The eco-friendly nature of plant extracts has made them become sustainable alternative vector control agents. Anopheles mosquitoes serve as the vectors for human malaria. Malaria still kills more than 600,000 individuals every year, 90% of this in sub-Saharan Africa and mostly children under five. In the present study, larvicidal activity of leaf extract of Telfairia occidentalis was evaluated against the malarial vector, Anopheles coluzzi. The adult blood-fed Anopheles mosquitoes were collected and each gravid mosquito was forced to lay eggs separately in a prepared tube. Each mosquito that laid an egg was morphologically identified prior to the molecularly identification by SINE 200 PCR. 540 of the F1 generation colonies were used for this study. The Highest larval percent mortality (100%) was observed in 600 ppm concentration and above at 24 h of time of exposure. The lowest mortality of 8.3% was observed in the lowest concentration of 20 ppm. This indicates that the extract is concentration-dependent. The LC50 and LC90 against the larvae were 75.56 ppm and 266.31 ppm respectively. At P > 0.05 level, the Chi-square value was significant. This shows the potential of the extract in controlling mosquito vectors in the environment. Amplification of Short Interspaced Nuclear Element 200, i.e. SINE 200 region in Anopheles gambiae and their F1 generation shows that 100% (n = 40) of the tested population was A. coluzzi. This represents the predominant species of Anopheles in the area of study. The data obtained shows that the phytochemicals derived from T. occidentalis leaf extracts could be applied as an alternative mosquito vector control agent or in integrated pest management programs.
Anopheles coluzzi, F1 generation, Leaf extract, Malaria, vector control, Telfairia occidentalis SINE 200 PCR
Mosquitoes serve as vectors for pathogenic organisms to man and animals resulting in major infectious diseases in their hosts. Mosquitoes constitute a severe nuisance for human and domestic animals [1]. Approximately, 228 million cases of malaria were recorded worldwide in 2018. Malaria still kills more than 600,000 individuals every year, 90% of this in sub-Saharan Africa and mostly children under five [2]. Nigeria has the highest-burden, and mortality due to malaria alone accounting for ~27% of all global cases [3]. Insecticide resistance has been developed in mosquito populations as a result of repeated use of synthetic chemicals in mosquito vectors or and agricultural pest controls [4]. Plants are rich in bioactive compounds and hence have been used as alternative mosquito vector control agents [5]. Traditional communities across the world including Nigeria have been using plant products such as Moringa olifera against insect vectors. The bioactive compounds derived from these plants could be used to control larvae, pupae or adult mosquitoes as larvicides, pupicides and repellant agents respectively as observed in many studies worldwide [6].
Telfairia occidentalis (Hook fluted pumpkin), is belongs to the family Cucurbitaceae and is a dicotyledonous plant. It consists of more than 700 species distributed across the warm regions of the world [7]. It is an indigenous plant to South-eastern part of Nigeria and gradually spread all over Nigeria. T. occidentalis is an important seed and leaf vegetable and seems to be propagated in the entire Nigeria due to its nutritional, health, medicinal and commercial benefits. Additionally, it can be propagated in both dry and wet seasons in Nigeria. Telfairia leaf is rich in Magnesium, Iron and fibers [8] and is used as food supplements. In a recent study, it has been demonstrated that Telfairia leaf contains free soluble phenol content that exceeds the bound phenols counterpart. These free soluble polyphenols exhibit higher antioxidant activity as characterized by their free radical scavenging and higher reducing power ability compared to their bound phenol counterpart. It has also been recorded that the leaf extract of Telfairia contained a significant amount of phenols, flavonoids and vitamin C among other bioactive compounds [9].
The A. gambiae sensu lato (A. gambiae complex) consists of: A. bwambae, A. melas, A. arabiensis, A. merus, A. quadriannulatus, A. coluzzi, A. gambiae sensu stricto (ss) and A. ampharicus [10] which are known as the major vectors of malaria. Information on the larvicidal effects of T. occidentalis on the malaria vectors has been limited in different parts of the world despite the threats posed by the mosquito vectors and the need to adjust to sustainable vector control. This study aims to assess the larvicidal activity of the leaf extract of T. occidentalis on the larvae of the major human malarial vector, Anopheles coluzzi. To the best of our knowledge, this is the first evidence-based report of the larvicidal assessment of the leaf extract of T. occidentalis against A. coluzzi from Nigeria.
The leaf of T. occidentalis was shade dried immediately after collection from the Nayi-Nawa area Gombe, Nigeria. The dried leaf was powdered with the aid of an electrical blender until finely ground.
300 mL of methanol was used to extract the 100 g of finely ground leaf powder for 8 hours in a Soxhlet apparatus. A rotary evaporator was used to remove the solvents from the crude extracts, which was then collected in a beaker. 1 g of the crude extract was dissolved in 100 mL of methanol to form a stock solution (i.e. 1% extract). Different concentrations i.e. 20, 50, 100, 200, 400, 600, 800 and 1000 ppm were prepared from that stock solution.
The adult blood-fed Anopheles mosquitoes were collected indoors in about six rooms at Kwadon village in Yamaltu Deba Gombe State with the consent of the owners of the rooms. The blood-fed female Anopheles mosquitoes were transported to the insectary in Gombe State University College of Medical Sciences. The mosquitoes were fed with 10% sucrose. After two days, each gravid mosquito was put into a prepared 1.5 mL microcentrifuge tube for forced egg-laying. Each mosquito that laid the egg was morphologically identified as A. gambiae sensu lato prior to molecular species Identification. The identification was focused on the mosquitoes' legs, tarsus, abdomen, tufts, palps, veins, terga and wing. The genomic DNA of these mosquitoes was extracted and identified molecularly by SINE PCR. The emerged larvae of Anopheles mosquitoes were reared in the insectary of Gombe State University. A 3:1 ratio of biscuit and yeast was used to feed the mosquito larvae. The mosquitoes were maintained in the insectary condition at 75-85% relative humidity, 28 ± 2 ℃, as well as 14 h light and 10 h dark photoperiod cycle. The larvicidal activity of T. occidentalis was assessed using the first filial generation of the mosquito samples.
The larvicidal activity of T. occidentalis against A. coluzzi was evaluated using the method recommended by World Health Organization [11]. The condition in the bioassay lab was maintained at 75-85% R.H. and 27 ± 2 ℃. To each an individual disposable paper cup containing 99 mL of deionized water and 1 mL of desired target concentration of plant's extract, twenty of the third instar larvae was transferred and the larvicidal activity was recorded. Three replicates (Table 1) were set up for each concentration and the controls were set up with 99 mL of deionized water and 1 mL of methanol. Abbott's formula was used to correct the control mortalities [12] and the regression equation, lethal concentrations (LC50, LC90), Chi square as well as 95% Confidence Limit were calculated using Probit analysis [13].
Table 1: Assessment of larvicidal activity of the T. occidentalis leaf extract on A. coluzzi. View Table 1
The species of A. gambiae s. l. were identified morphologically using Gillies and Coetze [10] Morphological keys with aid of stereomicroscope with a clearer magnification of eye lens. The dark spot at the upper margins of the wings which is common to all anopheles was focused on identifying the species. The observation was focused on the mosquitoes' legs, tarsus, abdomen, tufts, palps, veins, terga and wing.
The genomic DNA of the adult parent A. gambiae s.l. followed by some of their larval colony (A. coluzzi) were extracted using TAN Bead Nucleic Extraction Kit (Taiwan Advanced Nanotech, Taiwan) and all their molecular forms were determined according to the SINE PCR method with primer sequences forward - SINE200_F- TCGCCTTAGACCTTGCGTTA and Reverse SINE 200_R- CGCTTCAAGAATTCGAGATAC. Using the above specific primers adopted elsewhere [14]. SINE 200 region was amplified in Anopheles gambiae population. Briefly, to the 14 μL of the prepared PCR Master Mix (Sigma-Aldrich, Germany), 1 μL of genomic DNA was added. The DNA Thermal Cycler (DCL25, Dynalon) was used for the PCR amplification using the following program condition: One cycle of Initial denaturation step at 95 ℃ for 5 min; 35 cycles of three steps: Denaturation at 94 ℃ for 30 s, annealing at 54 ℃ for 30 s, extension at 72 ℃ for 60 s; and a finally one cycle of final extension at 72 ℃ for 10 min. The PCR amplicons were run on gel electrophoresis for 30 min in 2% agarose gel (GeneON, UK) mixed with DNA loading dye (Blue, Bioline USA) at voltage = 110 volts and Current = 300 mAh for 30 min in the Gel tank (SCIE-PLAS, UK). The hyper-ladder IV, a 100 bp increment Ladder (Bioline, USA) a ready-to-use 10 fragments (100-1000 bp) DNA marker was used to compare the sizes of the amplified fragments. The gel was visualized with UV-trans-illuminator (biostep, bioview) and photographed under the UV-trans-illuminator.
The mortality observed during the larvicidal assessment of the plant leaf extract was corrected using Abbott's formula. The SPSS software was used for the analysis of the experimental data to determine the lethal concentrations, chi square value, 95% confidence intervals and regression equations (Y = mortality; X = Concentrations).
The blood-fed Anopheles mosquito samples collected indoor at Kwadom Village Yamaltu Deba Gombe used in this research were morphologically identified using Gillies and Coetze morphological keys [10] and found that all the Anopheles samples were A. gambiae s.l. This study is further to give a little insight into the diversity of morphological and morphologically indistinguishable species in the area of study.
Table 2 shows the total results obtained from the amplification of the SINE 200 region in A. gambiae complex to identify three of its member's i.e. A. gambiae s.s. (S form), A. coluzzi (M form) and An. arabiensis. The first lane (M) constitutes a hyper ladder IV that calibrates 100 bp size. Lanes L2 to L5 represents the amplified SINE 200 region of A. gambiae larvae while a lane L6-L11 represents the adult A. gambiae (Figure 1).
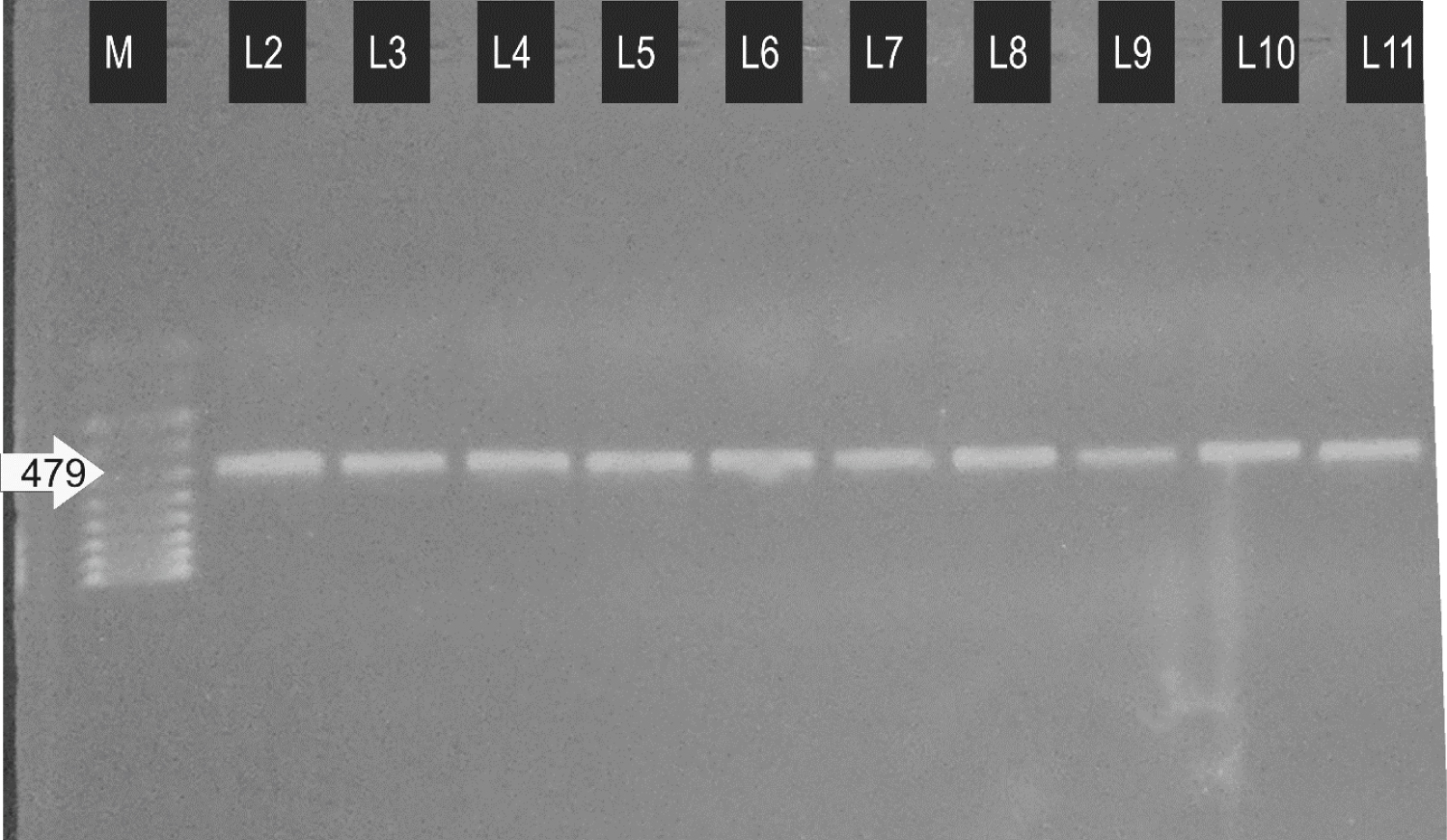 Figure 1: PCR amplification of SINE 200 for identification of A. gambiae complex species.
Figure 1: PCR amplification of SINE 200 for identification of A. gambiae complex species.
M = 100 bp Ladder; L2-L5 = A. coluzzi (larvae); L6-L11 = A. coluzzi (Adult)
View Figure 1
Table 2: Molecular species identification of A. gambiae s.l. by SINE 200 PCR. View Table 2
The result in Figure 1 showed that 100% (n = 40) of the analyzed twenty (20) adult parent mosquitoes and their 20 randomly selected F1 colonies (larvae) were all identified as Anopheles coluzzi with an average band size of 479 bp on agarose gel [14].
The assessment of larvicidal activity of the T. occidentalis (methanolic) leaf extract on malarial vector, A. coluzzi was presented in Table 1. Different levels of mortality have been observed across the three replicates exposed to the extract. 0% dead has been observed in the control group (0% extract concentration). At the lowest concentration i.e. 20 ppm, only approximately 8.3% mortality was observed in the mosquito population. Similarly, at 50 ppm concentration, only approximately 15% mortality was observed with little increase compared to the 20 ppm concentration. This result indicates that other higher concentrations of the extract have higher activity compared to the 50 ppm concentration. In 100 ppm, an increase in mortality was observed of about 20% of the 50 ppm (i.e. 35%). Mortality of more than 50% of larvae was observed in at least 200 ppm while 95% in at least 400 ppm with 100% mortality of at least 600 ppm concentration.
Table 3 showed the LC50 and LC90 values exhibited by the T. occidentalis were 75.56 ppm and 266.31 ppm respectively. This data suggest that the extract has lethal concentration of about 50% and 90% of the mosquito population at 75.56 ppm and 266.31 ppm respectively. The regression equation was Y = -4.39988 + 2.34248X. The 95% lower and upper confidence limit for both LC50 and LC90 were 63.15 ppm and 91.02 ppm as well as 161.44 ppm and 306.63 ppm respectively. The Chi square value for the assessment was 5.21.
Table 3: LC50 and LC90 of T. occidentalis against human malaria vector, Anopheles coluzzi. View Table 3
The use of plants in place of synthetic chemicals in bio-control of mosquitoes has been gaining attention across the world including Nigeria which is due to the sustainable nature of the plant pesticides [15]. The methanolic plant's leaf extract showed significant larval mortality against the Anopheles mosquito species. The result indicated that the increase in mortality was observed with increase in the concentration of the extract. This data suggests that the activity of the T. occidentalis leaf extract was concentration-dependent. The mortality observed in the tested larvae could be associated with the availability of bioactive compounds present in the leaf extract [16]. No mortality has been observed in the control group (0% concentration of the extract). This could validate the claim that the mortality observed in the treatment group was due to the effect of the extract. Thus, the leaf extract of this plant exhibited larvicidal activities on A. coluzzi. Similarly, several plants have been evaluated for larvicidal pupcidal as well as adulticidal activity against several pest insects including mosquitoes. In one separate study, the ethanolic and aqueous extract from moringa has been found to exhibit larvicidal properties against the Aedes mosquitoes [17]. Chromolaena ordata has been observed to have insecticidal activity against adult stage of American cockroach, Periplaneta Americana [18] and its oil extracts against the larvae and adult of mosquitoes of family Culicidae [19]. Moreover, V. amygdalina leaf powder has been reported to have potential in controlling of C. maculatus (Cowpea bruchid) infesting stored seed [20].
The Chi-square value was significant at P > 0.05 level. This could suggest that there was a significant difference in the activity of the plant's extract at different concentrations. Due to the non-toxicity nature of plant-derived chemicals (phytochemicals), they serve as the useful material in the development of sustainable and safer insecticides and pesticides [21]. T. occidentalis, as compared to other plant extracts, such as M. olifera leaf extract which has been studied extensively to have exhibited both larvicidal and pupicidal activity against different larval instars of different groups of mosquitoes including Aedes (Aedes egypti and A. albopictus), Culex quinque fasciatus, and Anopheles mosquitoes [22]. The result obtained in this study was encouraging as it showed the activity of the extract against the target mosquito population. The observed mortality in the larval population could be associated with the presence of bioactive compounds such as flavanoid, phenolic compounds etc. contained in the plant [23]. The leaf extract of T. occidentalis has been demonstrated to exhibit antimalarial activity in vivo in mice model. The mice were infected with Plasmodium berghei to establish an infection. The T. occidentalis leaf extract was shown to exhibit antimalarial (antiplasmodial and blood schizontal) activity in both early and established malaria infections [24].
Most of the highly toxic plant extracts against the mosquitoes are likely equally toxic to human and domestic animals. Since water served as a breeding site for these mosquitoes especially Anopheles which mostly bred in clean water body (which could be a drinking water), there is a risk factor concerning the application of these toxic plants in mosquito vector control intervention [16]. T. occidentalis is very less or nontoxic to human and important animals [25]. More importantly, the result of this study shows that the extract exhibited a promising larvicidal activity as evident in its effect observed in the present study. Plant-derived chemicals offer a promising advantage over the synthetic chemicals pesticides due to their unique features such as eco-friendly, biodegradability, lesser or nontoxicity, and lesser chance to develop resistance by the vectors. Therefore, the leaf extract of T. occidentalis could play an important role in mosquito vector control intervention program.
In all sample being tested (Figure 1), the amplicon size of 479 bp was observed. This result suggests that they were A. coluzzi population. It has been previously reported that, there exist a remarkable difference among the molecular forms of A. gambiae at locus S200 × 6.1. The PCR assay employed in this study could identify three (3) different members of A. gambiae complex due to the differences in the amplicon size upon PCR and electrophoresis [14]. Several methods have been developed for the identification of individual members of A. gambiae complex such as ribosomal DNA PCR (rDNA PCR) method. This rDNA PCR method identifies the five majorly distributed members of A. gambiae which are morphologically indistinguishable and are the major malarial vector in Sub-Saharan Africa. The rDNA PCR method is effective and is based on the species-specific sequences in the rDNA intergenic spacers. This could identify both species and intergenic hybrid species of A. gambiae members via PCR amplification with specific primers. The method could identify A. arabiensis, A. gambiae s.s., A. quadriannulatus, A. melas or A. merus using a cocktail of five 20 nucleotides [26].
The consensus analysis of the sequence of SINE200 signifies that it is a characteristic transfer RNA-related (tRNA-related) Short Interspaced Nuclear element. It contains A and B boxes in the promoter region of polymerase III at the 5' end of tRNA-related region. At the 3' end, there is also a variable number of the tandem repeat of AAG which is also a typical tRNA-related SINE element [27]. There also exists a conserved sequence at the middle of the SINE200 element which is not a tRNA-related sequence as demonstrated by recent studies in other eukaryotic SINEs [28]. Moreover, the intentional mismatch primers (PCR-IMP) and Restriction Fragment Length Polymorphism (RFLP-PCR) assays have been employed currently in identification of A. gambiae have been based on the single/or few nucleotide mutation recognition along the sequences. Therefore, it is subjected to homoplasy [29]. As opposed to those assays, the present study employed the PCR assay that is based on the specific and irreversible insertion of some nucleotide sequence along the SINE200 region of the members of A. gambiae complex. A 230 bp insertional element has been observed in the A. coluzzi (M form) and absent in S form. This specific and irreversible insertion has made it possible to differentiate among the three members of the A. gambiae complex: A. coluzzi, A. arabiensis and A. gambiae s.s, as described elsewhere [30]. The observed band size of 479 bp from Figure 1 indicates the presence of specific and irreversible insertion of a 230 bp element in the SINE200 region of the mosquito sample being tested. Figure 1 indicated that the species (sample) collected in the study area were A. coluzzi (with 479 bp) band size. This data could suggest that the predominant Anopheles species in the study area was A. coluzzi (M form).
The finding of the present study shows that the leaf extracts of T. occidentalis exhibited larvicidal activity (lesser concentration with high efficacy) against A. coluzzi and hence could be applied as a mosquito vector control agent or employed in integrated pest management programs. It also indicates that the predominant species in the study area was A. coluzzi population that is susceptible to leaf extracts of T. occidentalis.
The authors are thankful to Gombe State University for providing the facilities as well as Dr. Sulaiman Muhammad of Department of Biology and Dr. Lazarus J. Goje of Department of Biochemistry Gombe State University Nigeria for reading through the manuscript.
The authors declare no competing and conflict of interest.
All authors have contributed equally in this study.