International Journal of Clinical Cardiology
Favorable Plaque Sealing Process after Implantation of Endeavor Zotarolimus-Eluting Stent into Chronic Total Occlusion of Sirolimus-Eluting Stent
Minoru Ichikawa* and Yoshiyuki Kijima
Department of Cardiology, Higashi-osaka City General Hospital, Japan
*Corresponding author: Minoru Ichikawa, MD, PhD, Department of Cardiology, Higashi-osaka City General Hospital, 3-4-5 Nishi Iwata, Higashi-osaka, Osaka 578-8588, Japan, Tel: +81-6-6781-5101, Fax: +81-6-6781-2194, E-mail: michikawa419@gmail.com
Int J Clin Cardiol, IJCC-2-040, (Volume 2, Issue 4), Case Report; ISSN: 2378-2951
Received: June 26, 2015 | Accepted: July 20, 2015 | Published: July 23, 2015
Citation: Ichikawa M, Kijima Y (2015) Favorable Plaque Sealing Process after Implantation of Endeavor Zotarolimus-Eluting Stent into Chronic Total Occlusion of Sirolimus-Eluting Stent. Int J Clin Cardiol 2:040. 10.23937/2378-2951/1410040
Copyright: © 2015 Ichikawa M, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
We here report a case with in-stent chronic total occlusion (CTO) of sirolimus-eluting stent (SES). The SES-CTO lesion was successfully treated with additional implantation of Endeavor-zotarolimus-eluting stent (E-ZES) into the lesion. During the follow-up period of 50 months, coronary angioscopy and optical coherence tomography revealed the favorable plaque sealing process after E-ZES implantation into SES-CTO.
Keywords
Sirolimus-eluting stent, Zotarolimus-eluting stent, Coronary angioscopy, Chronic total occlusion
Introduction
The best treatment strategy for in-stent chronic total occlusion (CTO) after drug-eluting stent (DES) implantation has not yet been established. One option would be implantation of the newer generation DES into DES-CTO. We here report a case with in-stent CTO of the first generation DES, sirolimus-eluting stent (SES) (Cypher�, Cordis, Miami Lakes, FL, USA). He was successfully treated with implantation of another DES, an Endeavor-zotarolimus-eluting stent (E-ZES, Medtronic, Santa Rosa, CA, USA) into SES-CTO. E-ZES is characterized by more rapid release of the bio-suppressive drug and the greater late loss than other DES. Coronary angioscopy (CAS) and optical coherence tomography (OCT) revealed the favorable plaque sealing process during the follow-up period of 50 months.
Case Description
In 2004, a 69-year-old man with unstable angina underwent percutaneous coronary intervention (PCI). The target lesion was severe stenosis of the first diagonal branch of his left coronary artery. His coronary risk factors were hypertension, dyslipidemia, and cigarette smoking. The lesion was successfully treated with the first generation DES, SES of 2.5x18mm.
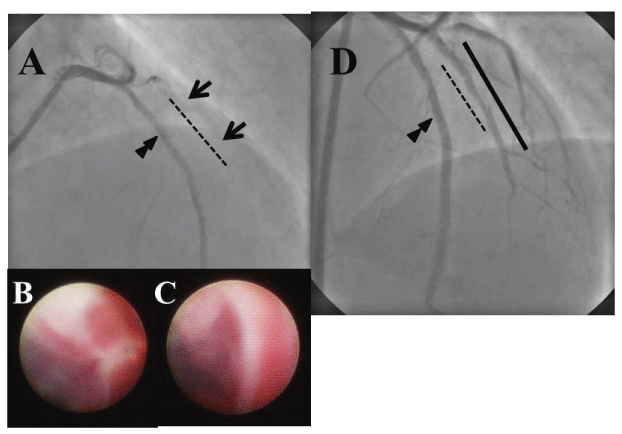
In 2010, prior to his major surgery, he was subjected to coronary angiography (CAG). It showed CTO at the stented site, indicating silent ischemic insult (Figure 1A, see dotted line). After angioplasty with a balloon of small diameter, CAS (Visible™, Fibertech, Tokyo, Japan) revealed the inner appearance of SES-CTO, characterized by massive mural thrombi and yellow atherosclerotic plaques (Figure 1B,1C). We subsequently deployed the longer E-ZES of 2.5x24mm (Figure 1D, solid line) into the shorter SES (Figure 1D, dotted line), so that the luminal side of SES was fully covered by E-ZES.
.
Figure 1: CAG and CAS at 6 years after SES deployment. CAG showed CTO at the SES-implanted diagonal branch of left coronary artery (dotted line in A). After recanalization with a balloon of small diameter, CAS revealed massive thrombi and yellow neointimal in SES (B, C). Proximal and distal arrows in A indicated the sites of angioscopic image B and C, respectively. The longer E-ZES (solid line in D) was successfully implanted into the SES-CTO. Angiograms were recorded from an anteroposterior and 30-degree cranial projection (A) and from 20-degree left anterior oblique and 30-degree cranial projection (D). Double arrowheads indicated left anterior descending artery.
View Figure 1
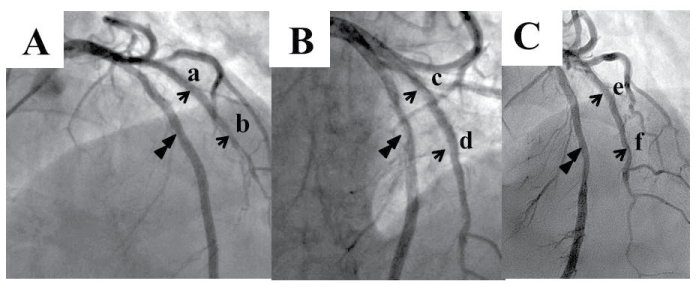
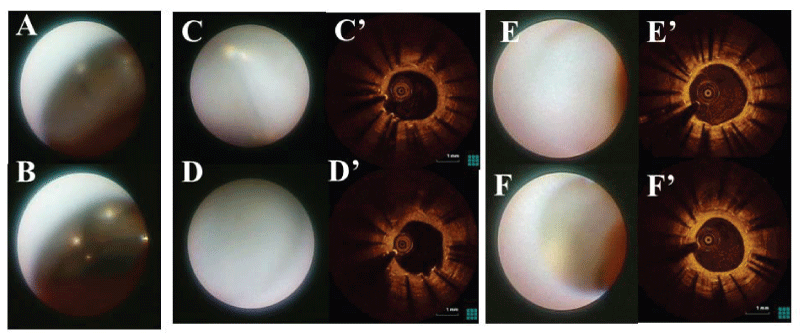
We performed follow-up CAG and intracoronary imaging (CAS and OCT) at 8 months (Figure 2A), 15 months (Figure 2B), 33 months (data not shown) and 50 months after the E-ZES deployment (Figure 2C). Dual antiplatelet therapy (DAPT) had been continued since 2004, i.e. 100mg aspirin and 75mg clopidogrel once daily. CAG showed no in-stent restenosis (Figure 2). Intracoronary imaging data at 33 months were similar to those at 50 months. CAS showed the luminal appearance of E-ZES (Figure 3). At 8 months, stent struts were covered with the thin transparent neointima (Figure 3A,3B). At 15 months, stent struts were covered with white thicker neointimal but still bulged into the lumen without glossy appearance (Figure 3C,3D). At 50 months, stent struts were fully embedded with white neointimal and not visible (Figure 3E,3F). OCT (Dragonfly Imaging Catheter, St, Jude Medical, St. Paul, MN, USA) showed the relatively thinner neointimal stent coverage at 15 months (Figure 3C',3D') and the thicker homogeneous coverage at 50 months (Figure 3E',3F'), confirming the favorable plaque sealing after E-ZES implantation.
.
Figure 2: Follow-up CAG after E-ZES deployment into SES. The patient underwent CAG at 8 month (A), 15 months (B), and 50 months (C) after implantation of E-ZES into SES. The E-ZES at the diagonal branch had been patent throughout the follow-up period. Angiograms were recorded from an anteroposterior and 30-degree cranial projection (A) and from 20-degree left anterior oblique and 30-degree cranial projection (B,C). Double arrowheads indicated left anterior descending artery. Arrows (a-f) indicated the sites of angioscopic and OCT images in figure 3.
View Figure 2
.
Figure 3: Follow-up CAS and OCT after E-ZES deployment into SES. In-stent CAS images were recorded at 8 months (A,B), 15 months (C,D), and 50 months (E,F). In-stent OCT images were recorded at 15 months (C',D') and 50 months after the E-ZES deployment (E',F'). Sites of angioscopic and OCT images were indicated by arrows in figure 2.
View Figure 3
Discussion
Up to 50 months after E-ZES implantation into SES-CTO, CAG showed patency of the treated vessel (Figure 2). CAS and OCT showed the favorable plaque sealing with white neointima (Figure 3). Previously-proposed strategies for DES-CTO were cutting balloon angioplasty [1], additional implantation of SES or everolimus-eluting stents [2], and paclitaxel-coated balloon [3]. We would propose that additional E-ZES implantation into SES-CTO was another option for SES-CTO treatment.
We previously reported with CAS that SES-CTO was characterized by massive thrombi and yellow plaque (Figure 1B, C), indicating that neoatherosclerosis progressed after SES implantation [4]. We also reported the relatively rapid healing process of E-ZES-related coronary perforation [5] and pseudoaneurysm [6]. Although a comparative study with many patients is necessary to verify our proposal, this case report suggested that E-ZES implantation into SES-CTO might lead to favorable plaque sealing with white neointimal (Figure 3). Characteristics of E-ZES are rapid release of zotarolimus and the greater late loss at chronic phase, allowing us to speculate that vascular healing process after E-ZES implantation would be similar to that after implantation of bare metal stents (BMS). Previous study with CAS reported the plaque sealing phenomenon after BMS implantation in patients with acute myocardial infarction [7]. It still remains unclear whether BMS implantation into SES-CTO would result in a similar outcome to E-ZES implantation.
In-stent neoatherosclerosis has been well characterized in vivo with CAS and OCT. The former visualizes endothelial coverage on stent struts, in-stent mural thrombi, and yellow plaque [8]. The latter quantifies in-stent plaque burden. To diagnose plague vulnerability, CAS is superior to OCT.
In conclusion, we report a case with the favorable plaque sealing of SES-CTO after additional E-ZES implantation.
References
-
Adamian M, Colombo A, Briguori C, Nishida T, Marsico F, et al. (2001) Cutting balloon angioplasty for the treatment of in-stent restenosis: a matched comparison with rotational atherectomy, additional stent implantation and balloon angioplasty. See comment in PubMed Commons below J Am Coll Cardiol 38: 672-679.
-
Song HG, Park DW, Kim YH, Ahn JM, Kim WJ, et al. (2012) Randomized trial of optimal treatment strategies for in-stent restenosis after drug-eluting stent implantation. See comment in PubMed Commons below J Am Coll Cardiol 59: 1093-1100.
-
Mamuti W, Jiamali A, Rao F, Zhang W, Pei X, et al. (2014) Drug-coated balloon angioplasty for drug-eluting stent restenosis: insight from randomized controlled trials. See comment in PubMed Commons below Ann Med 46: 679-683.
-
Ichikawa M, Kijima Y, Mishima M (2012) Repetitive angioscopic observations of a sirolimus-eluting stent deployed in a patient with silent chronic thrombotic occlusion. See comment in PubMed Commons below Cardiovasc Interv Ther 27: 201-204.
-
Ichikawa M, Kijima Y (2013) Optical coherence tomographic and angioscopic assessments of arterial healing in coronary artery perforation after implantation of zotarolimus-eluting stent. See comment in PubMed Commons below Int Heart J 54: 332-333.
-
Ichikawa M, Kijima Y (2015) Spontaneous resolution of pseudoaneurysm after zotarolimus-eluting stent implantation: imaging evidence at 13 months of follow-up. See comment in PubMed Commons below Cardiovasc Interv Ther 30: 168-170.
-
Sakai S, Mizuno K, Yokoyama S, Tanabe J, Shinada T, et al. (2003) Morphologic changes in infarct-related plaque after coronary stent placement: a serial angioscopy study. See comment in PubMed Commons below J Am Coll Cardiol 42: 1558-1565.
-
Ichikawa M, Takei Y, Hamasaki T, Kijima Y (2015) Characterization of patients with angioscopically-detected in-stent mural thrombi - Genetics of clopodogrel responsiveness and generations of drug-eluting stents. Circ J 79: 85-90.





