International Archives of Nursing and Health Care
Exploring the Contribution of Treatment Factors to Preferences for Smoking Cessation Interventions
Souraya Sidani*, Joan Brewster, Joyal Miranda, Shelley Walkerly and Emily Belita
School of Nursing, Ryerson University, Toronto, Canada
*Corresponding author: Souraya Sidani, PhD, Professor and Research Chair, School of Nursing, Ryerson University, 350 Victoria Street, Toronto, ON, M5B 2K3, Canada, Tel: 416-979-5000 x 2572, Fax: 416-979-5344, E-mail: ssidani@ryerson.ca
Int Arch Nurs Health Care, IANHC-2-028, (Volume 2, Issue 1), Review Article; ISSN: 2469-5823
Received: August 05, 2015 | Accepted: January 18, 2016 | Published: January 23, 2016
Citation: Sidani S, Brewster J, Miranda J, Walkerly S, Belita E (2016) Exploring the Contribution of Treatment Factors to Preferences for Smoking Cessation Interventions. Int
Arch Nurs Health Care 2:028. 10.23937/2469-5823/1510028
Copyright: © 2016 Sidani S, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution,
and reproduction in any medium, provided the original author and source are credited.
Abstract
Background: Preferences for treatment affect the poor uptake, adherence, and outcomes of smoking cessation interventions. This study addressed the need to examine what smokers like and dislike about smoking cessation interventions. The study aimed to describe adult smokers’ preferences for three interventions: nicotine replacement therapy, brief individual advice and group behavioral therapy, and to identify treatment-related factors underlying treatment preferences.
Methods: A cross-sectional design was used. Eligible participants were given information about each smoking cessation intervention, completed items to rate it relative to four attributes (appropriateness, effectiveness, severity of side effects and convenience of use), and asked to indicate what made the preferred intervention appealing. Logistic regression was used to examine the treatment attributes that were related to preferences. Content analysis was applied to identify additional factors that contribute to preferences.
Results: The majority of participants had a preference, with the nicotine gum (45.6%) and group behavioral (54.4%) therapy most frequently selected. Only the attribute of appropriateness (OR > 2.0) was significantly associated with preferences for nicotine gum and group behavioral therapy. The perceived effectiveness, convenience of use and format in which it is delivered (e.g. group dynamics) were additional factors underlying preferences.
Conclusions: The findings highlight the importance of assessing the smokers’ perception and preferences when selecting cessation treatment. Future research should examine the impact of providing the preferred intervention on abstinence.
Keywords
Smoking cessation, Preference, Appropriateness, Effectiveness, Convenience, Risk, Nicotine gum, Brief individual advice, Group behavioral therapy
Introduction
Pharmacological (e.g. nicotine replacement therapy) and behavioral (e.g. individual counseling) therapies have demonstrated low success rates, reaching up to 26% in clinical trials and 7% in practice [1,2]. Poor uptake and suboptimal adherence are two of many reasons for the observed low success rates [3]. Preferences for treatment, representing choice of intervention, have recently been acknowledged as factors affecting the poor uptake, adherence and outcomes of smoking cessation interventions [4]. Accordingly, Etter and Schneider (2013) highlighted the need to determine “what users like and dislike” about these interventions in order to “maximize their use and effectiveness” (p. 59) [5]. Whereas several studies investigated smokers’ preferences for a range of interventions, very few provided answers to the question: What makes a smoking cessation intervention desirable? This study addressed this question by exploring smokers’ preferences for pharmacological (i.e. nicotine replacement therapy) and behavioral therapies, and identifying treatment-related factors that contribute to expressed preferences. The selection of the latter factors was guided by a conceptualization of treatment preferences [6].
Adult smokers’ preferences for cessation interventions have been examined in four sets of studies. Each set was concerned with specific types of interventions. The first set included pharmacological interventions; the results consistently showed that a larger proportion of smokers chose NRT over bupropion [7-10]. The second set consisted of different modes of administrating NRT such as inhaler, spray, lozenge, and gum; the most preferred mode varied across studies [2,4,11]. The third set involved a variety of strategies to obtain assistance in smoking cessation; participants were interested in self-instructional over face-to-face strategies [12,], professional advice [13], individual or group-based support [14], educational programs and one-on-one consultation with health professionals [15], and abrupt quitting method [16]. The fourth set comprised pharmacological and behavioral therapies; the findings suggested an overall preference for behavioral support and NRT over medication [17-19].
Few studies explored factors contributing to the expressed preferences for smoking cessation interventions. The identified factors were perceived effectiveness of the intervention [10], length of using the intervention to achieve abstinence [9], and previous use of the intervention [8]. Other treatment-related factors that may influence smokers’ preferences include perceived attributes (appropriateness, effectiveness, risk, convenience) and reported prior use of the intervention [20]. Gaining an understanding of these factors is useful in modifying cessation interventions to make them attractive to smokers and to enhance their uptake, adherence, and effectiveness [6].
The overall purpose of this study was to explore smokers’ preferences for pharmacological and behavioral cessation therapies. Three interventions were selected: nicotine gum (4 mg), brief individual advice (given by a health professional), and group behavioral therapy. The specific study aims were:
1. To describe adult smokers’ preferences for the cessation therapies.
2. To examine the relationship of treatment-related factors (i.e. perception of attributes and previous use) with preferences expressed for each smoking cessation intervention.
3. To identify factors, in addition to treatment attributes measured in this study, that smokers take into consideration when selecting a cessation intervention.
Methods
Design
A cross-sectional design was used. Smokers interested in the study contacted the research assistant by telephone. The research assistant explained the study, obtained oral consent for administering questions to determine eligibility, assessed for the preset inclusion and exclusion criteria, and invited eligible participants to a data collection session held at the study research office. At the session, the research assistant obtained written consent and administered the questionnaire. The questionnaire contained items assessing socio-demographic characteristics and nicotine dependence, as well as attributes, previous use and preferences for the smoking cessation interventions. Open-ended questions were incorporated to identify additional factors underlying participants’ preferences. Participants were given $5 to cover public transportation expenses to attend the data collection session, and a $10 gift (coffee) certificate as a token for their participation. The order for presenting the three smoking cessation treatments was randomized to reduce the potential for a sequence effect.
Sample
Adult smokers were selected if they met the preset eligibility criteria. Inclusion criteria were: 1) age ranging from 25 to 65 years, 2) community-dwelling, non-institutionalized with a permanent home address, 3) ability to read English, and 4) self-report of smoking one or more cigarettes per day. The exclusion criteria entailed: 1) pregnancy: pregnant women may have a particular motivation to quit smoking, and 2) cognitive impairment, ascertained with a score < 24 on the Mini-Mental State Exam [21]: persons with such impairment may have difficulty understanding the study, providing informed consent, and responding to the questions.
Two strategies were used to recruit adult smokers. The first consisted of community media advertisement made in local newspapers with a wide distribution and on widely accessed websites. The second strategy involved distribution of flyers advertising the study at community centers and hospital outpatient clinics. All advertisements specified the general selection criteria (age and smoking status) and provided the study contact information.
Sample size
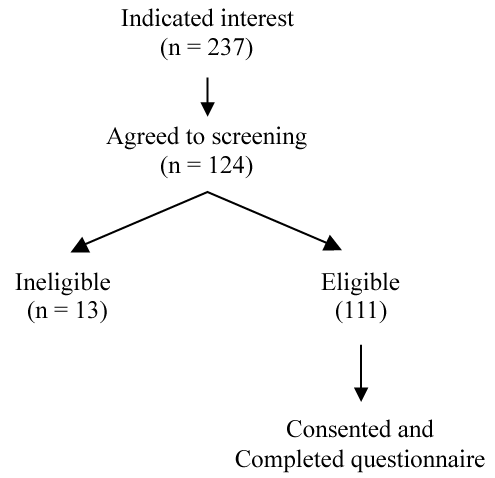
The flow of participants is reported in Figure 1. Reasons for ineligibility were: age not within the pre-specified range (n = 3); not having a permanent address (n = 6); or not smoking daily (n = 4). The sample size (n = 90, 81% of eligible) was considered acceptable to address the study aims. It provided adequate power to detect the influence of treatment-related factors on preferences, using the rule of having 5-10 cases per predictor included in the regression model [22]. As stated in study aim 2, the number of predictors was 5 and a sample size ranging from 25-50 was considered suitable for the planned regression analysis.
Smoking cessation interventions
The three smoking cessation interventions were selected to represent a range of treatments that varied in terms of: active ingredients, mechanisms of action, mode and dose of delivery, attributes (i.e. effectiveness in promoting abstinence and dealing with craving, ease of implementation, benefits, and risks), and availability (i.e. ease of access). A description was developed for each intervention. The description entailed the intervention’s goal (what it is set to achieve), activities (what it involves), schedule (how it is delivered and by whom), benefits (how effective it is), and risks (what are possible side effects). The information was synthesized from relevant literature and summarized in Table 1.
![]()
Table 1: Descriptions of smoking cessation interventions.
View Table 1
Variables and measures
Socio-demographic characteristics: Age, sex, marital status, education, employment status, and race were assessed with standard questions. Education was quantified by the number of years of formal schooling.
Nicotine dependence: Nicotine dependence was measured with the 6-item Fagerstrom Test of Nicotine Dependence [23]. Participants with a total score ≥ 6 are categorized as nicotine dependent (possible range of scores: 0-10). The test demonstrated acceptable psychometric properties [24]. In addition, participants reported on the number of cigarettes smoked per day.
Intervention attributes, previous use and preferences: These variables were measured with an adapted version of the Treatment Acceptability and Preference (TAP) measure [25]. The adaptation involved changes in terminology to make it relevant to smoking cessation. The TAP measure had four sections. Each of the first three sections presented the description of one smoking cessation intervention (as explained previously) and a set of nine items to rate the following intervention’s attributes: a) appropriateness (i.e. logic and suitability) in assisting with smoking cessation, within the context of individual lifestyle (3 items; e.g. How suitable does this treatment seem to be to help you quit smoking? How suitable does this treatment seem to your lifestyle?); b) short and long term effectiveness in helping quit smoking and dealing with craving (3 items; e.g. How effective, in the short-term, do you think this treatment will be in helping you quit smoking? How effective do you think this treatment will be in dealing with the craving for cigarettes you may experience when you quit smoking?); b) severity of side effects (1 item; How severe (bad) do you think are the risks or side effects of this treatment?); and d) convenience of implementation (i.e. ease of application and willingness to comply with treatment recommendations; 2 items; e.g. How easy do you think it will be for you to apply all activities of this treatment?). A five-point response scale, ranging from not at all (0) to very much (4) was used for each item. Higher scores reflect a favorable appraisal of the intervention relative to its appropriateness, effectiveness, and convenience, and unfavorable view of its side effects. The items measuring attributes of each intervention were internally consistent in this study, evidenced by Cronbach’s alpha coefficients > .80 (after reverse coding the item on severity of side effects). After rating each intervention, participants indicated whether or not they have used it in the past and the extent to which they found it effective, using an 11-point numeric rating scale, anchored with not at all (0) to very much (10). The last section of the TAP measure inquired about their preference for each or none of the three smoking cessation interventions.
Additional factors: To identify additional factors that smokers take into consideration when selecting an intervention, an open-ended question was posed, requesting participants to elaborate, in their own words, what made the preferred intervention appealing to them.
Data Analysis
Descriptive statistics were used to characterize the participants’ socio-demographic profile, level of nicotine dependence and perception of each intervention’s attributes. The number and percentage of participants expressing preferences for nicotine gum, brief individual advice, and group behavioral therapy were computed (aim 1). Logistic regression was applied to examine the relationship of treatment-related factors with preferences expressed for each intervention (aim 2). The predictors were entered into the regression model in two blocks. The first consisted of the four attributes (appropriateness, effectiveness, risks, convenience) and the second included previous use of the respective intervention. Perceived effectiveness of a previously used intervention was excluded as a predictor due to missing data and collinearity with the predictor reflecting prior use. Participants who reported not using an intervention previously did not rate its effectiveness, resulting in missing data and reduced sample size. Significant predictors had odds ratio (OR) > 1.0, p-value associated with the respective Wald test ≤ .05, and a rather narrow 95% confidence interval. Participants’ responses to the open-ended question were content analyzed to identify factors that smokers take into consideration when selecting a cessation intervention. Specifically, the responses were carefully read and coded; similar codes were lumped into categories reflecting non-overlapping factors; and the number of participants mentioning the same factor was counted.
Results
Characteristics of participants
As shown in Table 2, participants were middle-aged (46.8 ± 10.5, range: 25-65) white men. Most were single, unemployed, with 11-15 years of education. The mean score on the Fagerstrom Test of Nicotine Dependence was 4.7 (± 2.4 range: 0 to 10); less than half of the participants were considered nicotine dependent based on the cut-off score (≥ 6) proposed by Heatherton et al. (1991) and reported smoking 11-20 cigarettes per day [23].
![]()
Table 2: Characteristics of participants.
View Table 2
Sequence of presenting the smoking cessation treatments
The number of participants randomized to the three sequences of treatment presentation was balanced. About one third of the participants received the following sequences: 1) nicotine gum, brief individual advice, group behavioral therapy (n = 31, 34.4%); 2) brief individual advice, group behavioral therapy, nicotine gum (n = 30, 33.3%); and 3) group behavioral therapy, nicotine gum, brief individual advice (n = 29, 32.2%). There were no differences in the mean ratings of the treatment attributes across the three sequences; therefore, pertinent data were pooled across sequences and analyzed for each treatment.
Treatment-related factors
The mean ratings of the treatments’ appropriateness, effectiveness, severity of side effects, and convenience are presented in Table 3. On average, participants rated the nicotine gum as convenient to use but somewhat appropriate and effective in helping them quit smoking and deal with craving. They rated its side effects as severe. About 55.6% of participants reported having used the nicotine gum in the past and perceived it as minimally effective (2.70 ± 2.37, range: 0-8). Participants judged the brief individual advice rather unfavorably, having low levels of effectiveness, appropriateness and severity of side effects but moderate level of appropriateness. One third of the participants reported receiving such advice, which they perceived as minimally effective (2.82 ± 3.24, range: 0-10). In general, participants rated the group behavioral therapy as appropriate, moderately effective and convenient, with low severity of side effects. A small percentage (14.4%) of participants have used it in the past and perceived it as moderately effective (4.30 ± 3.01, range: 0-10).
![]()
Table 3: Mean (SD) ratings of the treatments’ acceptability.
View Table 3
Treatment preferences
The majority (87.8%) of participants stated they have a preference and 12.2% indicated they had no preference for any of the three smoking cessation interventions. The percentage of participants expressing a preference varied across the interventions: 45.6% preferred the nicotine gum, 18.9% the individual brief advice and 54.4% the group behavioral therapy.
Factors related to preferences
The results of logistic regression showed that only one treatment attribute, appropriateness, was significantly related to preferences for nicotine gum, model χ2(5) = 28.87, p < .01. Participants who rated this intervention as appropriate were 2.3 times more likely than those who did not, to prefer nicotine gum [95% confidence interval: 1.9–2.7]. None of the treatment-related factors predicted preferences for individual brief advice, model χ2(5) = 8.07, p > .05. Appropriateness was the only treatment factor that was associated with preferences for group behavioral therapy, model χ2(5) = 34.16, p < .01. Participants who rated this intervention as appropriate were 3 times more likely to choose it [95% confidence interval: 2.5 - 3.5].
Content analysis of participants’ responses to the open-ended question revealed factors that make the preferred interventions appealing. Nicotine gum appealed to participants because it: helps in dealing with craving and withdrawal (n = 15); is convenient (n = 18) as it is readily available, easy to follow, and can be used in any place, at any time; replaces the habit of smoking (n = 7); and allows the use of nicotine without having to smoke (n = 6). The group format for delivering the behavioral therapy was attractive to participants; it offered a context in which: persons give specific advice on ‘how to quit’ (n = 23) and provide support and feedback (n = 8). Being in a group facilitated ‘hearing others’ problems’ (n = 5) and promoted the feeling of ‘not being alone in the quitting process’ (n = 6).
Discussion
The study attempted to elucidate the treatment-related factors underlying adult smokers’ preferences for pharmacological and behavioral cessation therapies. Overall, the findings indicated that the majority of middle-aged, well-educated men reporting a moderate level of nicotine dependence had preferences for group behavior therapy and nicotine gum. The perceived effectiveness and convenience of use of smoking cessation interventions, and the format in which they are given were factors that influenced preferences.
The majority of participants expressed preferences, and a small number of smokers had no preference for any of the three interventions. This finding is consistent with previous research [26] and highlights the importance of assessing smokers’ preferences and accounting for these preferences in treatment selection [27]. Cumulating evidence supports the benefits of providing interventions that are congruent with the persons’ choice, in improving outcomes of medical, psycho-educational and behavioral interventions for a variety of health problems [28,29], including depression [30] and muscle pain [26].
In general, participants reported a more favorable perception and preference for the group behavioral therapy, followed by the nicotine gum, then the brief individual advice. The favorable perception of behavioral therapy for smoking cessation is comparable to the general tendency to view non-pharmacological interventions as more acceptable than pharmacological ones for the management of psychological problems such as depression [30,31] and insomnia [32]. However, the favorable perception and preference for group behavioral therapy found in this study are inconsistent with results of previous research on smoking cessation therapies. The findings of three studies that examined preferences for pharmacological and behavioral therapies suggested a preference for consultation with a physician for smoking cessation [17,18,33]. The across-study differences in findings may be related to variability in the target population (e.g. nurses, general public, smokers with moderate level of nicotine dependence), the type of interventions presented to participants (e.g. prescribed medication, over-the-counter NRT, self-help approaches), the information about the interventions given to participants (which was not clearly described in previous research) and the method used to elicit preferences (e.g. stating the treatment participants like most, choosing among alternatives). A systematic process was used to assess preferences in this study. The process involved providing smokers with information on interventions and having them appraise the interventions relative to the same set of attributes, that is, appropriateness and effectiveness in improving abstinence and dealing with craving, severity of side effects, and convenience of use, which is of relevance to persons engaged in treatment selection. Accordingly, the expressed treatment preferences are well-informed, based on a critical appraisal of the treatment attributes [34].
Although a very small number of participants have used the group behavioral therapy in the past, most rated it as appropriate for quitting smoking, moderately effective and convenient, and associated with low risk. Slightly more than half expressed a preference for this intervention. Behavioral therapies have also been found to be appealing to pregnant women [14] and Filipino men [15] contemplating smoking cessation. Two features made the group behavioral therapy attractive to smokers: its appropriateness (i.e. extent to which it was perceived as logical and suitable) and the group dynamic. Participants clarified that group dynamic facilitates exchange of information on specific strategies to help quit, peer support, and a sense of ‘not being alone’ during the quit process. Previous use did not significantly predict preference for group behavioral therapy in this study, which may be due to the unequal number of participants who did and did not report exposure to this intervention in the past. Yet, those who used it found it moderately effective in helping them quit smoking. Therefore, participants’ lack of familiarity or previous experience with group behavioral therapy may also have contributed to its attractiveness, as suggested by Cupertino et al. (2008) and McEwen and West (2009) [7,8]. Despite its demonstrated effectiveness in increasing abstinence more so than medication or NRT [35], favorable perception and desirability (as found in this study), group behavioral therapy is not widely accessible in community and primary care settings. Clinical decision makers are encouraged to undertake initiatives to make this treatment more readily available to smokers.
About half of the participants have used the nicotine gum previously but found it minimally effective in quitting smoking. On average, they rated this intervention less favorably, perceiving it as low-moderately appropriate, effective and convenient, and associated with what they judged as severe side effects. Slightly less than half of the participants expressed preferences for nicotine gum, a finding similar to the one reported by Schneider et al. (2008) [4]. The factors that contributed to the expressed preference were not consistently identified in the quantitative and qualitative data analyses. Whereas appropriateness significantly predicted preference for nicotine gum, the following features of this intervention made it appealing to smokers: its effectiveness in dealing with craving in that it provides the nicotine without exposure to the smoke; and its convenience in that it is readily available and easy to use in different contexts of daily life. The reason for this discrepancy in findings is not clear. It may be related to possible multi-collinearity in the quantitative ratings of the treatment attributes entered as predictors in the regression model. It is worth noting that participants perceived high risks associated with nicotine gum. Risk is an important factor that persons take into account in treatment-related decision making [2]. This pattern of findings also suggests that the complexity and individuality of treatment preferences may not be well captured through group-based analyses; that is, persons may differ in their perceived importance or weighing of the treatment attributes when choosing among interventions. For instance, some may value appropriateness and effectiveness more so than risk and convenience. This observation highlights the need to devise simple tools to explore the individuals’ perception and valuing of different attributes prior to assessing their preferences.
Participants rated the individual brief advice as having no risk but appraised it as low-moderately convenient and appropriate, yet minimally effective in promoting smoking cessation. Consistent with these ratings, a small number of participants preferred this intervention, which differs from previous findings showing that more than a quarter of respondents chose consultation with physicians for smoking cessation [13,15,18]. Two interrelated factors may account for the difference in findings: characteristics of this study’s participants and of the brief individual advice. Participants were middle-aged smokers with low-moderate levels of nicotine dependence. The brief advice was presented as a discussion of the importance of quitting and provision of resources, which were held for a short time during a regular visit. Therefore, participants may have perceived a lack of fit between the educational nature of the advice (as depicted in this intervention’s description) and their need for a strategy that would help them stop smoking and effectively deal with cravings. Although none of the treatment-related factors significantly predicted preferences for brief individual advice, a few participants found it appealing because it is given by a health professional who may be considered as an authoritative figure, and in an individualized way. The participants’ perspective has implications for practice. As suggested by Fiore and Baker (2011) [1], the advice may be offered to smokers with low levels of nicotine dependence, as the first component in a stepped approach to smoking cessation. However, modifications in its delivery are necessary to enhance its acceptability; such modifications may include the incorporation of some elements of motivational interviewing, which was found more effective than brief advice in increasing abstinence rate, and the allocation of more time during the visit to smoking cessation intervention. In addition, smoking cessation and strategies to address barriers to cessation should be discussed at follow-up visits. Further, training of healthcare professionals in alternative, patient-centered, ways to provide advice and in additional interventions for smoking cessation (such as motivational interviewing) should be considered.
Conclusion
In conclusion, the findings suggest that the nicotine gum (a form of NRT) and group behavioral therapy were appealing to smokers (i.e. perceived as appropriate, effective and convenient) and therefore were chosen by most participants to assist with quitting. Participants considered the treatments’ attributes when selecting treatments, as indicated by the quantitative and qualitative study findings. These were related primarily to effectiveness, convenience of use, and the format in which the treatment is delivered. The extent to which these findings are applicable to smokers with varying levels of nicotine dependence and selecting among a range of smoking cessation therapies, within the context of clinical practice, is yet to be determined. The impact of providing treatment congruent with smokers’ preferences in improving short and long term abstinence should be evaluated in future research.
Funding
The Canadian Lung Association
Declaration of Interests
None to be reported by the authors
Acknowledgement
None to be reported
Disclaimer
The views and opinions expressed in this publication are those solely of the authors and do not necessarily represent the views and opinions of Halton Region Health Department, Regional Municipality of Halton.
References
-
Fiore, MC, Jaén, CR, Baker, TB, et al. (2008) Treating tobacco use and dependence: 2008 update. Clinical Practice Guideline. Rockville, MD: US Department of Health and Human Service
-
Schneider NG, Olmstead RE, Nides M, Mody FV, Otte-Colquette P, et al. (2004) Comparative testing of 5 nicotine systems: initial use and preferences. Am J Health Behav 28: 72-86.
-
Fiore MC, Baker TB (2011) Clinical practice. Treating smokers in the health care setting. N Engl J Med 365: 1222-1231.
-
Schneider NG, Cortner C, Justice M, et al. (2008) Preferences among five nicotine treatments based on information versus sampling. Nicotine Tob Res 10: 179-186.
-
Etter J-F, Schneider NG (2013) An internet survey of use, opinions and preferences for smoking cessation medications: Nicotine, varenicline, and bupropion. Nicotine Tob Res 15: 59-68
-
Sidani S, Miranda J, Epstein DR, Bootzin RR, Cousins J, Moritz P (2009a) Relationships between personal beliefs and treatment acceptability, and preferences for behavioral treatments. Behav Res Ther 47: 823-829
-
Cupertino PA, Richter KP, Cox LS, Nazir N, Greiner AK, et al. (2008) Smoking cessation pharmacotherapy preferences in rural primary care. Nicotine Tob Res 10: 301-307.
-
McEwen A, West R (2009) Do implementation issues influence the effectiveness of medications? The case of nicotine replacement therapy and bupropion in UK Stop Smoking Services. BMC Public Health 9: 28.
-
Paterson RW, Boyle KJ, Parmeter CF, Neumann JE, De Civita P (2008) Heterogeneity in preferences for smoking cessation. Health Econ 17: 1363-1377.
-
Vogt F, Hall S, Marteau TM (2008) Understanding why smokers do not want to use nicotine dependence medications to stop smoking: Qualitative and quantitative studies. Nic Tob Res 10: 1405-1413
-
Bolliger CT, van Biljon X, Axelsson A (2007) A nicotine mouth spray for smoking cessation: a pilot study of preference, safety and efficacy. Respiration 74: 196-201.
-
Schwartz JL, Dubitzky M (1967) Expressed willingness of smokers to try 10 smoking withdrawal methods. Public Health Rep 82: 855-861.
-
Owen N (1989) Behavioral intervention studies and behavioral epidemiology research to improve smoking-cessation strategies. Health Educ Res 4: 145-153.
-
Ussher M, West R, Hibbs N (2004) A survey of pregnant smoker’s interest in different types of smoking cessation support. Patient Educ Couns 54: 67-72
-
Maxwell AE, Garcia GM, Berman BA (2007) Understanding tobacco use among Filipino American men. Nicotine Tob Res 9: 769-776.
-
Etter J-F (2011) Comparing abrupt and gradual smoking cessation: a randomized trial. Drug Alcohol Depend 118: 360-365.
-
Lawrance KG1 (2001) Adolescent smokers' preferred smoking cessation methods. Can J Public Health 92: 423-426.
-
Marques-Vidal P, Melich-Cerveira J, Paccaud F, Waeber G, Vollenweider P, Cornuz J. (2011) High expectation in non-evidence-based smoking cessation interventions among smokers - The Colaus study. Prev Med 5: 258-261.
-
Zhang B, Chaiton M (2015) Use of quit aids among Ontario smokers. The Ontario Tobacco Research Unit.
-
Corrigan PW, Salzer MS (2003) The conflict between random assignment and treatment preference: Implications for internal validity. Eval Program Plann 26: 109-121.
-
Crum RM, Anthony JC, Bassett S, Folstein MF (1993) Population-based norms for the Mini-Mental State Examination by age and educational level. J Amer Med Assoc 269: 2386-2391.
-
Norman GR, Streiner, DL (2008) Biostatistics: The bare essentials (3rd ed.). Shelton, CT: PMPH US
-
Heatherton TF, Kozlowski LT, Frecker RC, Fagerström KO (1991) The Fagerström Test for Nicotine Dependence: a revision of the Fagerström Tolerance Questionnaire. Br J Addict 86: 1119-1127.
-
Wiggers LCW, Stalmeier PFM, Oort FJ, Smets EMA, Legemate DA & de Haes JCJM (2005) Do patients’ preferences predict smoking cessation? Prev Med 41: 667-675.
-
Sidani S, Epstein DR, Bootzin RR, Moritz P, Miranda J (2009b) Assessment of preferences for treatment: validation of a measure. Res Nurs Health 32: 419-431.
-
Preference Collaborative Review Group (2009) Patients’ preferences within randomized trials: Systematic review and patient level meta-analysis. Brit Med J: a1864.
-
McClure JB, Swan GE (2006) Tailoring nicotine replacement therapy. Rationale and potential approaches. CNS Drugs 20: 281-291.
-
King M, Nazareth I, Lampe F, Bower P, Chandler M, et al. (2005) Impact of participant and physician intervention preferences on randomized trials: a systematic review. JAMA 293: 1089-1099.
-
Swift JK, Callahan JL, Vollmer BM (2011) Preferences. J Clin Psychol 67: 155-165.
-
Kwan BM, Dimidjian S, Rizvi SL (2010) Treatment preference, engagement, and clinical improvement in pharmacotherapy versus psychotherapy for depression. Behav Res Ther 48: 799-804.
-
Raue PJ, Schulberg HC, Heo M, Klimstra S, Bruce ML (2009) Patients' depression treatment preferences and initiation, adherence, and outcome: a randomized primary care study. Psychiatr Serv 60: 337-343.
-
Vincent N, Lionberg C (2001) Treatment preference and patient satisfaction in chronic insomnia. Sleep 24: 411-417.
-
Bialous SA, Sarna L, Wewers ME, Froelicher ES, Danao L (2004) Nurses' perspectives of smoking initiation, addiction, and cessation. Nurs Res 53: 387-395.
-
Bowling A, Rowe G (2005) "You decide doctor". What do patient preference arms in clinical trials really mean? J Epidemiol Community Health 59: 914-915.
-
Zernig G, Wallner R, Grohs U, Kriechbaum N, Kemmler G, Saria A (2008). A randomized trial of short psychotherapy versus sustained-release bupropion for smoking cessation. Addiction 103: 2024-2031.