Chlorpyrifos is a broad-spectrum chlorinated organophosphate pesticide with a multipurpose use worldwide. However, its use and/or misuse could put a real threat on non-targets including humans and domestic animals.
To assess the toxic effect of chlorpyrifos on serum glucose, bilirubin, liver enzymes, renal parameters, protein profile, and some electrolytes in adult male domestic rabbits.
The oral LD50 of chlorpyrifos was determined, and then a daily dose of 1/10 LD50 was given orally to rabbits for 6 weeks. Control animals were given distilled water. Blood samples were collected and analyzed weekly.
Administration of 1/10 LD50 chlorpyrifos (33.3 mg kg-1 body weight) provoked a general increase in serum glucose and bilirubin compared to control rabbits, with maximum % differences of 27.2 and 15.2%, and p-values of 0.002 and 0.044, respectively. Alanine amino transferase (ALT), aspartate amino transferase (AST), gamma-glutamyl transferase (γ-GT), and alkaline phosphatase (ALP) showed significant elevation registering maximum % differences of 30.2, 37.1, 29.4 and 26.8%, and p-values of 0.007, 0.003, 0.008 and 0.002, respectively. Conversely, cholinesterase (ChE) was significantly inhibited recording a maximum % difference of 64.6% (p < 0.001). Serum urea and creatinine displayed a significant increase (maximum % differences = 43.6 and 26.4%, p-values = 0.002 and 0.018, respectively), whereas total protein, albumin, and globulin exhibited significant decreases (maximum % differences = 25.5, 27.3 and 28.2%, p-values of 0.013, 0.010 and 0.009, respectively). Hypercalcemia and hypophosphatemia were also observed in chlorpyrifos-treated rabbits.
Exposure to chlorpyrifos even at a low dose can impair functions of vital mammalian organs.
Chlorpyrifos, Rabbits, Glucose, Liver biomarkers, Renal parameters, Electrolytes
Chlorpyrifos (O,O-diethyl O-3,5,6-trichloro-2-pyridyl phosphorothioate), a synthetic organophosphorus insecticide was first registered in the United States in 1965. The most common trade name is dursban, and it is absorbed via the gastrointestinal tract and to a lesser extent through intact skin and by inhalation [1]. The US Environmental Protection Agency categorized chlorpyrifos as a Class II toxin: Moderately toxic [2]. A common measure of acute toxicity is the lethal dose (LD50). The oral LD50 (the dose that kills 50% of test animals population when administered by the oral route) is expressed in terms of weight of test substance per unit weight of test animal mg kg-1 body weight. The acute oral LD50 of chlorpyrifos was estimated to be 135-163 mg kg-1 body weight in rats, 504 mg kg-1 body weight inguinea pigs, and up to 2000 mg kg-1 body weight in rabbits [3]. However, no previous study determined the exact or the specific oral LD50 in adult male domestic rabbit.
Chlorpyrifos affects the nervous system by inhibiting the breakdown of the neurotransmitter acetylcholine. When target and non-target organisms are exposed, chlorpyrifos inhibits irreversibly the cholinesterase (ChE) enzyme, which prevents breakdown of acetylcholine in the synaptic cleft. The resulting accumulation of acetylcholine at cholinergic receptor sites causes overstimulation of the neuronal cells and cholinergic crisis, which leads to neurotoxicity and eventually death [4,5]. Chlorpyrifos affects also ChE levels differently in various systems throughout the body. Scientists have observed plasma and red blood cell ChE inhibition in experimental animals at doses lower than those required to cause ChE inhibition in the brain [6,7].
After exposure to the toxic doses of chlorpyrifos, clinical signs of poisoning appear within a few minutes or after few hours and this depends on the degree of absorption, distribution, metabolism of the insecticide inside the body and its excretion [8]. Chlorpyrifos poisoning shows the cardinal signs of organophosphorus compounds that is represented by muscarinic signs, which appears early that includes: salivation, lacrimation, sweating, colic with diarrhea, dyspnea with profuse nasal discharges, urination, defecation, miosis and bradycardia [9,10], followed by nicotinic signs that includes: muscle fasciculation, muscle twitching, abnormal gait, in coordination and tremors [9,11], eventually nervous system effects which includes hyperactivity, anxiety, tremors followed by convulsions, depression and finally death as a result of respiratory failure [12,13].
Most previous studies focused on the assessment of neurotoxicity of chlorpyrifos and little attention has been paid to its toxic effect on other mammalian organs. Alterations of hepatic and renal biomarkers, and metabolic profile have been reported in response to organophosphorus pesticides exposure including chlorpyrifos in experimental animals as well as in humans [14,15]. This investigation has been undertaken in the purpose of expansion of our awareness and knowledge on the physiological hazards of chlorpyrifos, which is being broadly used in Gaza Strip for the control of coleoptera, diptera, homoptera and lepidoptera in soil or on foliage in a wide range of crops including fruits, vegetables, flowers, palm, alfalfa, cereals, maize, sorghum, glasshouse and outdoor ornamentals, mushrooms, turf, and in forestry. Also chlorpyrifos is used for control of household pests, mosquitoes, and in animal houses [16].
Throughout the year 2020, more than 544.4 metric tons of pesticides are used in the Gaza strip. The insecticides represent 232.5 metric tons of these pesticides, 18.1 metric tons of these insecticides are chlorpyrifos [16]. The use and/or misuse of such highly toxic compounds either in the agricultural fields or for domestic purposes constitute a real threat on humans and domestic animals. Several cases of chlorpyrifos poisoning or death were previously recorded among farm workers in Gaza Strip [17,18]. Therefore, this study is aimed to assess the toxic effect of a sublethal dose of chlorpyrifos on serum glucose, bilirubin, liver enzymes, renal parameters, protein profile and some electrolytes in adult male domestic rabbits. The rationale for selecting rabbit as the model/experimental animal for the present study resides in the facts that this animal is very docile and non-aggressive and hence easy to handle and observe, has less health problems, very economical compared with the expense of the large animal category that needs stringent rules of the local ethical committee. The findings will raise our understanding on the extent of chlorpyrifos toxicity, and can then be extrapolated to human beings to assess the potential hazards in the human populations due to chlorpyrifos exposure.
Healthy adult male domestic rabbits weighting 1,000-1,200g were used in this study. Animals were maintained under the ambient conditions in the animal house in the Department of Biology, Islamic University of Gaza. They were fed on a commercial balanced diet specially prepared for rabbits (Anber). The diet and tap water were lead free and offered ad libitum throughout the experimental period. The Institutional Animal Ethics Committee approved the experimental protocol (No. 04/2019).
The study had two phases: The first phase was to determine the oral LD50 of chlorpyrifos and the second phase was to investigate the toxic effect of a sublethal dose of chlorpyrifos (1/10 LD50) on serum glucose, bilirubin, liver enzymes, renal parameters, protein profile and some electrolytes in male domestic rabbits. Analytical-grade chlorpyrifos (98% purity) used in this study was purchased from Payer AG Chemical Company, Germany.
A total number of 80 rabbits were used for the determination of LD50 of chlorpyrifos. Animals were divided into 10 groups (8 rabbits/group). The first nine groups (1-9) were administered different single doses of chlorpyrifos ranging from 150 to 550 mg.kg-1 body weight as shown in Table 1. The 10th group was served as a control group. Chlorpyrifos was given orally using a special stomach tube with a smooth tip to protect the interior lining of the oral and buccal cavity from injury. The rabbit was held between its two ears so that the esophageal opening was clearly and unobstructively opened. The gastric tube was filled with the required dose of chlorpyrifos and then smoothly inserted until it adequately enters the upper part of the esophagus where its contents were emptied. The animals were observed for mortality during the 48-h observation period. The LD50 was determined by the graphical method [19].
Table 1: The first nine groups (1-9) were administered different single doses of chlorpyrifos. View Table 1
After the determination of chlorpyrifos LD50 in the first phase, a sublethal oral dose of chlorpyrifos (1/10 LD50) was given daily in the second phase to assess the toxic effect of chlorpyrifos on serum glucose, bilirubin, liver enzymes, renal parameters, protein profile and some electrolytes in male domestic rabbits. Animals were divided into two groups: Control and experimental groups. The control group comprised 36 rabbits (six rabbits were housed in each cage) and the experimental group also included 36 rabbits (six rabbits were housed in each cage). The experimental groups were orally administrated 1/10 LD50 chlorpyrifos daily throughout the experimental duration of 6 weeks. Control animals were given distilled water.
Bodyweight of both control and experimental rabbits was measured using a sensitive balance (Model: ONA-15, made in Istanbul 2013) and weights were recorded to the nearest gram. Toxicity symptoms, abnormalities, and mortalities of both control and experimental animals were observed.
Six animals were taken from each control and experimental groups every week. Blood samples were collected into centrifuge tubes (without any anticoagulant) from rabbit marginal ear vein using the incision method [20]. The samples were left for about 15 min to allow blood coagulation. Then, the samples centrifuged at 3,000 rpm for 20 min. Serum samples were separated into glass tubes, labeled, and kept in the refrigerator for biochemical assay.
Various methods of serum analysis of different biochemical parameters in male domestic rabbits are summarized in Table 2.
Table 2: Biochemical assay of different parameters in male domestic rabbits. View Table 2
Data were statistically analyzed using IBM SPSS statistics version 22. Means were compared by an independent-sample t-test. Probability values (P) were obtained from the student's table of "t" and significance was at p < 0.05. The percentage difference was calculated according to the formula: percentage difference equals the absolute value of the change in value, divided by the average of the two numbers, all multiplied by 100. Percent difference = (|(V1-V2)|/((V1 + V2)/2))*100. The graph logarithmic scale of oral LD50 of chlorpyrifos was plotted using the Microsoft Excel program 2013.
The experimental trials for oral LD50 determination of chlorpyrifos after 48 h of administration in male domestic rabbits revealed that the mortality commenced at 200 mg kg-1 body weight, recording mortality percentage of 12.5% (Table 3). Increasing chlorpyrifos dose to 250, 300, 350, 400, and 450 resulted in mortality percentages of 37.5%, 50.0%, 50.0%, 62.5, and 75.0%, respectively. The mortality rate was a function of dose increase. The maximum concentration of chlorpyrifos that kills all animals in the group was found to be 500 mg kg-1 body weight. The calculated oral LD50 of chlorpyrifos in male domestic rabbits from the linear regression was found to be 333 mg kg-1 body weight (Figure 1).
Table 3: Mortality percentage of male domestic rabbits after 48 h of oral administration of different doses of chlorpyrifos. View Table 3
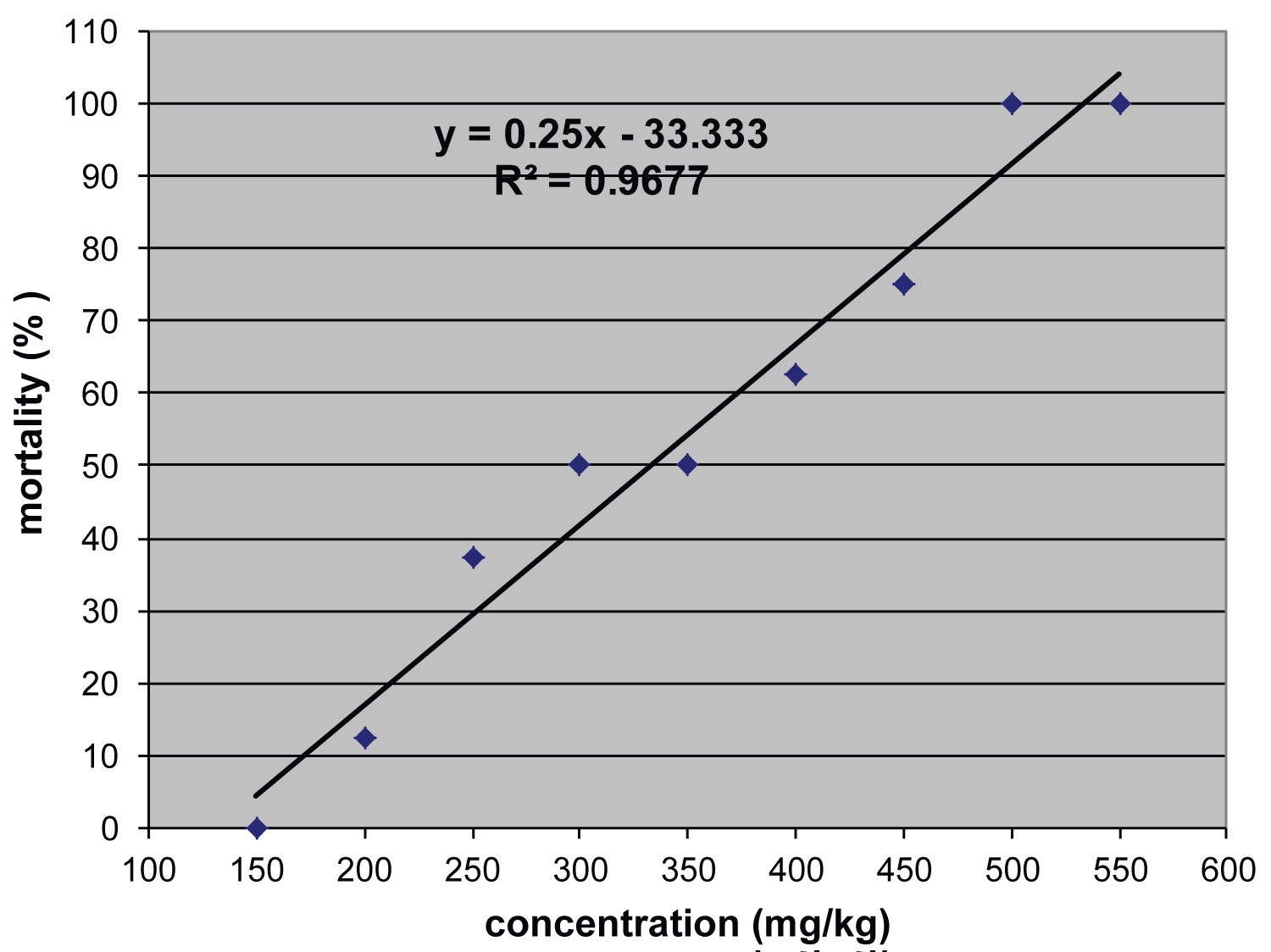 Figure 1: Logarithmic scale of oral LD50 of chlorpyrifos in male domestic rabbits. (LD50 = 333 mg kg-1 body weight).
View Figure 1
Figure 1: Logarithmic scale of oral LD50 of chlorpyrifos in male domestic rabbits. (LD50 = 333 mg kg-1 body weight).
View Figure 1
The bodyweight of rabbits after 6 weeks of daily oral administration of 1/10 LD50 chlorpyrifos (33.3 mg kg-1 body weight) was significantly decreased compared to controls (816.7 ± 35.8 versus 960.1 ± 33.4, p = 0.032). Chlorpyrifos-fed rabbits showed varying degrees of toxicity symptoms a few hours after dosing, which included diarrhea, disorientation, drowsiness and mild tremor. However, control rabbits did not display any toxicity symptoms. The mortality records showed that 3 chlorpyrifos-treated rabbits died during the 6-week experimental duration from the total number of 36 rabbits used in the experiment: 1 rabbit in the fifth week and 2 rabbits in the sixth week. However, none of the control rabbits died throughout the experiment.
Table 4 shows that oral administration of 1/10 LD50 chlorpyrifos increased the mean level of serum glucose in male domestic rabbits along the experimental period of 6 weeks compared to control animals. This increment was significant in the last four weeks, with a maximum % difference of 27.2% in the 3rd week (p = 0.002). Serum bilirubin was also increased, reaching a maximum % difference of 15.2 during in the 5th week of the experiment (p = 0.044).
Table 4: Effect of chlorpyrifos(1/10 LD50, 33.3 mg kg-1 body weight) on serum glucose and bilirubin levels in male domestic rabbits. View Table 4
As indicated in Table 5, ALT, AST, γ-GT and ALP activities were generally increased in chlorpyrifos-fed rabbits than controls, showing maximum significant % differences of 30.2, 37.1, 29.4 and 26.8 in the 5th, 3rd, 6th and 4th, weeks, respectively (p = 0.007, p = 0.003, p = 0.008 and p = 0.002). Conversely, ChE activity was significantly inhibited all over the experimental periods examined, with a maximum % difference of 64.6 in the 4th week of the experiment (p < 0.001).
Table 5: Effect of chlorpyrifos (1/10 LD50, 33.3 mg kg-1 body weight) on serum ALT, AST, γ-GT, ALP, and ChE activities in male domestic rabbits. View Table 5
The mean levels of serum urea and creatinine were elevated in response to chlorpyrifos intake throughout the whole experiment (Table 6). The significant increase in urea commenced from the 3rd week and reached its maximum % difference of 43.6 at the end of the experiment (p = 0.002). Serum creatinine fluctuated across the experimental period reaching its maximum % difference of 26.4% in the 4th week of the experiment (p = 0.018).
Table 6: Effect of chlorpyrifos (1/10 LD50, 33.3 mg kg-1 body weight) on serum urea and creatinine levels in male domestic rabbits. View Table 6
Table 7 revealed a general decrease in the mean levels of total protein, albumin and globulin in chlorpyrifos-treated rabbits all over the weekly intervals of the experiment. The significant decreases were detected during the last four weeks, recording maximum % differences of 25.5, 27.3 and 28.2 in 4th, 6th and 6th weeks for total protein, albumin and globulin, with p = 0.013, p = 0.010 and p = 0.009, respectively.
Table 7: Effect of chlorpyrifos(1/10 LD50, 33.3 mg kg-1 body weight) on serum total protein, albumin and globulin levels in male domestic rabbits. View Table 7
As illustrated in Table 8, serum calcium concentration was increased whereas serum phosphorus was decreased upon chlorpyrifos administration along the experimental intervals examined. The significant change in serum calcium appeared in the 4th and 5th weeks with maximum % difference of 16.8 in the 4th week (p = 0.034). Serum phosphorus exhibited some variations throughout the experiment displaying a maximum % difference of 14.3 in the 2nd week of the experiment (p = 0.032).
Table 8: Effect of chlorpyrifos (1/10 LD50, 33.3 mg kg-1 body weight) on serum calcium and phosphorus concentrations in male domestic rabbits. View Table 8
Extensive and multipurpose use and/or misuse of organophosphorus pesticides mainly chlorpyrifos in Gaza Strip [34], poor follow of protective measures [35,36], and lack of knowledge and awareness about the harmful impact of chorpyrofis [37], necessitated the conduction of this research for better understanding the extent of chlorpyrifos toxicity on various mammalian organs and general health hazards of humans and domestic animals. The first step is to determine the LD50 which is a major parameter in measuring acute toxicity and also as an initial procedure for general screening of chemical and pharmacological agents for toxicity [38]. The acute oral LD50 of chlorpyrifos was roughly estimated at a wide range of up to 2000 mg kg-1 body weight in rabbits [3]. However, no published data are available on the exact or the specific oral LD50 of chlorpyrifos in adult male domestic rabbit. In this study, the logarithmic scale showed that the oral LD50 of chlorpyrifos in adult male domestic rabbits was 333 mg kg-1 body weight. This coincides with the fact that chlorpyrifos was categorized as one of the moderately toxic organophosphorus compounds, their oral LD50 ranging between 50-500 mg kg-1 body weight. Chlorpyrifos is moderately toxic to mice, rats, sheep and guinea pigs [13,39].
Daily oral administration of 1/10 LD50 (33.3 mg kg-1 body weight) chlorpyrifos caused a significant reduction in the bodyweight of rabbits. Similar result was previously reported [40,41]. Such reduction in the body weight may be a result of the combined action of cholinergic and oxidative stress, and/or due to the overall increased degradation of lipids and proteins as a result of the direct effect of chlorpyrifos exposure [42,43]. The observed decrease of protein content in chlorpyrifos-treated rabbits does support this view. The mortalities occurred in the last two weeks of the experiment as a result of repeated chlorpyrifos intake may be largely attributed to diarrhea that may be related to the cholinergic crisis, a consistent sign in organophosphate poisoning [34,44]. Diarrhea is a main clinical sign observed in male Wister rats orally supplemented with chlorpyrifos [45].
Results presented in this study demonstrated significant increase in serum glucose commencing at the 3rd week of chlorpyrifos intake, implying that this organophosphorus compound disturbs glucose homeostasis. Such finding is in agreement with that reported by other authors [46,47]. The proposed mechanisms involved in chlorpyrifos-induced hyperglycemia may include: 1) Decrement in insulin secretion as a result of the degenerative action of chlorpyrifos on the beta cells of Langerhans islets in the pancreas [48]; 2) Stimulation of hepatic gluconeogenesis and glycogenolysis [49], and 3) Activation of the hypothalamic-pituitary-adrenal axis and secretion of glucocorticoids from adrenal cortex in response to adrenocorticotropic hormone release from the anterior pituitary gland. These adrenal cortical glucocorticoids act on the hepatocytes for activation of the gluconeogenesis pathway and in turn increase blood glucose levels [50]. Serum bilirubin was relatively increased in chlorpyrifos-treated rabbits, and this increase is in the line with other studies [43]. The change in serum bilirubin which is accepted as an indicator of liver function may provide an evidence on hepatotoxicity induced by chlorpyrifos [15].
In general, serum ALT, AST, γ-GT and ALP activities were significantly elevated in chlorpyrifos-fed rabbits with respect to controls. This elevation in liver enzymes was documented elsewhere in the literature [43,51,52]. The normal function of both ALT and AST is to catalyze the conversion of alpha-ketoglutarate and an amino acid to glutamate and another product. In the case of ALT, the amino acid and product are alanine and pyruvate. In the case of AST, the amino acid and product are aspartate and oxaloacetate. ALT plays an important role in glucose-alanine cycle, and AST in malate aspartate shuttle within cells [53]. γ-GT is involved in glutathione metabolism and drug and xenobiotic detoxification [54]. ALP removes the phosphate group from various proteins and nucleotides. ALP is a leading biomarker of hepatobiliary injury [55]. Consequently, alteration of serum ALT, AST and γ-GT is largely accepted as a conventional indicator of hepatotoxicity [56,57]. In this context, the observed elevation of these liver function biomarkers as a result of chlorpyrifos exposure could be attributed to their release from the cytoplasm into the blood circulation indicating a necrosis and inflammatory reactions [39], and reflect the alteration of the membrane permeability of the hepatocytes [58]. In contrary to elevation of transaminases, γ-GT and ALP, serum ChE activity was significantly inhibited in chlorpyrifos-supplemented rabbits. This anticholinesterase action is a good indication of intoxication with chlorpyrifos as organophosphorus insecticide, and was reported by other authors [34,59]. Inhibition of ChE activity results in accumulation of acetylcholine, the neurohumoral mediator of the cholinergic junctions, and consequent excessive activation of acetylcholine nicotinic and muscarinic receptors at neuromuscular junction and in the autonomic and central nervous system. The clinical signs and symptoms of such cholinergic crisis are broadly mentioned in the literature [11,50,60]. Hence, one can say that diarrhea, disorientation, drowsiness, mild to severe tremors, and some cases of death observed among chlorpyrifos-fed rabbits are clinical manifestation of chlorpyrifos toxicity.
The tested renal parameters showed significant increase in serum urea and creatinine towards the end of the experiment in chlorpyrifos-treated rabbits, implying the detrimental effects of chlorpyrifos on kidney function. Elevation of urea and creatinine was reported in chlorpyrifos-treated Wistarrats [15,61]. Urea and creatinine are waste products of protein metabolism that are excreted through the kidney. As essential markers of kidney function, the recorded elevation of urea and creatinine following chlorpyrifos exposure ensures renal dysfunction [62]. Elevated creatinine in the serum reflects decreased glomerular filtration rate, while increased urea indicates dysfunctional reabsorption [63]. Urea levels can also be increased by other factors, such as dehydration, antidiuretic drugs and diet; however, increased creatinine is more specific to kidney damage [64,65]. Therefore, a combined elevation in urea and creatinine suggests a moderate to severe degree of kidney damage induced by chlorpyrifos.
In this investigation, the chlorpyrifos-supplemented rabbits exhibited significantly lower total protein, albumin and globulin levels than the control rabbits. Albumin, which is the most abundant blood plasma protein, is produced by the liver and several studies have shown that its production can be decreased by chlorpyrifos [66-68]. Since reductions in albumin levels are generally suggestive of liver disease, it is possible that chlorpyrifos alters protein and free amino acid metabolism and their synthesis within the liver. The hepato- and nephrotoxic effects of chlorpyrifos, as observed by impairment of liver and kidney biomarkers, do support this idea. The decline of total protein is further supported by the reduction in the body weight noted in chlorpyrifos-fed rabbits. Hypercalcemia and hypophosphatemia were also found in chlorpyrifos treated rabbits, implying that chlorpyrifos interferes with calcium and phosphorus homeostasis. Such alterations in serum calcium and phosphate were reported in male Wister rats, and explained on the basis that exposure to chlorpyrifos may disturb parathyroid glands and calcitonin cells of thyroid gland [69].
A pervasively-used chlorpyrifos helps in minimizing the damages caused by pests, but its exposure could be harmful for animal as well as human population. The present investigation provides an insight of chlorpyrifos induced toxicity on liver and kidney, and possibly the pancreas, the vital organs for maintaining the homeostasis. Increased damage to these vital organs even at low doses of chlorpyrifos could be compared with the adverse effect of chlorpyrifos on human population.
The authors declare no conflict of interest.