Background: Colorectal Cancers (CRC) are one of the top ten cancers to increase the burden on the health care system in India. Often patients experience symptoms after carcinoma has significantly progressed. Timely and accurate screening in the target population remains a challenge for clinicians.
Methods: The present study was conducted in one of the largest tertiary care hospitals in Asia. We studied the medical records of a total of 162 patients who had undergone surgical resection of the colon. 81 cases of CRC confirmed with histopathology reports were included in the Malignant group. The Control group had patients with benign and inflammatory lesions of the bowel. Hemograms of these patients at admission before the surgical procedure were taken in the study.
Results: We found a negative correlation (r-value of -0.46; p-value < 0.001) between Hemoglobin and Platelet count in the malignant group. The sensitivity of mean platelet volume to platelet count ratio (MPV/Pc) was 72%, followed by Platelet to hemoglobin ratio (PHR) of 70% and Platelet to Lymphocyte ratio (PLR) of 69%. AUC was highest for MPV/Pc (AUC = 0.731) among the ratios. Platelet count, PLR, and PHR were significantly (p-value < 0.001) high in the CRC cases. MPV/Pc was low compared to benign cases (p-value < 0.001).
Conclusion: We are advocating for the ratios to be assessed further for the possible additional screening tool for CRC.
Complete blood count, Colorectal carcinoma, Platelet to hemoglobin Ratio, Platelet to lymphocyte ratio, Mean platelet volume to platelet count ratio
CRC: Colorectal Cancer; CBC: Complete Blood Count; MPV: Mean Platelet Volume; NLR: Neutrophil to Lymphocyte Ratio; PLR: Platelet to Lymphocyte Ratio; PHR: Platelet to Hemoglobin Ratio; MPV/Pc: Mean Platelet Volume to Platelet count ratio
Carcinoma of the large bowel is common in northwest Europe, North America [1]. In the United States, it is the third most common cancer among men and women and the second leading cause of death from cancer. It is the most curable form of carcinoma of the gastrointestinal tract. Males and females are affected equally, and the mean age at diagnosis is in the sixth to seventh decades of life [2]. In India, the Annual Incidence Rates (AARs) for colon cancer and rectal cancer in men are 4.4 and 4.1 per 100000, respectively. The AAR for colon cancer in women is 3.9 per 100000. Colon cancer ranks 8th and rectal cancer ranks 9th among men. For women, rectal cancer does not figure in the top 10 cancers, whereas colon cancer ranks 9th [3].
A study identified a set of 24 genes mutated in a significant number of cases. In addition to genes found through prior research (e.g., APC, ARIDIA, TP53, KRAS, and PIK3CA), the researchers identified new genes such as SOX9, FAM123B/WTX, ERBB2, and IGF2. These genes were involved in regulating cell proliferation and can therefore serve as potential therapeutic drug targets [4]. Genetic factors contributing to CRC are colonic polyposis-associated syndromes i.e. Familial Adenomatous Polyposis (FAP) and its variants (Turcot, Gardner, and attenuated FAP), MYH-associated polyposis and non-colonic polyposis syndromes Hereditary Nonpolyposis Colon Cancer (HNPCC), or Lynch syndrome [5]. Environmental factors such as age, cigarette smoking, ulcerative colitis, diabetes mellitus, and obesity are also important contributors to the development of CRC.
According to ICMR guidelines, the workup of suspected CRC patients consists of digital rectal examination which has a high positive predictive value for the presence of rectal tumors. Other screening tests to detect early-stage colorectal cancer include Complete Blood Counts (CBC), stool-based tests (gFOBT, FIT, and FIT-DNA), liver and kidney function tests, Carcinoembryonic Antigen (CEA) tests, carbohydrate antigen 19.9 (CA19.9), direct visualization tests (flexible sigmoidoscopy, alone or combined with the FIT; colonoscopy; and CT colonography) and serology tests (SEPT9 DNA test).
The colon is one of the most common sites of the gastrointestinal tract biopsied during endoscopic evaluation. The majority of colonic biopsies are performed to exclude either inflammatory or preneoplastic or neoplastic processes. The most common forms of colitis encountered include inflammatory bowel disease (both ulcerative colitis and Crohn's disease) and infectious colitis, ischemic colitis, and lymphocytic/collagenous colitis. In addition, a variety of polyps can arise in the colon or rectum, most commonly hyperplastic polyps, adenomatous polyps, and sessile serrated polyps/adenomas [2].
There are many screening tests that can help diagnose early staged CRC. Centers for Disease Control and Prevention (CDC) recommends colonoscopy every 5 years or every 10 years with a Fecal Immunochemical Test (FIT) every year in individuals between the ages 45-75 years. In this study, we aim to explore correlations between different parameters of CBC in patients of CRC in order to investigate the role of novel CBC ratios as additional tools for CRC.
This is a retrospective, analytical study conducted in B.J. Medical College and Civil hospital - a tertiary care hospital in Gujarat, India. Medical reports of 81 patients with Colorectal cancer and 81 patients with intestinal benign conditions (benign intestinal tumors, polyps, inflammatory conditions, etc.) from September 2017 to March 2020 were taken from medical records for this study.
Based on the medical records of histopathology reports, a total of 162 patients were selected. Data were divided into two groups based on the benign and malignant etiology of the histopathology reports. Cases diagnosed with inflammatory etiology such as ulcerative colitis (without the histologic evidence of dysplasia); infectious etiologies including gastrointestinal tuberculosis, and amebiasis; non-neoplastic polyps such as hyperplastic polyps, inflammatory polyps and hamartomatous polyps were included in the benign group. The malignant group had cases of adenocarcinoma of the colon.
A routine hemogram was performed at the time of hospital admission. Hemograms were performed on automated analyzers (Horiba Pentra XLR, Automatic Hematology Analyzer, Horiba Yumizen H500, Automatic Hematology Analyzer, Horiba Yumizen H2500, Automatic Hematology Analyzer). Presenting complaints on admission were derived from the medical records which included combinations of abdominal pain, weight loss, blood in stool, constipation, unable to pass stool, diarrhea and vomiting.
World Health Organization classification of colorectal carcinoma and AJCC (American Joint Committee on Cancer) classification was followed for histopathology reporting.
Any patient older than 10 years of age with bowel resection with a definitive diagnosis of colorectal cancer on histopathology report. Data of patients whose CBC were performed on the hospital admission before the treatment, surgery or blood transfusion were taken in the study.
Inconclusive histopathology reports, and cases without CBC report at the admission prior to any intervention were excluded from the study.
Statistical analysis was done by two sample (independent) t-test. Correlations were analyzed by Spearman's correlation test. Analysis was done using MS Excel and SPSS software. The Receiver Operating Characteristic (ROC) curve was used to calculate the Area Under the Curve (AUC). Statistical significance was defined as p < 0.05. P-values mentioned in the study is 1-tailed.
In malignant cases, the average age was 50.52 ± 14.40 years with a male to female ratio of 1.13 whereas, in the benign group the average age was 44.68 ± 16.49 years of age with a male to female ratio of 2.68 Figure 1.
 Figure 1: Age and sex distribution of benign and malignant cases.
View Figure 1
Figure 1: Age and sex distribution of benign and malignant cases.
View Figure 1
Conventional adenocarcinoma was the most common (79.01%) histopathological subtype in the malignant group. The rest of the 21% included other variants such as mucinous adenocarcinoma, and signet adenocarcinoma. Out of all 81 cases, 65 cases were moderately differentiated adenocarcinoma. The most common primary tumor stage in the study was T3 Figure 2, Figure 3.
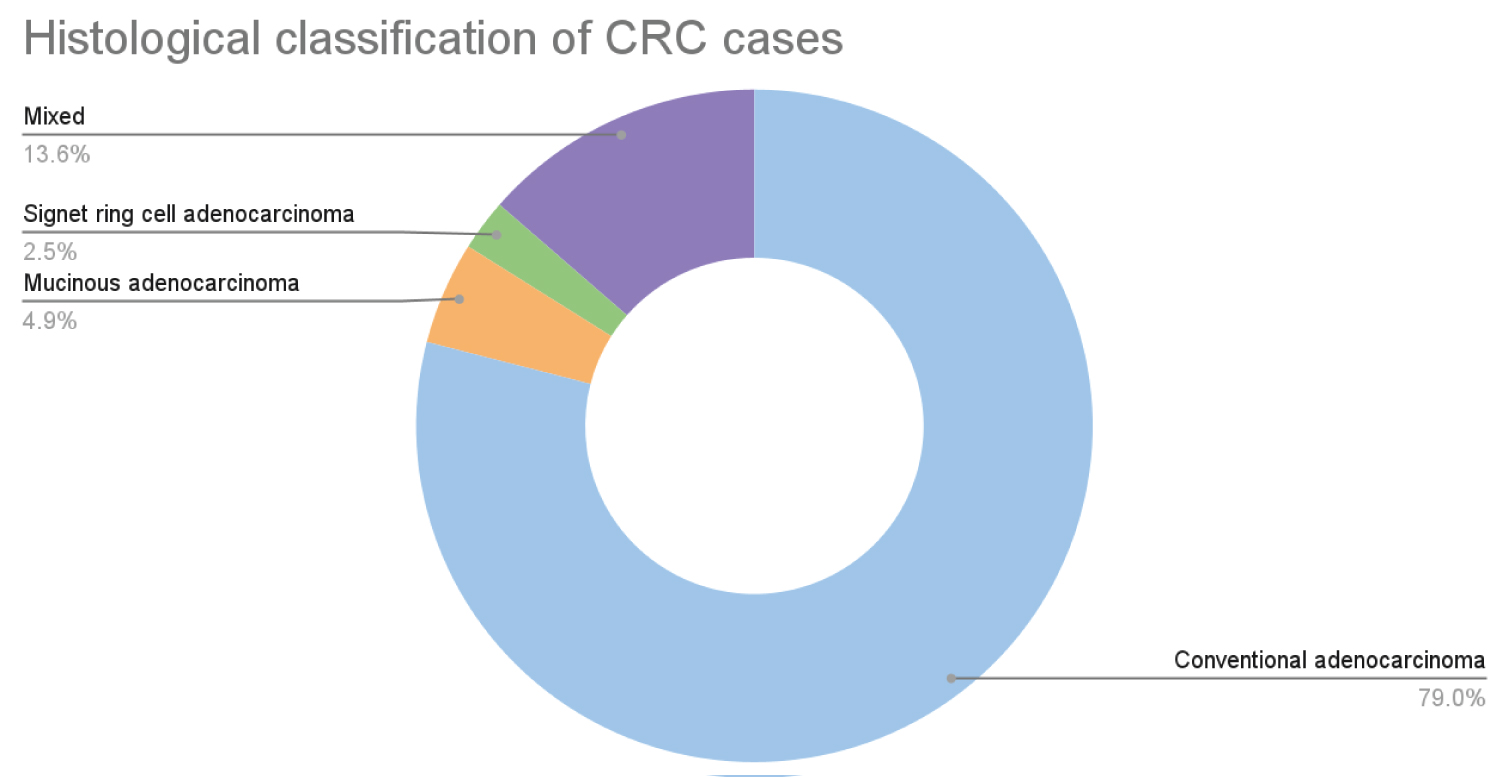 Figure 2: Histological classification of CRC.
View Figure 2
Figure 2: Histological classification of CRC.
View Figure 2
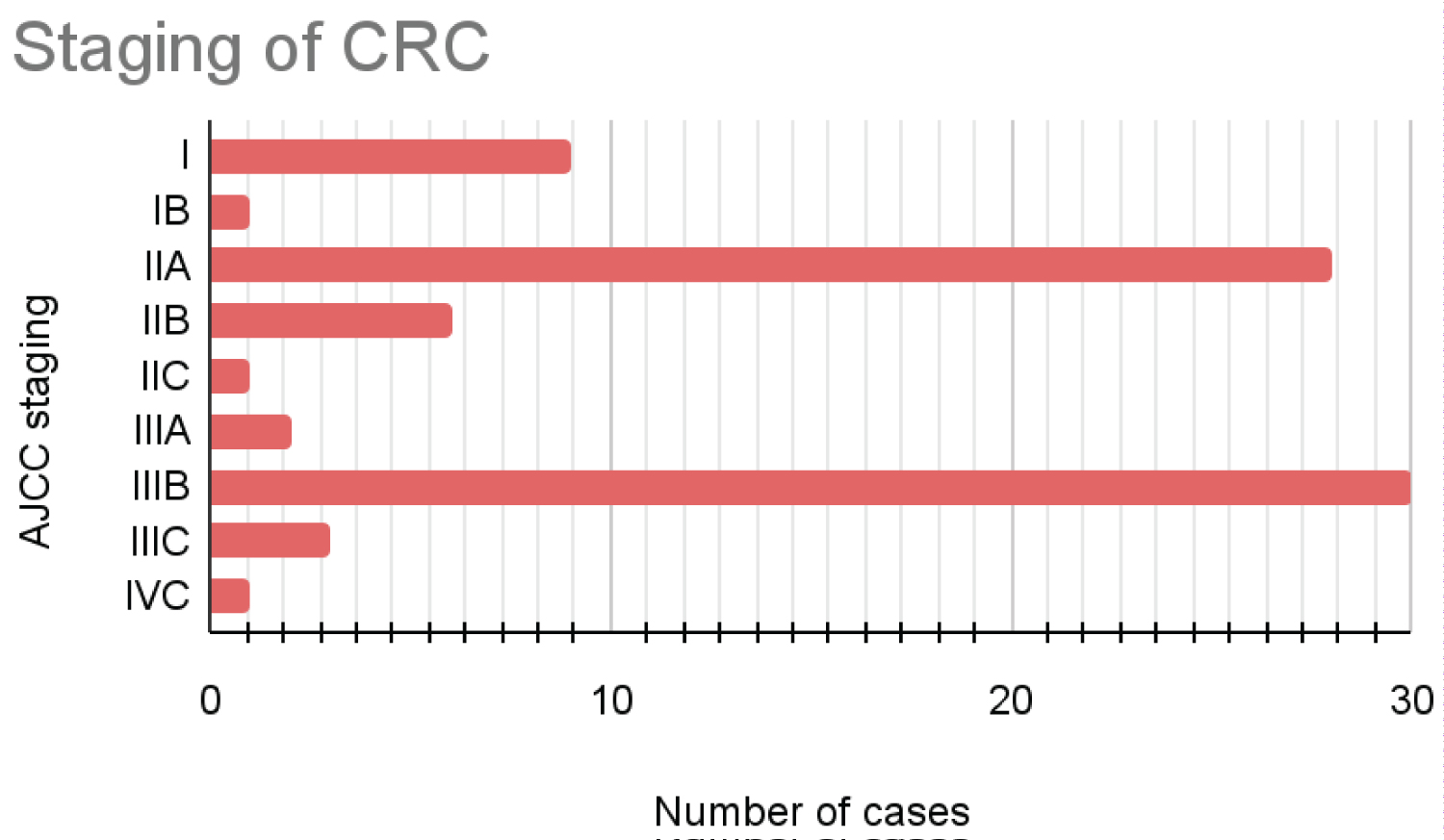 Figure 3: AJCC prognostic stage-wise distribution of CRC cases.
View Figure 3
Figure 3: AJCC prognostic stage-wise distribution of CRC cases.
View Figure 3
In the Benign group, the maximum cases (79.01%) were of inflammatory changes in the intestinal wall Figure 4.
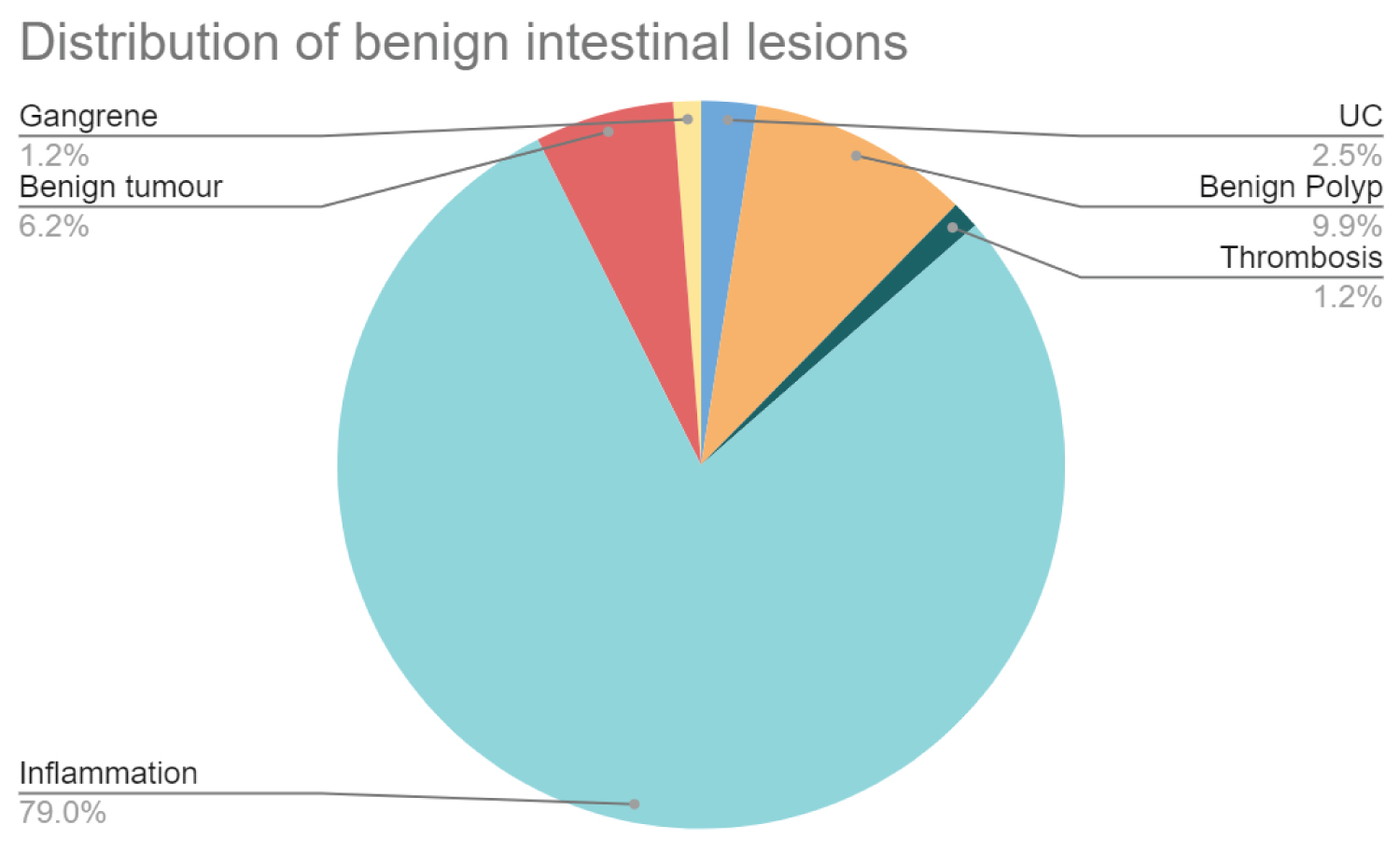 Figure 4: Benign lesions.
View Figure 4
Figure 4: Benign lesions.
View Figure 4
Based on hemograms of cases, Neutrophil-Lymphocyte ratio (NLR), Platelet-Lymphocyte ratio (PLR), Platelet-Hemoglobin Ratio (PHR) and Mean platelet volume-Platelet count ratio (MPV/Pc) were derived and compared.
The following tables show the mean ± SD value of said parameters. Two groups have been compared with the help of two sample (independent) t-test and t-values and one tailed p-values have been mentioned Table 1.
Table 1: Laboratory parameters and new ratios of malignant and benign cases. View Table 1
No significant difference was seen in PLR, PHR, NLR and MPV/Pc based on gender in CRC patients Table 2.
Table 2: Comparison of parameters in the malignant group based on gender. View Table 2
By two sample (independent) t-test, only MPV showed a statistically significant increase with advanced stage and presence of lymph node metastasis Table 3, Figure 5.
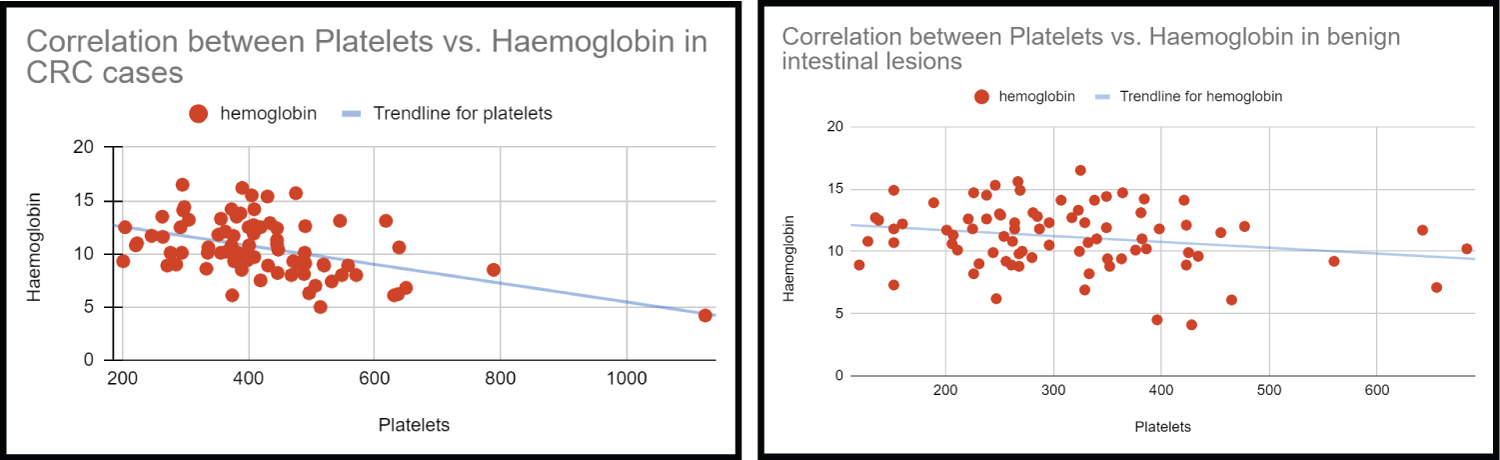 Figure 5: Correlation between Platelets and Hemoglobin.
View Figure 5
Figure 5: Correlation between Platelets and Hemoglobin.
View Figure 5
Table 3: Ratios based on AJCC staging of malignant cases. View Table 3
The negative correlation between Platelet (Plt) and Hb in patients with CRC had an r-value of -0.46 (p-value - < 0.001) and with the benign condition, it was -0.21 (p-value - 0.059). Linear regression equation for correlation between Plt and Hb in CRC cases is derived to be – Hb = -0.00885*Plt + 14.32.
The sensitivity of all three ratios combined was 48 and specificity was 37. Cut-off values for PLR, PHR, MPV/Pc were considered to be 175, 3 and 0.024 respectively. The sensitivity of all three ratios combined was 48 and specificity was 37. ROC shows AUC of 0.679, 0.723, 0.731 for PLR, PHR an MPV/Pc respectively Table 4, Figure 6.
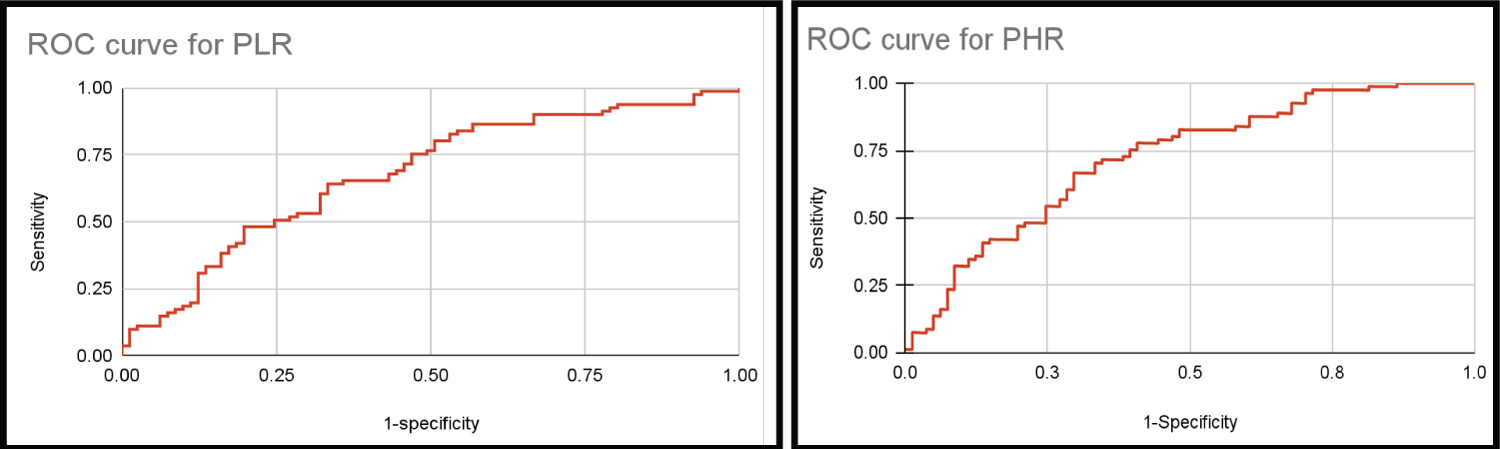 Figure 6: ROC curves for the proposed ratios (PLR, PHR, MPV/Pc).
View Figure 6
Figure 6: ROC curves for the proposed ratios (PLR, PHR, MPV/Pc).
View Figure 6
Table 4: Sensitivity and Specificity of the proposed ratios. View Table 4
Previously, many studies have pointed out some correlation between platelets and various carcinomas. Multiple papers have studied the relationship between mean platelets volume, platelets and hemoglobin in colon cancer, hepatic carcinoma and bladder cancer to name a few.
A significant amount of research has been done to help prove MPV as an early indicator of activated platelets. One possible explanation could be based on the fact that the platelets with elevated MPV contain more α-granules and release more prothrombotic substances, and these substances aggravate inflammation.
The vital role of activated platelets in cancer metastasis through the release of cytokines, chemokines and expression of several adhesion receptors makes a compelling argument to further explore platelets parameters in a routine hemogram.
Li JY, et al. argued about the role of Pro-inflammatory cytokines such as interleukin-6 (IL-6) in colon cancer patients. Furthermore, various tumor cell-secreted mediators, such as cysteine proteinases or ADP, contribute to platelet activation. Tumor cells could also activate platelets through the production of thrombin triggered by tumor-associated tissue factor.
We observed that high MPV is associated with higher prognostic tumor stage and lymph node involvement (< 0.05) which is backed by a Li N, et al study that also linked MPV with tumor differentiation (p < 0.001). Similarly, they did not report significant differences between the groups with regard to age, gender, NLR, PLR [6]. Li JY, et al. observed lower MPV values associated with the presence of colon cancer. We had a lower MPV value in the malignant group than in the benign group but it was not found to be statistically significant (p-value 0.83) [7].
A study by Wu-YY, et al. [8], reported significantly lower MPV values in the colorectal cancer group in comparison to controls. Similarly, we observed lower MPV values in the CRC group. However, this difference was not statistically significant (p-value 0.83). When MPV was compared among subgroups of malignant cases, MPV was significantly (p-value < 0.05) high with increased tumor staging. The mean platelet volume to platelet count ratio in the present study (p-value < 0.001) showed comparable results with Wu-YY, et al. who also reported a significantly lower MPV/Pc ratio in the colorectal cancer group in comparison to control groups. This was also in alignment with levels of MPV/Pc ratio in esophageal squamous cell carcinoma [9] and non-small cell lung cancer [10] in other studies Table 5, Table 6.
Table 5: Comparison of CBC parameters with other studies. View Table 5
Table 6: Comparison of MPV, MPV/Pc according to AJCC prognostic staging with another study. View Table 6
Milica, et al. reported that NLR and PLR were significantly higher in all individual TNM stages (I, II, III, and IV) compared to controls (p < 0.01), and MPV values were significantly lower (p < 0.01) [11]. PLR was also significantly high in the CRC group in the present study. Aside from that, the present study reported lower NLR than the benign control group. This could be explained by infections, and inflammatory etiology in the benign group. They also reported a significant difference between early stages (I, II) and more advanced disease stages (III and IV) in values of NLR and PLR (< 0.05) [11]. Li MX, et al. concluded Increase in NLR is also significantly associated with the poorer differentiation of the tumor (OR: 1.574, 95% CI: 1.226-2.022) [12]. In contrast to these findings, present study showed higher MPV values with advanced staging. NLR, PLR, MPV/Pc did not show any statistically significant increase with the stage of the tumor Table 7.
Table 7: Comparison of sensitivity and specificity of proposed parameters with another study. View Table 7
We argued that Platelets play a role in tumorigenesis and progression in CRC. Platelets are increased in number and secret vascular endothelial growth factor and platelet-derived growth factor. Conversely, lymphocytes are involved in tumor cell apoptosis in the antitumor reaction of the immune system. Hence, the ratio of elevated platelets and decreased lymphocytes can give us information about underlying malignancy. Meta-analysis by Tan D, et al. indicated that increased PLR is significantly associated with poor tumor differentiation [odds ratio (OR) 2.12; 95% CI, 1.45-3.08, P < 0.001)]. This meta-analysis suggested that a high peripheral blood PLR can be used as a predictor of overall survival [13]. In the present study, PLR was significantly high in the malignant group (p-value < 0.001) yet it failed to detect prognostic staging of the tumor. (p-value 0.47).
Platelet-to-Hemoglobin Ratio (PHR) was studied by Li Xing, et al. [14] and reported a worse prognosis with higher PHR in patients with hepatocellular carcinoma receiving transcatheter arterial chemoembolization. In the present study, we noticed a statistically significant difference (p-value - < 0.001) between the PHR of the two groups. Mean hemoglobin in CRC patients is low probably due to bleeding per rectum. Increased platelets can be explained by the presence of ongoing inflammation. A significant p-value of the aforementioned novel ratio is attributable to these findings in CRC patients. CRC cases had a higher PHR ratio compared to the control group. Despite that, PHR fell short in differentiating tumor stage in CRC cases (p-value 0.16).
Based on the results assessed, it is clear that to suspect CRC at the time of screening, various variables from the hemogram need to be taken into consideration in conjunction with other known screening methods.
Having studied these new parameters, we realize the limitations of our study. Ours was a retrospective study only from one tertiary care center. Further studies are required to establish this relationship with more certainty. The present study does not take into account the possible medical treatment taken by CRC patients before the surgery.
Novel markers such as NLR (p-value < 0.05); PLR, PHR, and MPV/Pc (p-value < 0.001) all had statistically significant differences between malignant and benign groups. CRC was associated with higher PLR and PHR whereas lower MPV/Pc ratio. Lower MPV/Pc is 72% sensitive to raise suspicion of CRC followed by High PLR and PHR being sensitive at about 70%. Among the subgroups of CRC according to TNM staging, elevated MPV points at an advanced stage of colorectal cancer (p-value < 0.05). Based on the results of this study, it is apparent that a routine hemogram has an ample amount of unexplored data. Since CBC is a cheap, easy to perform, easily accessible, low-risk procedure, the addition of these calculated ratios can aid in screening for colorectal cancer.
Medical records and laboratory information system, Civil hospital, Ahmedabad.
None.