International Journal of Clinical Cardiology
Pioglitazone Improves Endothelial Function in Patients with Paroxysmal Atrial Fibrillation and Type 2 Diabetes Mellitus Following Catheter Ablation
Jun Gu1, Wei Hu2, Xu Liu3 and Chang-Qian Wang1*
1Department of Cardiology, Shanghai Ninth People's Hospital, Shanghai Jiaotong University School of Medicine, People's Republic of China
2Department of Cardiology, Shanghai Minhang Hospital, Fudan University, People's Republic of China
3Department of Cardiology, Shanghai Chest Hospital, Shanghai Jiaotong University, People's Republic of China
*Corresponding author: Chang-Qian Wang, Department of Cardiology, Shanghai Ninth People's Hospital, Shanghai Jiaotong University School of Medicine, No. 639 Zhizaoju Road, Shanghai, 200011, People's Republic of China, Tel: +8621 23271699, E-mail: shxkliuxu@126.com
Int J Clin Cardiol, IJCC-2-046, (Volume 2, Issue 4), Research Article; ISSN: 2378-2951
Received: July 30, 2015 | Accepted: August 26, 2015 | Published: August 30, 2015
Citation: Gu J, Hu W, Liu X, Chang-Qian W (2015) Pioglitazone Improves Endothelial Function in Patients with Paroxysmal Atrial Fibrillation and Type 2 Diabetes Mellitus Following Catheter Ablation. Int J Clin Cardiol 2:046. 10.23937/2378-2951/1410046
Copyright: © 2015 Gu J, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
In our previous study, pioglitazone, one of the peroxisome proliferator-activated receptor-gamma activators was shown to exert beneficial effects on the clinical outcome of catheter ablation in patients with paroxysmal atrial fibrillation (PAF) and type 2 diabetes mellitus (T2DM). In the current study, we aimed to investigate the influences of pioglitazone treatment on endothelial function in these patients. This study enrolled 150 patients with drug-refractory PAF and T2DM, who were grouped based on whether they received pioglitazone before ablation or not. Then 51 patients treated with pioglitazone and 99 control subjects were followed up after catheter ablation. To determine the effects of pioglitazone on endothelial function, high-frequency ultrasound was performed at baseline and after 12-month following-up. The parameters measured were brachial artery flow-mediated dilation (FMD) and nitrate mediated vasodilation (NMD). Furthermore, serum endothelial nitric oxide synthase (eNOS) level and the quantity of circulating endothelial progenitor cells (EPCs) were also determined. All baseline variables of the two groups were comparable. After 12 months of following-up, FMD improved significantly in the pioglitazone group compared to the control group as well as baseline. Besides, pioglitazone treatment also led to higher circulating EPCs number and serum eNOS level. In conclusion, pioglitazone brings about favorable effects on endothelial function in patients with PAF and T2DM after catheter ablation.
Keywords
PPARγ agonist, Atrial fibrillation, Catheter ablation, Endothelial function
Introduction
Atrial fibrillation (AF) is the most common type of sustained heart rhythm disorder confronted in clinical practice, which confers a high risk of mortality and morbidity from abnormal hemodynamics and thromboembolism [1]. The beat-to-beat alteration of flow dynamics in AF has disadvantageous effects on cardiac haemodynamics and is associated with endothelial dysfunction [2]. Endothelial dysfunction may contribute to a prothrombotic state in patients with AF and cardiovascular events [3]. Besides, the presence of diabetes mellitus (DM) augments endothelial dysfunction in AF patients [4].
Pioglitazone, one of peroxisome proliferator-activated receptor-gamma (PPARγ) agonist, is now widely used in the treatment of type 2 DM (T2DM). Recent clinical studies have shown that pioglitazone improves endothelial function [5-8] as well as ameliorates the quantity and function of endothelial progenitor cells (EPCs) [9,10]. Our previous study indicated that pioglitazone ameliorated the preservation of sinus rhythm (SR) and decreased the reablation rate in patients with paroxysmal AF (PAF) and T2DM after catheter ablation [11]. For those enrolled patients, we further determined the effects of pioglitazone use on the endothelial function as well as the magnitude of EPCs mobilization during pre- and post-ablation period.
Methods
Study design and study populations
The methodology for this clinical trial was previously described in detail [11]. From September 2007 to July 2009, a total of 150 consecutive patients with drug-refractory PAF and T2DM were enrolled in this prospective observational single-centre cohort study. The study protocol was approved by the local ethics committee and informed consent was obtained from all patients. Major exclusion criteria were age >75 years old, unstable angina, acute myocardial infarction, congestive heart failure, pregnancy or severe renal or hepatic dysfunction. Patients were grouped based on whether they received pioglitazone therapy before ablation or not: 51(34%) patients received pioglitazone preablation were in the pioglitazone group and pioglitazone was continued post-ablation (30 mg/day), while 99(66%) patients without previous pioglitazone therapy formed the control group, in which patients taking standard therapy except pioglitazone during this study. All enrolled patients were kept free from newly prescribed statin, and angiotensin converting enzyme inhibitor/angiotensin II receptor blocker (ACEI/ARB) during the follow-up.
Mapping and catheter ablation procedure
Electrophysiological study and catheter ablation procedure were performed as we previously described [11]. Briefly, all antiarrhythmic drugs (AADs) except amiodarone were discontinued for at least five half-lives. Oral anticoagulation was ceased 3 days pre-procedure, and full-dose weight-adjusted (1.5 mg/kg daily) subcutaneous low-molecular-weight heparin was used to bridge the procedure until therapeutic oral anticoagulation was achieved following ablation. The presence of atrial thrombus was excluded by transoesophageal echocardiography before ablation. During the procedure, intravenous unfractionated heparin was administered to maintain an activated clotting time between 300 and 350 sec. Circumferential pulmonary vein isolation (CPVI) was performed at the posterior wall 1 cm and at the anterior wall 5 mm away from the angiographically defined PV ostia under the guidance of the CARTO system (Biosense Webster). The endpoint of circumferential PV ablation was PV isolation, which was defined as disappearance of all PV potentials or left atrial (LA)-PV potential dissociation. After the CPVI, if AF was sustained or induced with coronary sinus burst pacing at a cycle length down to 180 ms during the administration of intravenous isoproterenol (1.0 to 3.0 mg/min) and lasted more than 3 min, additional ablation, including linear ablation of the left atrial roof and/or superior vena cava isolation, was conducted. The cavotricuspid isthmus was ablated in patients with a history of atrial flutter (AFL). If the AF did not terminate or was inducible after aforementioned procedures, SR was restored by transthoracic cardioversion. Patients were administered oral anticoagulation (warfarin) (international normalized ratio: 2.0-3.0) for 3 months. AAD was administered in all cases for 1 month after the ablation and was withdrawn 1-month later in cases without AF recurrence, but was continued otherwise.
Following-up
Patients were discharged in SR after 2 days of monitoring. After discharge, all patients underwent following-up at 2 weeks post-procedure and then every month thereafter. At each hospital visit, the patients underwent 12-lead ECG and intensive questioning regarding any arrhythmia-related symptoms. Holter monitoring for 24 h was performed at 2 weeks and 1, 3, 6, 9, and 12 months. An open access to arrhythmia specialists was available subsequently. All patients enrolled in our study received both periodical ECG/Holter recording and ECG/Holter monitoring prompted by symptoms. As early recurrences often settle spontaneously, a 1-month blanking period was observed, during which recurrences were managed medically. After the 1-month blanking period, any episode of symptomatic or asymptomatic atrial tachyarrhythmias [ATa, including AF, atrial tachycardia (AT) and AFL] with ECG and Holter recording that lasted over 30 s was considered as a recurrent event [11].
Endothelial function study
Enrolled patients were asked to fast and discontinued vasoactive medications for 12 h prior to the study. In brief, the patients were instructed to lie quietly in a supine position for 10 min before the testing. All testings were carried out in a temperature-controlled room (20 to 25°). Arterial endothelial and smooth muscle function were evaluated by examining brachial artery responses to endothelium-dependent (flow-mediated) and endothelium-independent [glyceryl trinitrate (GTN)-mediated] stimuli. All images were conducted using a hand-held 10 mHz vascular ultrasound probe (GE Vingmed System 5 ultrasound machine) as previously described [12]. Endothelium-dependent, post ischemic flow mediated vasodilation (FMD) was determined by the maximal brachial artery diameter after 60 sec of reactive hyperemia, compared with baseline vessel diameter. Endothelium-independent, nitrate mediated vasodilation (NMD) was expressed as the percent change in baseline vessel diameter 3 min after sublingual nitrate administration (0.3 mg glyceryl trinitrate) [12].
Enzyme-Linked Immunosorbent Assay (ELISA) for endothelial nitric oxide synthase (eNOS)
Peripheral blood samples were obtained at baseline at the start of the procedure and at 12 months post-procedure during outpatient following-up. Laboratory personnel who conducted the testing were blinded to patient characteristics. The acquired blood samples were centrifuged at 2500g for 15 min at 4° and stored at -80° for batch ELISA analysis. Serum eNOS level was measured in accordance to company's instructions (R&D Systems, Minneapolis, MN, USA).
Ciuculating EPCs number
Ciuculating EPCs were quantified by phenotype in whole blood samples utilizing flow cytometry [13]. The cells were analyzed for phenotypic expression of surface antigens using monoclonal antibodies. The EPCs were defined as CD34+/CD133+/KDR+ cells. EDTA anticoagulant peripheral whole blood (100 μL) was stained with antibodies [anti-CD34-FITC (BioLegend, San Diego, CA, USA), anti-CD133-APC (Miltenyl Biotech, Germany) and anti-KDR (VEGFR2)-PE (Sigma, St Louis, MO, USA)] for 20 minutes in dark chamber at room temperature. Appropriate isotype controls were stained to establish positive stain boundaries. Erythrocytes were lysed (Becton Dickinson, Oxford, UK), and samples were centrifuged at 1500 g for 5 minutes at room temperature. Acquisition was performed by technicians blinded to subject identity using a FACS Valieur analyzer (Becton Dickison). Analysis was conducted by gating lymphocytes and monocytes on the basis of light-scattering properties. We acquired 100 000 events per gate. The quantity of EPCs was measured based on the percentage of CD34+CD133+KDR+ triple positive leucocytes and expressed as the number of cells per 105 mononuclear cells.
Statistical analysis
All continuous variables are expressed as mean ± SEM and categorical variables as proportions. Between-group comparisons were performed using the t-test or χ2 test as appropriate. A P-value of < 0.05 was considered statistically significant. Statistical analysis was performed using SPSS software (version 18; SPSS Inc, Chicago, IL).
Results
Baseline measurements and outcome after primary ablation
The baseline characteristics of the study population are shown in table 1. All baseline variables of the two groups were statistically similar. A total of 150 patients with PAF and T2DM were enrolled in this study. After 12 months of following-up, 46 patients (90.2%, maintaining SR subgroup) from pioglitazone group and 74 patients (74.7%, maintaining SR subgroup) from the control group had no ATa recurrence without AADs therapy (P = 0.025) as we previous described [11].
Changes in endothelial function after pioglitazone treatment
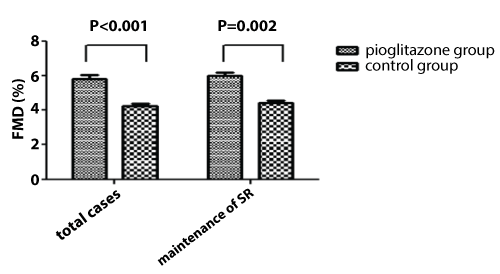
FMD and NMD were not statistically significant between the two groups at baseline (Table 1). At 12 months after catheter ablation, FMD was markedly increased in the pioglitazone group compared with control group (Table 2, Figure 1). Further subgroup analysis showed that in patients with successful maintaining SR following catheter ablation, pioglitazone therapy also gave rise to better FMD compared with control group (Table 2, Figure 1).
.
Figure 1: Comparison of FMD between the pioglitazone group and control group during following-up
View Figure 1
![]()
Table 1: Baseline patient characteristics
View Table 1
![]()
Table 2: Endothelial function after pioglitazone treatment
View Table 2
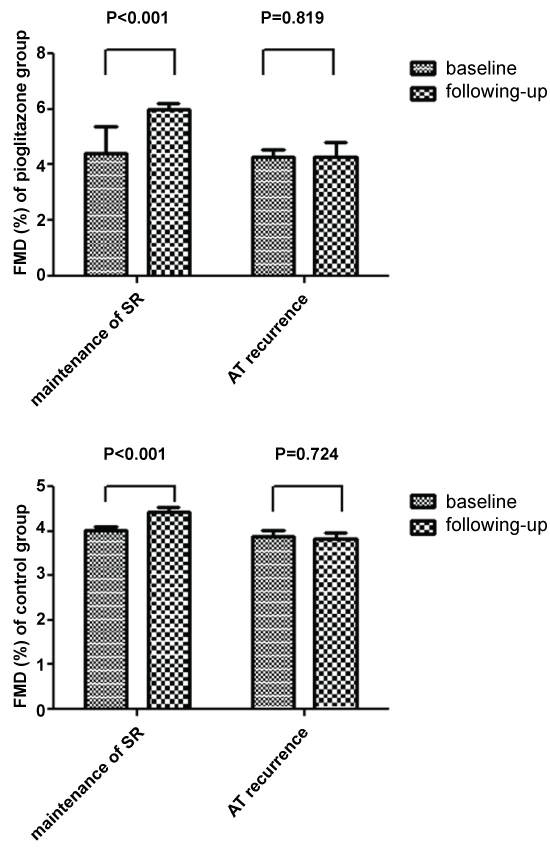
Moreover, catheter ablation with successful maintaining SR rather than ATa recurrence led to obvious higher FMD compared with baseline in both pioglitazone and control group (Table 3, Figure 2). However, NMD did not differ between the two groups during following-up (22.4 ± 0.7% vs 23.0 ± 0.4%, P = 0.429).
![]()
Table 3: Endothelial function between baseline and following-up
View Table 3
Serum eNOS level after pioglitazone treatment
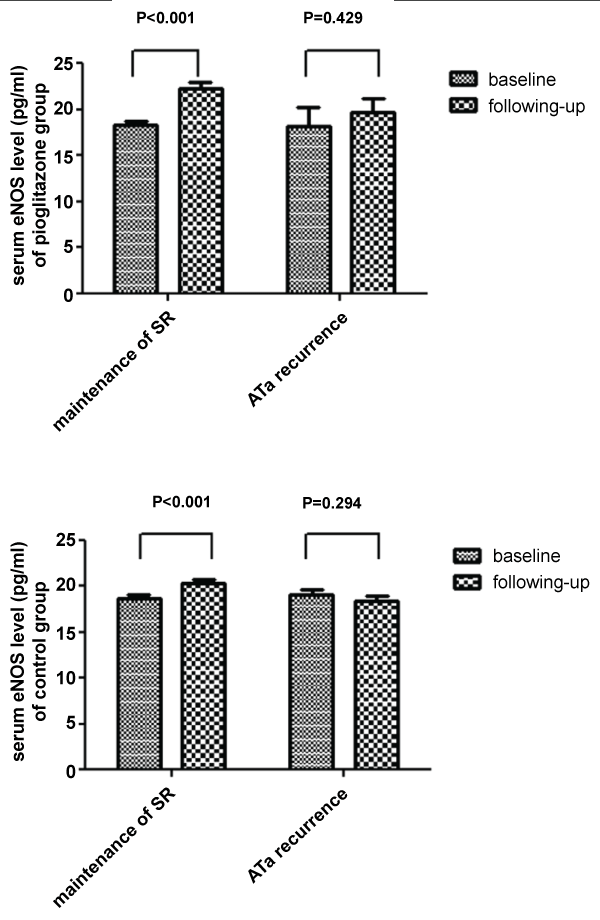
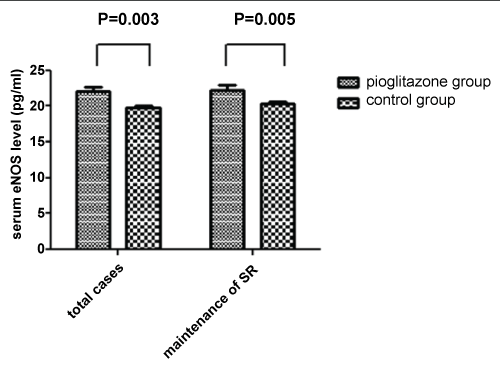
There was no marked difference in endothelial function marker eNOS at baseline between the two groups (Table 1). However, serum eNOS concentration was markedly higher in the pioglitazone group compared with the control group during the following-up (Table 2, Figure 3). Further subgroup analysis indicated that in patients with successful maintaining SR following ablation, pioglitazone therapy also resulted into increased serum eNOS level compared with the control group (Table 2, Figure 3). Besides, compared with baseline, serum eNOS level was obviously higher in patients with successful maintaining SR instead of ATa recurrence following catheter ablation in both pioglitazone and control group (Table 3, Figure 4).
.
Figure 3: Comparison of eNOS between the pioglitazone group and control group during following-up
View Figure 3
Effects of the pioglitazone treatment on circulating EPCs number
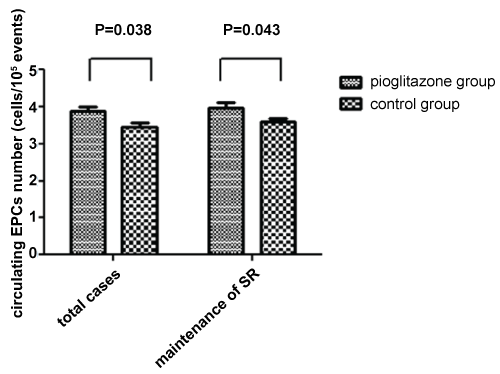
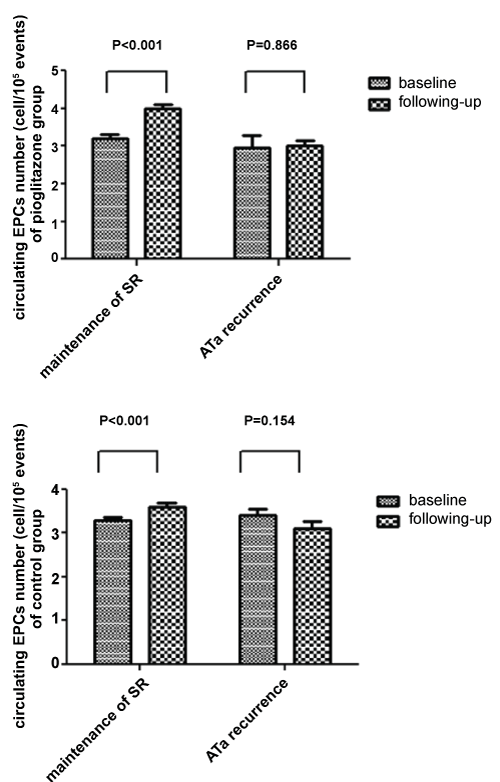
The number of circulating EPCs (CD133+CD34+KDR+) between the two groups was not significantly different before the start of therapy (Table 1). Pioglitazone treatment significantly increased the quantity of circulating EPCs compared with control group (Table 2, Figure 5). Further subgroup analysis showed that in patients with successful maintaining SR after ablation, pioglitazone therapy also brought about higher circulating EPCs number compared with control group (Table 2, Figure 5). Moreover, in both pioglitazone and control group, circulating EPCs level at 12-month following-up was significantly augmented compared with baseline level in patients with successful maintaining SR, but not with ATa recurrence following ablation (Table 3, Figure 6).
.
Figure 5: Comparison of ciuculating EPCs number between the pioglitazone group and control group during following-up
View Figure 5
.
Figure 6: Comparison of ciuculating EPCs number between baseline and following-up in both groups
View Figure 6
Discussion
The most notable findings of this study in patients with PAF and T2DM after catheter ablation were: (a) Pioglitazone treatment improved FMD; (b) Pioglitazone therapy led to a higher eNOS level; (c) Pioglitazone treatment augmented circulating EPCs number.
Accumulating evidences have indicated that AF patients have impaired endothelial function [2-4]. It is shown that patients with AF have diminished endothelial-dependent FMD and that this reduction is reversed by restoring SR through electrical cardioversion [14,15] as well as catheter ablation [16,17]. EPCs, as a specific group of cells generated from bone marrow, are suggested to play a key role in maintaining vascular integrity and in injury repair. They are thought to contribute to reendothelialization, neovascularization, and normalization of endothelial function and restoration of blood flow at the site of injury [18]. It also appears to be widely accepted that eNOS with normal function maintains vascular homeostasis by producing nitric oxide (NO). In the present study, our results indicated that FMD, ciuculating EPCs as well as serum eNOS level were ameliorated in patients with PAF and T2DM after successful catheter ablation. Several mechanisms may be responsible for AF induced endothelial dysfunction. First of all, laminar shear stress plays a key role in the process of eNOS activation in normal physiology, the beat-to-beat variation of AF induces a turbulent flow that may weaken arterial vessel distension and responsiveness. Secondly, disorganized atrial contraction induced by AF, markedly reduces eNOS expression and nitric oxide (NO) production in the atrial endocardium, which leads to endothelial dysfunction [19]. Finally, AF induces atrial inflammatory and oxidative stress that may generate a systemic noxious effect on endothelial cells. CRP, a biomarker of inflammation, directly inhibits endothelial cell NO production via destabilizing eNOS [20]. And ST-elevation myocardial infarction (STEMI) patient with a high C-reactive protein (CRP) peak value has compromised ability to stimulate eNOS-dependent NO production [21]. CRP may also serve to impair EPCs function and promote EPCs apoptosis through receptor for advanced glycation end products (RAGE) [22].
DM has been reported as one of the main risk factors for AF. Previous study identified that DM led to greater atrial arrhythmogenicity, longer intra-atrial activation time, and increased atrial fibrotic deposition in the animal model, which could be a major substrate for diabetes-related AF [23]. And patients with DM or an abnormal glucose metabolism had a lower atrial voltage and longer atrial activation time compared to those with a normal glucose metabolism [24]. Furthermore, endothelial dysfunction is greater in diabetes with AF [4]. There is a growing body of evidence elucidating the mechanisms underlying the development of endothelial dysfunction in T2DM. T2DM induced biochemical disturbances (dyslipoproteinemia, oxidative stress and inflammation) uncouple mitochondrial function in endothelial cells, generating reactive oxygen species (ROS) by increasing advanced glycation endproducts (AGEs), protein kinase C (PKC) and Nacetylglucosamine (glcNAC), impairing eNOS activity and inducing endothelial dysfunction [25].
There is increasing evidence that PPARγ activation plays an important role in the regulation of the vascular endothelial function. Both troglitazone and pioglitazone increased brachial artery FMD in T2DM patients without macrovascular disease [26-28]. In line with previous study, we also found that pioglitazone improved the FMD in patients with PAF and T2DM in the present study. Besides, our results indicated that pioglitazone treatment could increase the quantity of circulating EPCs after catheter ablation. And a reduced quantity and function of EPCs have been related to endothelial dysfunction as well as increased risk cardiovascular morbidity and mortality [18]. Several studies indicates that AF as well as DM is associated with impaired quantity and function of EPCs [29,30]. Likewise, pioglitazone was also shown to increase the circulating number of EPCs in patients with coronary artery disease [9] as well as exert beneficial effects in vitro function of EPCs isolated from impared glucose tolerance (IGT) subjects [10].
Regarding the favorable effect of pioglitazone on the FMD and circulating EPCs number in patient with PAF and T2DM after catheter ablation, its up-regulating effect on eNOS is supposed to be mentioned. In the present study, serum eNOS level was obviously increased with pioglitazone treatment. Recent studies have also suggested a potential regulatory role of PPARγ agonists on eNOS expression or activation in the vascular endothelium or EPCs [6,31,32]. PPARγ activation by rosiglitazone stimulation attenuated the apoptosis and dysfunction of EPCs induced by AGEs via up-regulation of protein kinase B (PKB) and eNOS signaling [31]. Likewise, Pioglitazone induced PPARγ activation up-regulated PKB and eNOS phosphorylation, resulting in the amelioration of vascular endothelial dysfunction and enhancement of blood flow recovery after tissue ischaemia in the diabetic mouse [32]. And pioglitazone abrogated the hypertensive effect of cyclosporine (CSA) via ameliorating detrimental changes in vascular eNOS/NO pathway caused by CSA [6]. Further study indicated that PPARγ-mediated activation of eNOS might involve HSP90 and adiponectin signalling mechanism [7].
It was reported that successful reversion of AF and maintenance of SR by catheter ablation or electrical cardioversion would lead to a decrease endothelial dysfunction [14-17]. So more cases with successful maintenance of SR following ablation might result in better endothelial function in the pioglitazone group of the present study. Accordingly, we further analysed the difference of endothelial function in patients with successful maintaining SR between the two groups. And the result also showed that pioglitazone markedly improved the endothelial function compared with the control group, which suggested that pioglitazone might possess the ability to enhance the beneficial effect of successful catheter ablation on endothelial function.
In the present study, patient characteristics (Table 1) show that there is tendency towards lower hsCRP levels in the pioglitazone group. And pioglitazone treatment for a period of time before ablation in these patients might account for the discrepant tendency of hsCRP. Likewise, another study also demonstrated that pioglitazone attenuated inflammation in patients with IGT or T2DM in a glucose-lowering-independent manner [33].
Study Limitation
At first, the following-up of 12 months might be too short and a longer period might be necessary to show the effects of the drugs on endothelial function. Secondly, the effect of various drugs (ACEI/ARB and statins) on endothelial function could not be fully controlled in the present study, as all subjects were not on the same drug therapy regimen. However, there were no marked differences in baseline medication regimens between the 2 groups. Thirdly, due to the limited quantity and nonrandomization of our study, a much larger, randomized and multi-centred trial is required to testify our conclusions.
Conclusions
In summary, our data indicates that pioglitazone improves endothelial function in patients with PAF and T2DM after catheter ablation, and our findings may also provide a potential rationale for clinical PPARγ agonists treatment in some particular conditions where the improvement of endothelial function should be taken into account.
Acknowledgment
This work was supported by grants from Shanghai city key medical specialties (ZK2012A24), special fund of atiral fibrillation from Chinese Medical Doctor Association (2013-2-12) and project of Shanghai Science and Technology Commission (14411972900).
References
-
Wattigney WA, Mensah GA, Croft JB (2003) Increasing trends in hospitalization for atrial fibrillation in the United States, 1985 through 1999: implications for primary prevention. Circulation 108: 711-716.
-
Watson T, Shantsila E, Lip GY (2009) Mechanisms of thrombogenesis in atrial fibrillation: Virchow's triad revisited. Lancet 373: 155-166.
-
Guazzi M, Arena R (2009) Endothelial dysfunction and pathophysiological correlates in atrial fibrillation. Heart 95: 102-106.
-
Varughese G, Patel JV, Tomson J, Lip GY (2005) The prothrombotic risk of diabetes mellitus in atrial fibrillation and heart failure. J Thromb Haemost 3: 2811-2813.
-
Marder W, Khalatbari S, Myles JD, Hench R, Lustig S, et al. (2013) The peroxisome proliferator activated receptor-γ pioglitazone improves vascular function and decreases disease activity in patients with rheumatoid arthritis. J Am Heart Assoc 2: e000441
-
El-Mas MM, El-Gowelli HM, Abd-Elrahman KS, Saad EI, Abdel-Galil AG, et al. (2011) Pioglitazone abrogates cyclosporine-evoked hypertension via rectifying abnormalities in vascular endothelial function. Biochem Pharmacol 81: 526-533.
-
Balakumar P, Kathuria S (2012) Submaximal PPARγ activation and endothelial dysfunction: new perspectives for the management of cardiovascular disorders. Br J Pharmacol 166: 1981-1992.
-
Nishio K, Sakurai M, Kusuyama T, Shigemitsu M, Fukui T, et al. (2006) A randomized comparison of pioglitazone to inhibit restenosis after coronary stenting in patients with type 2 diabetes. Diabetes Care 29: 101-106.
-
Werner C, Kamani CH, Gensch C, Böhm M, Laufs U (2007) The peroxisome proliferator-activated receptor-gamma agonist pioglitazone increases number and function of endothelial progenitor cells in patients with coronary artery disease and normal glucose tolerance. Diabetes 56: 2609-2615.
-
Spigoni V, Picconi A, Cito M, Ridolfi V, Bonomini S, et al. (2012) Pioglitazone improves in vitro viability and function of endothelial progenitor cells from individuals with impaired glucose tolerance. PLoS One 7: e48283.
-
Gu J, Liu X, Wang X, Shi H, Tan H, et al. (2011) Beneficial effect of pioglitazone on the outcome of catheter ablation in patients with paroxysmal atrial fibrillation and type 2 diabetes mellitus. Europace 13: 1256-1261.
-
Kaya H, Ozkan M, Yildiz M (2013) Relationship between endothelial dysfunction and prosthetic heart valve thrombosis: a preliminary investigation. Eur Rev Med Pharmacol Sci 17: 1594-1598.
-
Duan C, Du ZD, Wang Y, Jia LQ (2014) Effect of pravastatin on endothelial dysfunction in children with medium to giant coronary aneurysms due to Kawasaki disease. World J Pediatr 10: 232-237.
-
Skalidis EI, Zacharis EA, Tsetis DK, Pagonidis K, Chlouverakis G, et al. (2007) Endothelial cell function during atrial fibrillation and after restoration of sinus rhythm. Am J Cardiol 99: 1258-1262.
-
Guazzi M, Belletti S, Lenatti L, Bianco E, Guazzi MD (2007) Effects of cardioversion of atrial fibrillation on endothelial function in hypertension or diabetes. Eur J Clin Invest 37: 26-34.
-
Lim HS, Willoughby SR, Schultz C, Chakrabarty A, Alasady M, et al. (2014) Successful catheter ablation decreases platelet activation and improves endothelial function in patients with atrial fibrillation. Heart Rhythm 11: 1912-1918.
-
Shin SY, Na JO, Lim HE, Choi CU, Choi JI, et al. (2011) Improved endothelial function in patients with atrial fibrillation through maintenance of sinus rhythm by successful catheter ablation. J Cardiovasc Electrophysiol 22: 376-382.
-
Lee PS, Poh KK (2014) Endothelial progenitor cells in cardiovascular diseases. World J Stem Cells 6: 355-366.
-
Cai H, Li Z, Goette A, Mera F, Honeycutt C, et al. (2002) Downregulation of endocardial nitric oxide synthase expression and nitric oxide production in atrial fibrillation. Potential mechanisms for atrial thrombosis and stroke. Circulation 106: 2854-2858.
-
Fordjour PA, Wang Y, Shi Y, Agyemang K, Akinyi M, et al. (2015) Possible mechanisms of C-reactive protein mediated acute myocardial infarction. Eur J Pharmacol 760: 72-80.
-
Gomaraschi M, Ossoli A, Favari E, Adorni MP, Sinagra G, et al. (2013) Inflammation impairs eNOS activation by HDL in patients with acute coronary syndrome. Cardiovasc Res 100: 36-43.
-
Chen J, Jin J, Song M, Dong H, Zhao G, et al. (2012) C-reactive protein down-regulates endothelial nitric oxide synthase expression and promotes apoptosis in endothelial progenitor cells through receptor for advanced glycation end-products. Gene 496: 128-135.
-
Kato T, Yamashita T, Sekiguchi A, Sagara K, Takamura M, et al. (2006) What are arrhythmogenic substrates in diabetic rat atria? J Cardiovasc Electrophysiol 17: 890-894.
-
Chao TF, Suenari K, Chang SL, Lin YJ, Lo LW, et al. (2010) Atrial substrate properties and outcome of catheter ablation in patients with paroxysmal atrial fibrillation associated with diabetes mellitus or impaired fasting glucose. Am J Cardiol 106: 1615-1620.
-
Hamilton SJ, Watts GF (2013) Endothelial dysfunction in diabetes: pathogenesis, significance, and treatment. Rev Diabet Stud 10: 133-156.
-
Caballero AE, Saouaf R, Lim SC, Hamdy O, Abou-Elenin K, et al. (2003) The effects of troglitazone, an insulin-sensitizing agent, on the endothelial function in early and late type 2 diabetes. A placebo-controlled randomized clinical trial. Metabolism 52: 173-180.
-
Suzuki M, Takamisawa I, Yoshimasa Y, Harano Y (2007) Association between insulin resistance and endothelial dysfunction in type 2 diabetes and the effects of pioglitazone. Diabetes Res Clin Pract 76: 12-17.
-
Martens FM, Visseren FL, de Koning EJ, Rabelink TJ (2005) Short-term pioglitazone treatment improves vascular function irrespective of metabolic changes in patients with type 2 diabetes. J Cardiovasc Pharmacol 46: 773-778.
-
Siu CW, Watson T, Lai WH, Lee YK, Chan YH, et al. (2010) Relationship of circulating endothelial progenitor cells to the recurrence of atrial fibrillation after successful conversion and maintenance of sinus rhythm. Europace 12: 517-521.
-
Egan CG, Lavery R, Caporali F, Fondelli C, Laghi-Pasini F, et al. (2008) Generalised reduction of putative endothelial progenitors and CXCR4-positive peripheral blood cells in type 2 diabetes. Diabetologia 51: 1296-1305.
-
Liang C, Ren Y, Tan H, He Z, Jiang Q, et al. (2009) Rosiglitazone via upregulation of Akt/eNOS pathways attenuates dysfunction of endothelial progenitor cells, induced by advanced glycation end products. Br J Pharmacol 158: 865-873.
-
Huang PH, Sata M, Nishimatsu H, Sumi M, Hirata Y, et al. (2008) Pioglitazone ameliorates endothelial dysfunction and restores ischemia-induced angiogenesis in diabetic mice. Biomed Pharmacother 62: 46-52.
-
Nitta Y, Tahara N, Tahara A, Honda A, Kodama N, et al. (2013) Pioglitazone decreases coronary artery inflammation in impaired glucose tolerance and diabetes mellitus: evaluation by FDG-PET/CT imaging. JACC Cardiovasc Imaging 6: 1172-1182.