Background: Rectal cancer constitutes nearly one-third of all colorectal cancer diagnoses and certain clinical and molecular markers have been studied as potential prognosticators of patient survival. The main objective of our study was to investigate the relationship between the expression intensities of certain proteins: Growth Hormone-Releasing Hormone Receptor (GHRH-R), Hsp90, Hsp16.2, p-Akt and SOUL in specimens of locally advanced rectal cancer patients and time to metastasis as well as 10-year Overall Survival (OS). We also investigated whether these outcome measures were associated with the presence of other clinical parameters.
Methods: 109 patients were investigated retrospectively. Samples of pretreatment tumors were stained for proteins GHRH-R, Hsp90, Hsp16.2, p-Akt and SOUL using immunhistochemistry methods. Kaplan-Meier curves were used to show the relationship between intensity of expression of biomarkers, clinical parameters, time-to metastasis and 10-year OS.
Results: High levels of p-Akt, GHRH-R and Hsp90 were associated with significantly decreased 10-year OS (p = 0.001; p = 0.000; p = 0.004, respectively) and high expression levels of p-Akt and GHRH-R were correlated with significantly shorter time to metastasis. Tumors localized in the lower third of the rectum were linked to both significantly longer time to metastasis and improved 10-year OS.
Conclusion: Hsp 90, pAkt and GHRH-R as well as the lower-third localization of the tumor were predictive of 10-year OS in locally advanced rectal cancer patients. GHRH-R and Hsp90 expression were independent prognosticators of OS. Our results imply that GHRH-R could play a particularly important role both as a molecular biomarker and as a target for anticancer treatment of advanced rectal cancer.
Neoadjuvant radiochemotherapy, Rectal cancer, Hsp90, GHRH-R, Predictive markers, Overall survival
GHRH-R: Growth Hormone-Releasing Hormone Receptor; Hsp: Heat shock protein; LMR: Lymphocyte-to-Monocyte Ratio; NRCT: Neoadjuvant Radiochemotherapy; OS: Overall Survival; SOUL: Heme-Binding Protein 2; TRG: Tumor Regression Grade
Colorectal cancer is the third most common cancer in the world, and almost a third of all its diagnoses constitute rectal cancer [1]. The treatment protocol for rectal cancer depends primarily on the clinical stage of the cancer with neoadjuvant radiochemotherapy (NRCT) followed by total mesorectal excision being the standard of care for patients with locally advanced rectal cancer [2]. Patient survival and 10-year cumulative incidence of distant metastasis have been shown to correlate with the response to NCRT [3], which however, varies characteristically among individual patients. The histopathological Tumor Regression Grade (TRG) has been reported to be an independent predictor of disease-free survival with which pathological response to NRCT can be assessed [4-6].
Besides TRG, other clinical parameters, such as poor condition at the operation and certain molecular markers, like the expression of Matrilysin-2 have been implicated to be predictive factors for Overall Survival (OS) [7,8]. For example, the Lymphocyte to Monocyte Ratio (LMR), has been shown to correlate with the survival in different cancer types, including colorectal and rectal cancer [9,10].
In an earlier study, we analyzed the expression levels of the Growth Hormone–Releasing Hormone Receptor (GHRH-R), Heat shock protein (Hsp) 90, p-Akt, Hsp16.2 and Heme-binding protein 2 (SOUL) in pretreatment rectal tumor samples, and found that GHRH-R and Hsp90 were independent predictive factors of histopathological response to NRCT [11].
In addition to its neuroendocrine function, Growth Hormone–Releasing Hormone (GHRH) acts as an autocrine/paracrine growth factor in various cancers including gastrointestinal cancers [12,13]. The presence of GHRH-R and its splice variants - supporting the role of GHRH- has been demonstrated in different cancers, such as oesophageal and colorectal neoplasms [14,15].
Heat shock proteins and their members with lower molecular weight: Small Hsp-s, are chaperone molecules, expressed in response to different types of damage affecting the cells [16]. Increasing evidence has demonstrated that Hsps, including small Hsp-s play important roles in different types of tumors [17-20]. Hsp 90, the most abundant Hsp, chaperones 400 different client proteins, many of whom play crucial roles in malignancies [21]. In neuroectodermal tumors, the level of the expression of a previously characterized small Hsp, Hsp 16.2 was found to correlate with the histological grade of different types of brain tumors [22-24].
Members of cytoprotective pathways and those involved in tumor necrosis have also been investigated as possible molecular targets in cancer research [25,26]. The PI3K/Akt pathway is a major anti-apoptotic pathway and associated with the proliferation of malignant cells [27], while tumor necrosis has been shown to be an independent prognostic variable of cancer-specific survival [28]. A member of the BH3-domain-only protein family, Heme-binding protein 2 (SOUL), was shown to facilitate necrotic cell death in oxidative stress through the permeabilization of the mitochondrial membranes [29,30].
In our present investigation- analyzing a larger patient sample, than in our previous study [11] - we aimed to investigate the relationship between the intensities of protein expressions (GHRH-R, Hsp90, Hsp16.2, p-Akt and SOUL) in pretreatment tumor samples, 10-year Overall Survival (OS) and the time-to metastasis. It was also our objective to investigate whether patients' time-to metastasis and 10-year Overall Survival (OS) were associated with certain clinical parameters (gender, time-to-surgery, tumor localisation) of the patients.
Altogether 114 patients with locally advanced (cT3/T4 and/or cN + and cM0) rectal adenocarcinoma participated in our study (69 consecutive patients between January 2005 and December 2006 and 45 consecutive patients between January 2009 and March 2010). All patients received neoadjuvant radiochemotherapy (NRCT) followed by surgery. Pre-treatment examinations included digital rectal examination, sigmoidoscopy, biopsy, abdominal-pelvic CT, pelvic MRI, chest X-ray or CT.
3D planned conformal radiotherapy was carried out in all cases with the belly board in prone position, with 18 MV photons. The primary tumor as well as the lymph nodes at risk were covered with 3 irradiation fields and given 45 Gy-s in 25 fractions over a period of 5 weeks. Concomitant chemotherapy, 500 mg/m2 of 5-Fluorouracil in continuous infusion and 30 mg/m2 folic acid bolus on days 1-5 of the 1st and the 5th weeks of radiotherapy, were administered. Four weeks after the completion of NRCT, patients were re-staged and definitive surgical resection was performed 6-9 weeks after neoadjuvant therapy in altogether 109 cases (5 patients were excluded from the study). Curative resection was performed in all cases. All patients signed informed consent, which was approved by the local ethics committee. The main clinical characteristics of the patients who underwent operation are given in Table 1.
Table 1: Patient and tumor characteristics. View Table 1
The rectal radiotherapy grading system adapted from Mandard, et al. [31] was used for the histological evaluation of the resected specimens to determine the pathological response to neoadjuvant treatment. The five-point Tumor Regression Grading (TRG) is based on the presence of residual tumor cells and the extent of fibrosis. The TRG includes the following: TRG 1 (complete regression) is defined as the absence of residual tumor and fibrosis extending through the different layers of the rectal wall, TRG2 is characterized by the presence of rare residual tumor cells scattered throughout the fibrosis, TRG3 shows an increase in the number of residual tumor cells, but the fibrosis still predominates, TRG4 demonstrates residual tumor outgrowing the fibrosis, and TRG5 is characterized by the absence of any tumor regression. In line with earlier studies to facilitate statistical analysis, the TRG was combined into two groups: good responders consisting of TRG1-2 and poor responders comprising TRG 3-5 [31-33].
Rabbits were immunized subcutaneously at multiple sites with 100 pg of recombinant Hsp16.2/GST and SOUL/GST fusion proteins, which were expressed as described previously [11,22,29] in Freund's complete adjuvant. Four subsequent booster injections at 4-week intervals were given with 50 pg of protein in Freund's incomplete adjuvant. Blood was collected 10 days after boosting, and the antiserums were stored at 20 °C. IgGs were affinity-purified from sera by protein G-Sepharose chromatography (Sigma), according to the manufacturer's protocol.
Sections of the pre-treatment tumor tissue samples were fixed in formalin and embedded in paraffin. Subsequently, they were incubated with the following primary antibodies: GHRH-R primary antibody purchased from Abcam (Abcam Inc., Cambridge, MA), p-AKT and Hsp90 primary antibodies purchased from Cell Signaling (GHRH-R antibody detected the presence of both GHRH-R as well as the splice variants of the GHRH-R) and the self-developed anti-Hsp 16.2 and anti-SOUL polyclonal primary antibodies. Immunohistochemical staining was carried out according to the strepatavidin–biotin-peroxidase method with hydrogen peroxide/3-amino-9-ethylcarbazole development, using the Universal kit as previously described [34]. Only secondary IgG was incubated with the control sections. The evaluation of the slides was performed with the help of an Olympus BX50 light microscope with incorporated photography system (Olympus Optical Co., Hamburg, Germany). Staining intensity was recorded semiquantitatively as mild (+), moderate (++) or strong (+++), as described before [35]. For internal positive control, the normal cellular and vascular structures of the samples were used. Positive areas around necrotic fields were excluded due to their probable stress-related upregulation. All slides were assessed by the same experienced pathologist blinded to clinico-pathological data.
To increase the number of patients per group, the categories of the various variables were combined for these analyses: age over 60 years vs. 60 years or below, cT2 vs. cT3 vs. cT4, cN0 vs. cN1- 2, distance from the anal verge less than 5 cm versus between 5 and 10 cm versus more than 10 cm, time to surgery within 7 weeks versus over 7 weeks. For statistical testing, intensity values of immunohistochemistry were dichotomized into low-intensity (0, +) and high intensity (++, +++) categories. Overall survival (OS) was defined from the date of diagnosis until the time of death from any cause. Time to metastasis was defined from the date of diagnosis until the documented first appearance of metastasis.
Data relating to patient parameters and survival were extracted from the clinic's electronic medical database (eMedsol database) and the National E-health Infrastructure Database (EESZT). In cases where data regarding survival was not available from the above-mentioned databases, one researcher in the group contacted the patient or their family members for information.
Statistical analyses were performed with use of Statistical Package for the Social Sciences software 16.0 (SPSS, Chicago, IL). Univariate Chi-square test was used to compare clinical parameters and biological markers for tumor regression grade and clinical response. The relationship between the clinical- and biological markers and Overall Survival (OS) and time-to-metastasis were demonstrated using Kaplan-Meier curves and the level of significance was determined using the log-rank test. Following the proportional hazard assumption testing of clinical and biological parameters found to significantly affect OS, multivariate Cox regression analysis was performed (with a Confidence interval of 95%) to test for the independent influence of potential prognostic factors on overall survival. In the course of model estimation, the Enter method was used, meaning that all variables -excepting “tumor localization”- were used as possible covariants and included in the model and their combined effect was analyzed. Probability (p) values < 0.05 were considered statistically significant, and statistical tests were based on a two-sided significance level.
Immunohistochemical evaluation of the pre-treatment biopsy samples showed high-intensity staining (++, +++) for SOUL, Hsp 16.2, Hsp90, GHRH-R and for p-Akt in 60, 54, 67, 67% and 76% of the cases, respectively (Table 2). Good pathological response was found in 52, and poor response to NRCT was found in 57 cases out of the studied 109 specimens.
Table 2: Immunhistochemical expression of proteins in pre-treatment tumor specimens. View Table 2
High levels of GHRH-R expression in the pretreatment tumor specimens were significantly correlated with poor histopathological response (P = 0.049), while no significant relationship could be detected between the expression of other proteins and tumor regression grade (Table 3).
Table 3: Relationship between protein expression and histopathological response to NRCT (n = 109). View Table 3
Patients whose time to surgery was more than 7 weeks had a significantly higher chance of having a good response to NRCT (p = 0.003), than those who underwent surgery within 7 weeks after NRCT. None of the other pre-treatment clinical parameters was found to be significantly related statistically to histopathological response (Table 4).
Table 4: Relationship between clinical parameters and histopathological response to NRCT (n = 109). View Table 4
We examined the relationship between pre-treatment protein expression and 10-year OS. By 120 months, patients with tumors containing high levels of GHRH-R had a survival rate below 30%, compared to patients with cancers expressing low GHRH-R, where almost 90% of the patients were still alive. High levels of p-Akt, GHRH-R and Hsp90 were associated with significantly decreased 10-year OS (p = 0.001; p = 0.000; p = 0.004, respectively) (Figure 1a, Figure 1b and Figure 1c). The intensity of SOUL and Hsp16.2 staining did not affect the 10-year OS significantly (Figure 1d and Figure 1e).
 Figure 1: The relationship between the staining intensity of pre-treatment proteins a) p-Akt (p = 0.001), b) GHRH-R (p = 0.000), c) Hsp90 (p = 0.004) d) SOUL (p = 0.661), e) Hsp16.2 (p = 0.975) and 10-year OS. The effect of biological markers on overall survival was demonstrated using Kaplan-Meier curves and the level of significance was determined using the log-rank test. Probability (p) values < 0.05 were considered statistically significant.
View Figure 1
Figure 1: The relationship between the staining intensity of pre-treatment proteins a) p-Akt (p = 0.001), b) GHRH-R (p = 0.000), c) Hsp90 (p = 0.004) d) SOUL (p = 0.661), e) Hsp16.2 (p = 0.975) and 10-year OS. The effect of biological markers on overall survival was demonstrated using Kaplan-Meier curves and the level of significance was determined using the log-rank test. Probability (p) values < 0.05 were considered statistically significant.
View Figure 1
We evaluated the effect of the individual clinical parameters on 10-year OS. Good histopathological responses and the lower-third tumor localization were associated with significantly improved 10-year OS, compared to patients with poor histopathological response or upper- or middle-third tumors (p = 0.029; p = 0.015, respectively) (Figure 2a and Figure 2b). Although the association was not significant, female gender was linked with an improved 10-year OS (Figure 2c). Time to surgery did not significantly affect the 10-year OS (Figure 2d).
 Figure 2: The relationship between clinical parameters a) histopathological response (p = 0.029) b) tumor localization (p = 0.015) c) gender (p = 0.057) d) time to surgery (p = 0.568 and 10-year OS. The effect of clinical parameters on the overall survival was demonstrated using Kaplan-Meier curves and the level of significance was determined using the log-rank test. The probability (p) values < 0.05 were considered statistically significant.
View Figure 2
Figure 2: The relationship between clinical parameters a) histopathological response (p = 0.029) b) tumor localization (p = 0.015) c) gender (p = 0.057) d) time to surgery (p = 0.568 and 10-year OS. The effect of clinical parameters on the overall survival was demonstrated using Kaplan-Meier curves and the level of significance was determined using the log-rank test. The probability (p) values < 0.05 were considered statistically significant.
View Figure 2
The relationships between pre-treatment proteins, clinical parameters and OS using the log-rank test are shown in Supplementary Table 1.
The biological and clinical markers found to have a significant association with 10-year OS were analyzed using multivariate Cox regression analysis. Two biological markers, GHRH-R and Hsp90 were found to be significant independent prognostic factors of overall survival (in the case of GHRH-R: p: 0.000, EXP (B): -2.015, 95.0% CI: 0.044-0.401, and in the case of Hsp90 p: 0.005, EXP (B):-0.928, 95.0% CI: 0.206-0.757) (Table 5).
Table 5: Markers showing significant results after Cox regression analysis of the investigated biological and clinical parameters. View Table 5
A moderate correlation was found between p-Akt and GHRH-R, possibly explaining why p-Akt was not found to be a significant variable. However, when GHRH-R was excluded from the model, p-Akt remained significant (p = 0.000; Exp (B) = 0.204, 95.0% CI: 0.086-0.481) indicating that the prognostic role of p-Akt appears probable (Data not shown).
High expression levels of p-Akt and GHRH-R were associated with significantly shorter time-to-metastasis (Figure 3a and Figure 3b). No significant relationship could be detected between the expression levels of Hsp90, SOUL and Hsp16.2 and the time to metastasis (Figure 3c, Figure 3d and Figure 3e).
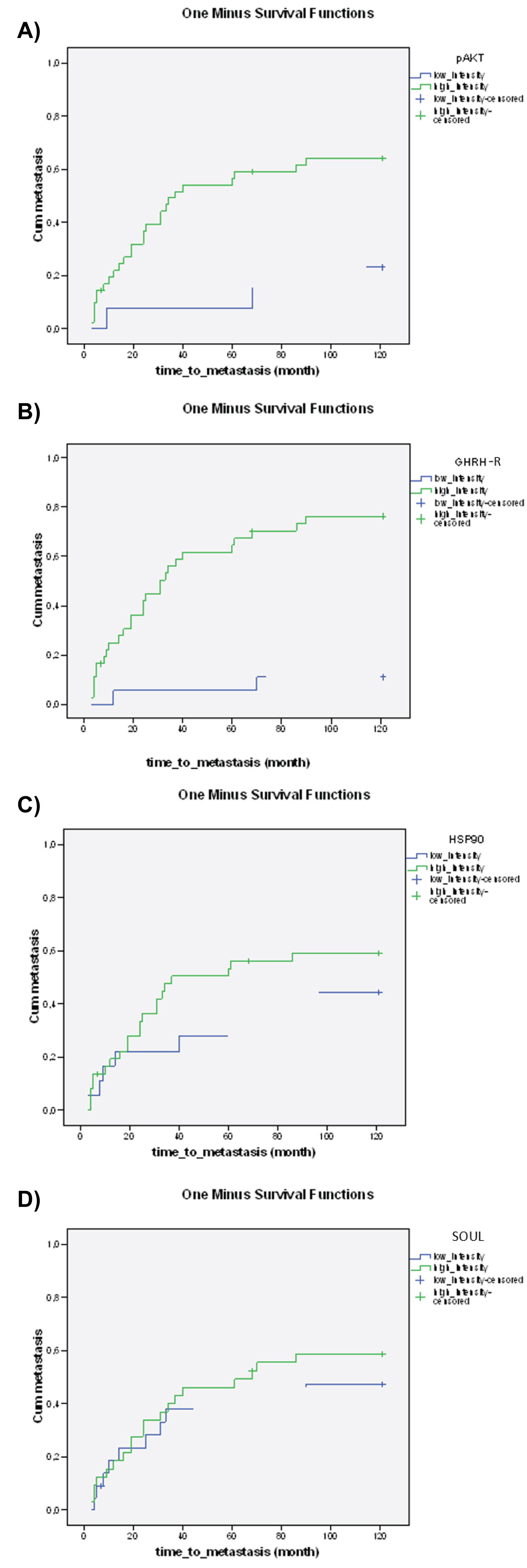 Figure 3: The relationship between the staining of pre-treatment proteins a) p-Akt (p = 0.000), b) GHRH-R (p = 0.000), c) Hsp90 (p = 0.115), d) SOUL (p = 0.310), e) Hsp16.2 (p = 0.328 and the time to metastasis. The effect of biological markers on the time to metastasis was demonstrated using Kaplan-Meier curves and the level of significance was determined using the log-rank test. Probability (p) values < 0.05 were considered statistically significant.
View Figure 3
Figure 3: The relationship between the staining of pre-treatment proteins a) p-Akt (p = 0.000), b) GHRH-R (p = 0.000), c) Hsp90 (p = 0.115), d) SOUL (p = 0.310), e) Hsp16.2 (p = 0.328 and the time to metastasis. The effect of biological markers on the time to metastasis was demonstrated using Kaplan-Meier curves and the level of significance was determined using the log-rank test. Probability (p) values < 0.05 were considered statistically significant.
View Figure 3
The tumors localized in the lower third of the rectum were associated with significantly longer time to metastasis. No significant relationship could be detected between the time to surgery, histopathological response, gender, and time to metastasis (Figure 4a, Figure 4b, Figure 4c and Figure 4d).
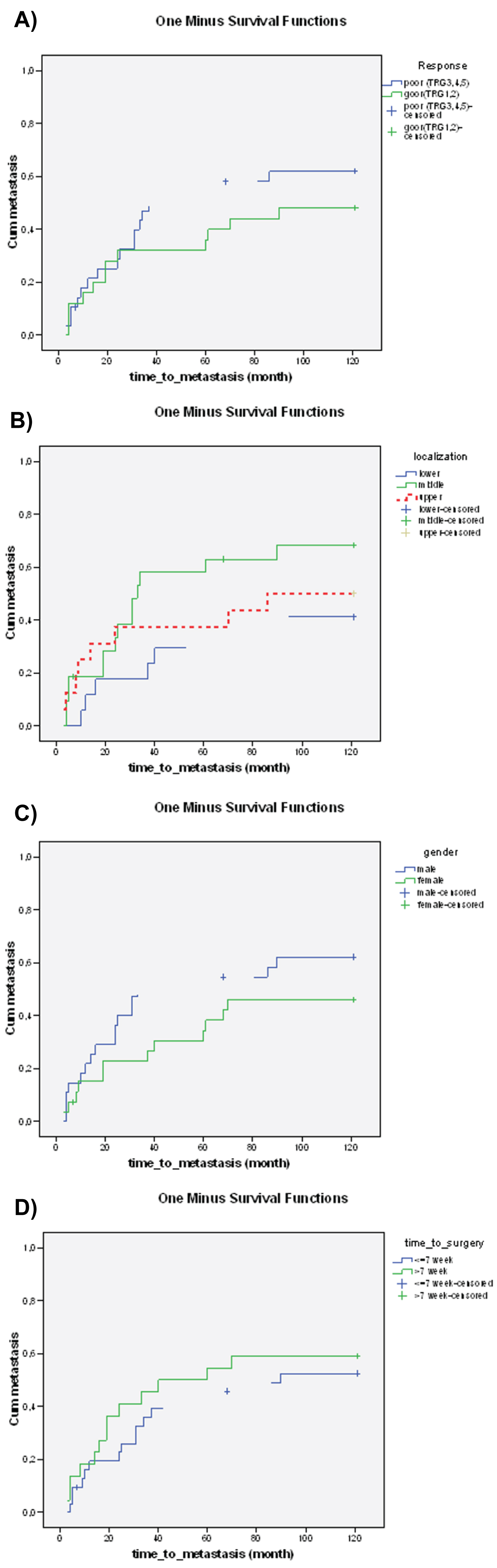 Figure 4: The relationship between clinical parameters a) Histopathological response (p = 0.170), b) Tumor localization (p = 0.048), c) Gender (p = 0.064), d) Time to surgery (p = 0.319), and time to metastasis. The effect of clinical parameters on time to metastasis was demonstrated using Kaplan-Meier curves and the level of significance was determined using the log-rank test. The probability (p) values < 0.05 were considered statistically significant.
View Figure 4
Figure 4: The relationship between clinical parameters a) Histopathological response (p = 0.170), b) Tumor localization (p = 0.048), c) Gender (p = 0.064), d) Time to surgery (p = 0.319), and time to metastasis. The effect of clinical parameters on time to metastasis was demonstrated using Kaplan-Meier curves and the level of significance was determined using the log-rank test. The probability (p) values < 0.05 were considered statistically significant.
View Figure 4
In our study, we identified new molecular markers and analyzed previously investigated clinical markers as predictors of time-to metastasis and OS in patients with locally advanced rectal cancer.
Our previous study showed that the expression levels of GHRH-R and Hsp90 were independent predictive factors of histopathological response to NRCT [11]. Besides the response to therapy, the appearance of the first metastasis and overall survival are both decisive outcomes for cancer patients. Supporting their possible role in locally advanced rectal cancer, we found that high levels of p-Akt, Hsp90 and GHRH-R were associated with significantly decreased 10-year OS, while increased staining intensities for p-Akt and GHRH-R were also linked to a significantly shorter time to metastasis. Malignant transformation and metastasis formation have been reported to be promoted by the growth hormone-stimulated Insulin-like growth factor I (IGF-I) in different malignancies [36-38] and antagonists of GHRH have been shown to suppress the tumoral production of IGF-I and IGF-II by inhibiting the secretion of GH and blocking the binding of autocrine GHRH to receptors on cancer cells [39-41]. An earlier study reported that increased GHRH-R in tumor samples of gastric cancer correlated with poor overall survival and was an independent predictor of patient prognosis [42]. The overexpression of GHRH-R in colorectal malignancies has been detected previously [43]. Furthermore, the antagonization of GHRH has been shown to induce DNA damage in human colon cancer cells and subsequently to lead to p21-mediated cell cycle arrest and apoptosis [44]. Our findings indicate that GHRH-R, consequently GHRH, may have an important part in both response to therapy and patient outcome measures. Although GHRH-antagonists have not yet been introduced into the clinical practice, the potential importance of GHRH-R both as a probable molecular target of therapy and -based on our present findings- as a predictor for survival in locally advanced rectal cancer, is highly plausible.
Cell proliferation, migration and angiogenesis are processes which contribute to metastasis formation [43]. Levels of pAkt have been shown to be overexpressed in CRC compared to normal colorectal mucosa [45], while angiogenesis of CRC was shown to be induced through the activation of the Akt/Erk signaling pathways [46]. Accordingly, in our tumor samples, intensive pAkt staining correlated with shorter time- to-metastasis and decreased 10-year OS. The client proteins of Hsp 90 have also been known to be associated with the development and progression of cancer cells [47,48]. Consequently, Hsp 90 may be overexpressed in tumors [49,50], as was shown in a study which found a positive correlation between overexpression of the mRNA for Hsp90 and metastasis and poor prognosis in CRC [51]. In line with these reports, our results indicated that high levels of Hsp90 levels in the pretreatment rectal cancer specimens were associated with decreased 10-year OS. Furthermore, along with GHRH-R, Hsp90 was found to be an independent prognosticator of OS in our study.
Although Hsp16.2 has been shown to inhibit cell death by binding to Hsp90 and through the activation of the PI-3kinase/Akt pathway [24] and was also been found to be overexpressed in esophageal cancer [52], the intensity of Hsp 16.2 staining showed no significant relationship with either the time to- metastasis or 10-year OS in our present study. Furthermore, both Hsp16.2 and SOUL were predictors of negative response to NRCT in esophageal cancer according to a previous report [52], yet neither protein correlated with the survival in our patients. It is plausible, thus, that in contrast to esophageal cancer cells, the main manner of tumor development in rectal cancer cells does not occur through the Hsp16.2-mediated anti-apoptotic or SOUL-mediated necrotic pathways. The exact molecular mechanisms require further investigation, however.
Among the studied clinical parameters, the time to surgery was significantly associated with the response to therapy but did not affect the time-to metastasis or 10-year OS in our study. Our findings are supported by other recent studies on rectal cancer patients in which no significant differences in disease-free or overall survival were found between patients with different time intervals to surgery [53-55], Furthermore, according to earlier reports, a longer than 6-8-week period between the completion of NRCT and surgery was associated with a significantly higher rate of response [56,57]. This may be explained by the phenomenon, that biological changes induced by radiation need time to develop.
In recent years several investigations have supported the prognostic role of TRG in locally advanced rectal cancer, where the degree of tumor regression following NRCT, indicating the histopathological response to NRCT, was found to correlate with the cumulative incidence of distant metastasis, disease-free and overall survival [3,6]. Corresponding to these reports, we also observed that good histopathological response leads to significantly improved 10-year OS.
There have been conflicting reports regarding the role of tumor localization in locally advanced rectal cancer patients. While an earlier study reported that the distance from the anal verge of more than 5 cm was associated with significantly lower downstaging rates [58], a more recent, but smaller study discovered that a distance more than 5 cm was related to a significantly higher complete pathological response rate and improved 5-year survival [59]. However, a very recent meta-analysis showed, that cancers closer to the anal verge were significantly more likely to achieve complete pathological response [60]. In accordance with this large analysis, we found that a distance less than 5 cm from the anal verge, that is lower-third localization, was related to a longer time to metastasis and increased 10-year OS. These findings may be explained by the clinical behavior of lower rectal cancers. The tumors in the lower third of the rectum can be reached more easily by physical examination and signs of bleeding may also be apparent sooner to the patient than tumors in the middle or upper third of the rectum, thus possibly leading to the earlier detection of the tumor.
Another clinical parameter that has been investigated as a potential prognostic factor, is the gender of the patient. Females have been found to have significantly better OS in CRC than males [61], which tendency we also observed among our patients, although the advantage was not found to be significant among our patients.
Although the parameters investigated as potential clinical prognosticators supported mostly the results from previous studies, our results highlight the possible importance of utilizing relatively easily accessible information, such as the localization of the tumor in everyday practice.
Our study has limitations, including the comparatively small sample size and the retrospective characteristics of our analysis.
We conducted the follow-up study of 109 patients with locally advanced rectal cancer over a 10-year period.
To our knowledge, this is the first study to report that Hsp90, pAkt and GHRH-R in pretreatment tumor samples are possible molecular predictive markers of decreased OS in locally advanced rectal cancer patients. Furthermore, GHRH-R and Hsp90 are independent prognostic factors of 10-year overall survival and pAkt may have an independent predictive role as well.
We also showed that the intensities of pAkt and GHRH-R expression were prognosticators of time-to-metastasis. These as well as our previous finding that the expression of GHRH-R was an independent prognostic factor for the response to therapy [11] all imply that GHRH-R could play a particularly important role both as a molecular biomarker and as a target for anticancer treatment of advanced rectal cancer.
This project was supported by the Janos Bolyai Research Scholarship of the Hungarian Academy of Sciences.
The authors declare that they have neither financial nor non-financial competing nor conflicting interests.
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
All authors were involved in delivery of the investigation. SB, ÁB, and EP designed the study. EG carried out immunohistochemistry. DF, ZL, ES, and DL collected the patient data and performed the statistical analysis. EP, LR and AVS analyzed the data and the relevant literature. SB and ÁB prepared the final figures and the tables. DF and EP prepared the manuscript. SB and AVS reviewed the manuscript. All authors have read and approved the final manuscript.
This study was performed in line with the principles of the Declaration of Helsinki. Ethics committee approval was obtained from the Regional Ethical Committee (Reference number: 7532-PTE2019).
Written informed consent was obtained from the patients for participation in this study.
Written informed consent was obtained from the patients or -in case of death- from the close relative for publication of this study.
None.