.041), however since there was an expected count of < 5, a Fischer Exact Test was used, showing that this association was statistically insignificant (p = 0.075) (Figure 3). TN cancers did however have a statistically significant association with extra-axillary (p = 0.00) and mediastinal/pleural (p = 0.00) metastases. The most infrequent site to be invaded was the brain (5%), mostly from TN breast cancer (Figure 4).
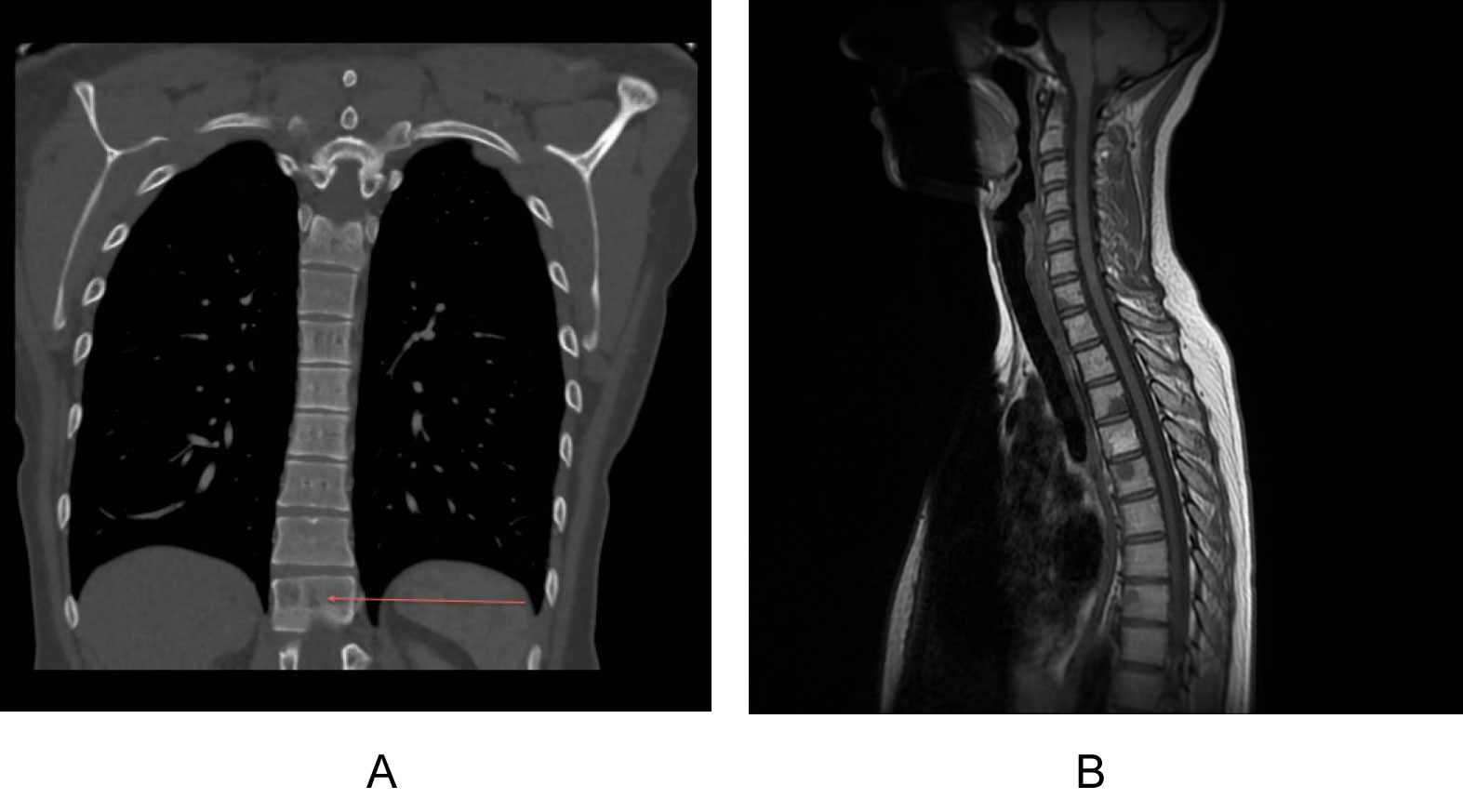 Figure 1: Computerised tomography staging scan in an ER+ cancer with indeterminate bony lesion (red arrow).
Figure 1: Computerised tomography staging scan in an ER+ cancer with indeterminate bony lesion (red arrow).
Computerised tomography staging scan in an ER+ cancer with indeterminate bony lesion (red arrow). View Figure 1
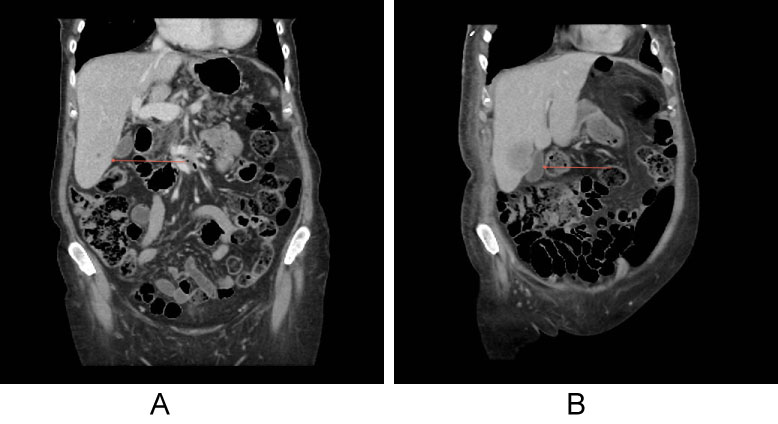 Figure 2: Computerised tomography staging in a HER+ cancer with hypodense liver lesion, which was too small to characterise (red arrow).
Figure 2: Computerised tomography staging in a HER+ cancer with hypodense liver lesion, which was too small to characterise (red arrow).
Three months later CT showed increase in size of the lesion suggestive of liver metastases. View Figure 2
 Figure 3: Multiple brain metastases in a triple negative breast cancer. This patient also had bony metastases (not shown). View Figure 3
Figure 3: Multiple brain metastases in a triple negative breast cancer. This patient also had bony metastases (not shown). View Figure 3
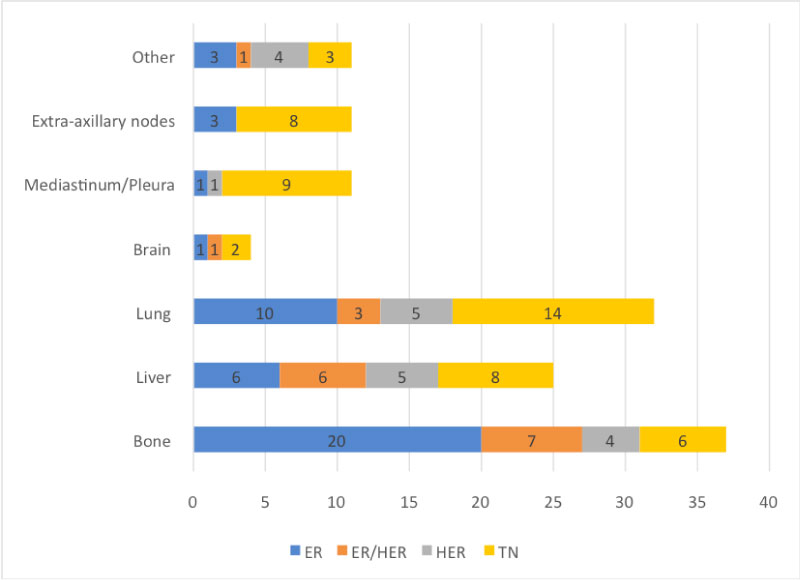 Figure 4: Proportions of organ specific metastases linked to specific molecular subtypes. View Figure 4
Figure 4: Proportions of organ specific metastases linked to specific molecular subtypes. View Figure 4
Table 1: Recurrence and metastasis in subtypes of breast cancer. View Table 1
Table 2: Organ-specific metastases in different subtypes of breast cancer (the two most common sites of metastases within each breast cancer subtype is depicted in bold). View Table 2
With respect to multiple metastases (bone, liver and lung), it was found that HER2-positive and TN cancers seemed to have the greatest proportion of multiple metastases. ER+/HER2+ breast cancer was associated with the most combined bone and liver metastases, TN with liver and lung metastases and HER2+ cancer with bone and lung metastases. ER-positive cancers had least number of multiple metastases. Due to the low number of multiple major metastases in the group, statistical significance between specific breast cancer subtypes and sites of multiple metastases was not evaluated (Figure 5).
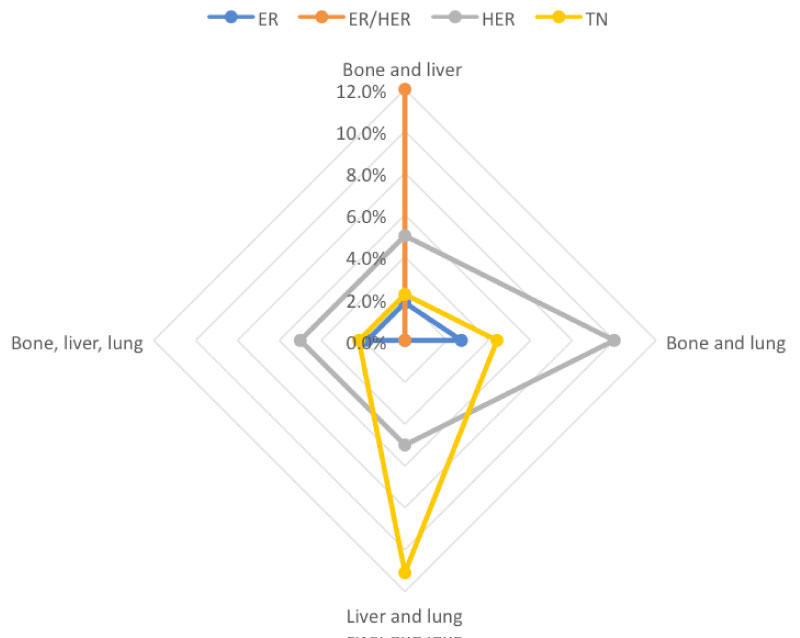 Figure 5: Diagram depicting multiple metastases in different subtypes of breast cancer. View Figure 5
Figure 5: Diagram depicting multiple metastases in different subtypes of breast cancer. View Figure 5
Out of the total of 201 patients in the study group, 50 (24.7%) patients had recurrent cancers. Of these 50 patients, 24 (48%) had local recurrence only, 16 (32%) had distant relapse only and 10 (20%) had both local and distant relapse. Majority (80.48%) of local recurrence were symptomatic with lump being the most common presentation followed by swelling, rash, pain or thickened scar. Rest of the recurrences (20.52%) were detected on mammographic follow up. The receptor type matched between primary and recurrent local relapse in 88.24% of patients. Lymphovascular invasion (LVI) and ductal cancer in situ (DCIS) was present in 58.63% and 72.26% of primary breast cancers respectively. 30% of primary cancers were lymph node positive, 62.1% had conservative surgery as primary treatment and 54% had received radiotherapy on the side of local recurrence. Within the local recurrence category, majority relapsed within 5 years from the primary diagnosis (13 patients), followed by more than 10 years (7 patients) with 4 patients in the 6-10 years time interval. In patients with distant relapse, ≤ 5 yrs; 6-10 yrs; > 10 yrs was 16:6:4 respectively.
Figure 6 depicts the recurrences with respect to different receptor subtypes. Median time till recurrence for HER2 cancers was 4 years (range 1-8 yrs). Most (6/8) of the recurrences of HER2 or ER/HER2 cancers were both systemic and local. Median time till recurrence was 3.1 yrs (range 1-15 yrs) for Triple negative (TN) cancer, which also had the highest percentage of recurrence (35%) (Table 1). Numbers were small in the subgroup analysis to calculate statistical significance for time trends.
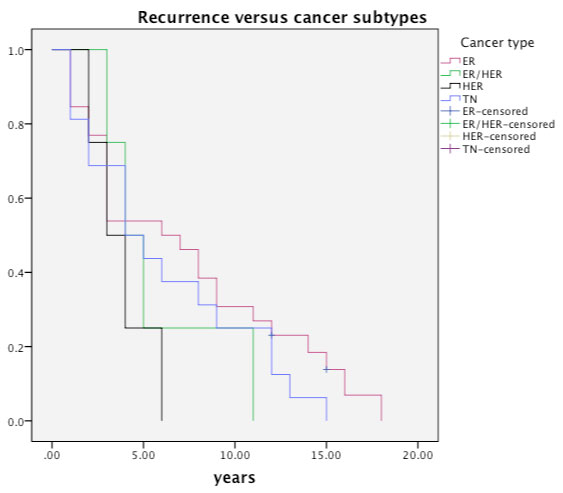 Figure 6: Recurrences with respect to different receptor subtypes of breast cancer (TN-Triple negative cancer; ER - oestrogen positive cancer; HER2- HER2 positive cancer; ER/HER2- Hybrid cancers). View Figure 6
Figure 6: Recurrences with respect to different receptor subtypes of breast cancer (TN-Triple negative cancer; ER - oestrogen positive cancer; HER2- HER2 positive cancer; ER/HER2- Hybrid cancers). View Figure 6
Breast cancer is a heterogeneous entity and has a distinct pattern of recurrence and development of distant metastases. There is a difference amongst subtypes of breast cancer with respect to time interval from presentation to occurrence of local or distant relapse [5,6]. Imaging plays a crucial role both at the time of initial diagnosis/staging and during follow up to detect local or systemic relapse. The results of this study can have important clinical implications. Having a propensity to metastasize to a particular visceral organ can forewarn the reporting radiologist to further investigate indeterminate lesions. A distinct time line from the primary diagnosis to recurrence could also lead to low threshold for investigating symptomatic patients within the time frame, leading to personalised care. The guidelines for monitoring breast cancer survivors in different countries are variable, with results from multi-centre studies like Mammo-50 [7,8] still awaited. Some countries like UK offer national guidelines regarding mammographic follow up of patients with breast cancer to detect local relapse [9]. However, it is important to bear in mind that these guidelines suggest annual mammographic follow up for 5 years [9] which detects local or contralateral relapse only. There is currently no universal consensus policy regarding systemic imaging follow up, especially since half of relapses were systemic in our series and detected on CT TAP. It is debatable whether the follow up regimen could be modified according to the subtype of breast cancer and if certain types of breast cancers could benefit from supplemental systemic follow up for 5 yrs. Our data suggests that certainly for HER2 and TN cancers supplemental systemic follow up imaging for 5 yrs would be beneficial. Larger studies are needed before we can put this into clinical practice.
This study demonstrated a statistically significant association between HER2 positivity and liver metastases in breast cancer, supporting previous evidence linking HER2 status and secondary liver cancer [10]. Research has shown that HER2 expressing breast cancers tend to spread to the liver more so than hormone-positive cancers [11-14]. In a study by Savei-heijink, et al. [15] involving 263 patients, authors evaluated metastatic behaviour of breast cancer subtypes. They did not find HER2 positivity as a strong predictor for site-specific metastases (liver or brain). The fact that none of the patients in their study received adjuvant HER2 targeted therapy may have played a role in their findings.
ER positive tumours are known to relapse later (upwards of 10-20 yrs) with higher rate of bone recurrences [11,12,16], similar to our experience. We found ER status to be an important predictor for bone only or bone first metastases, in agreement with other studies [11,12]. The high proportion of metastases appearing in bone supports current compelling evidence that bone is the most common area of metastasis from a breast primary [17]. Due to high number of false negatives and false positives on Tc-99m bone scans, the Royal College of Radiologists, London has recommended CT as a technique of choice for staging asymptomatic locally advanced breast cancer, with the use of bone scan and MRI as a problem solving tool in selected patients only [18,19].
The association between TN cancers and extra-axillary nodal and lung metastasis is supported by previous research [11,19], with statistically less chance of involving the liver or bone.
Similar to our study, the incidence of brain metastases in breast cancer patients varies from 5 to 30%, higher in TN breast cancer and HER2 positive cancers treated with Trastuzumab [5,12]. For HER 2 positive cancers, higher rate of CNS relapse has been demonstrated due to Trastuzumab inability to cross the blood brain barrier [20-22]. The link between TN cancer and brain metastasis was depicted in a study [23], where microarrays of brain metastases sufferers showed that the majority were within TN cancer patients, which was then supported by a further study which found that 46% of a specific population of TN breast cancer sufferers had brain metastases seen at the time of death [5].
In this study, TN cancers generally affected younger patients and relapsed early, consistent with current literature [5,6]. There is also a link between TN patients (commonly containing BRCA1 gene mutations) and metastasis [24]. These patients also had the highest proportion of recurrence and lowest median time to recurrence, which tends to occur within 5 years of initial diagnosis [25,26]. Studies have also demonstrated that TN cancers are more likely to present at physical examination rather than mammographic screening [10]. Moreover, TN breast cancers are more likely to develop distant metastasis before loco-regional recurrence [10,26], similar to our study. These results question the role of 'mammogram only' follow up in TN and HER2 subtypes of cancers and could be the domain of future studies.
Also in the present study, we found that most of the local recurrences presented with lump or pain, following which they were seen on screening mammograms or ultrasound. Our findings are similar to Chu, et al. [27], who found that in their 147 cases with recurrent breast cancer over 10 year period, imaging surveillance with supplemental imaging modalities would be beneficial for HER2 cancers but not in TN cancers (which presented with lumps).
Recurrence timeline of different subtypes of breast cancer suggest majority of recurrences for all subtypes within 5 years of primary diagnosis, although there is trickle beyond 5 years, which continues beyond 10 years. This is in keeping with current national guidelines regarding annual mammographic follow up till 5 years [9], that would pick up majority of local recurrences. Continuing annual mammographic screening beyond 5 years is contentious in an era of limited resources, more so when mammographic follow up does not pick up systemic recurrences and could falsely reassure patients. Moreover, local recurrences are more likely to be symptomatic, as shown in our study, underscoring the questionable role of mammograms only follow up without physical examination.
This study has certain advantages. Firstly, it is a large selective series focussing on the locally advanced and recurrent breast cancer. As recurrent cancers could occur in early as well as advanced primary cancer, by focussing this study on recurrences, we have focussed on the relevant group of patients. Moreover, all symptomatic patients where the possibility of metastases was raised were included in this study. Some of these were early stages cancers. In this respect, we have included the relevant group with high likelihood of relapse. Secondly, being a more recent series, the treatment received by most patients was standardised, in contrast with earlier series where HER2 targeted therapy may be lacking or newer generation of chemotherapeutic regimen was not utilised. Thirdly, we believe we have captured all cancers diagnosed and treated in the local community, as all cancers in the community were discussed in the multi-disciplinary meeting.
There are certain limitations to this study. Firstly, this study involved retrospective evaluation of data. Large prospective studies with longitudinal follow up over 15-20 years could be an optimal method of evaluating trends but difficult to organise. Secondly, PR statuses were not recorded, as the Trust's standard procedure for pathological analysis did not involve PR status analysis. Thirdly, Triple negative cancer is a heterogeneous group of tumour and it has been claimed that Triple negative cancer cannot be used as a surrogate for basal like features. Further studies are suggested to explore the role of immuno-histochemistry to further evaluate group heterogeneity within the triple negative group. Gene expression signatures and novel molecular mediators have shown to have associations with site-specific metastases but this is in research stage and translation to clinical practice has not yet been accomplished. Fourthly, diagnosis of metastases was an imaging based decision and histological proof was not available. In addition, conventional imaging may not have detected all signs of metastatic disease, so cases of subclinical metastasis may have been missed. Also, we have not evaluated different types of breast cancers based on ki67 status. Although these may be useful in the future for prognosis and treatment, at present, receptor status in the most commonly used system in most centres to evaluate breast cancers.
Lastly, this was a selective group of breast cancer with high likelihood of recurrence and metastases. Some early stage cancers with asymptomatic local or distant recurrence may have not been included in this analysis. It is also beyond the scope of this study to perform cost-effective analysis of adding CTTAP to the surveillance protocol but could be the domain of future studies.
Although, it is difficult to recommend imaging surveillance strategy based on this retrospective study with small numbers, we believe there is a void in the scientific domain in this field and future studies should look into evaluating the role of supplemental imaging surveillance with CT TAP with fast tracked physical examination with or without mammograms in patients with HER2 positive and TN cancers.
In summary, the molecular subtypes of breast cancer have distinct patterns of metastasis and clinical outcomes. This study questions the role of 'mammogram only' follow up in HER2 and Triple negative cancers and authors suggest more studies to evaluate the role of supplemental systemic follow up with CT TAP in these subtypes. It is prudent for radiologists to understand metastatic patterns in breast cancer subtypes and carry out appropriate and timely monitoring, especially for those at high risk of breast cancer relapse, so that future treatment and prognoses can continue to advance.
To all the patients and staff of the Breast centre.
The authors declare no conflict of interest or funding source.