International Journal of Clinical Cardiology
Ultrafiltration in Heart Failure: Experience in a French University Hospital
David Boutarin* Raphaelle -Ashley Guerbaai and Patricia Armand
Department of Cardiology, Centre Hospitalier Universitaire Grenoble, France
*Corresponding author: David Boutarin, Department of Cardiology, Centre Hospitalier Universitaire de Grenoble, BP 217, 38043 Grenoble Cedex 09, Tel: +33 476768466, Fax: +33 476765368, E-mail: dsboutarin@gmail.com
Int J Clin Cardiol, IJCC-2-054, (Volume 2, Issue 5), Perspective; ISSN: 2378-2951
Received: July 30, 2015 | Accepted: September 21, 2015 | Published: September 24, 2015
Citation: Boutarin D, Raphaelle-Ashley G, Armand P (2015) Ultrafiltration in Heart Failure: Experience in a French University Hospital. Int J Clin Cardiol 2:054. 10.23937/2378-2951/1410054
Copyright: © 2015 Boutarin D, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Keywords
Chronic heart failure, Ultrafiltration
Introduction
Venovenous ultrafiltration is an alternative therapy used in patients with chronic heart failure (CHF), persistent congestion and failing diuretics [1]. This therapy enables removal of excess water in order to reduce fluid overload [1]. Ultrafiltration has been used for over ten years in the United-States, however was only introduced in France three years ago, in very few hospitals [2]. Until recently, hemofiltration was only possible in high dependency resuscitation units with the use of hemodialysis machines, thus not available in cardiac intensive care. Ultrafiltration uses only 40 ml/hr of blood volume against 300 ml/hr with hemodialysis. Our center was among the first, to offer ultrafiltration to patients with end-stage heart failure, in the cardiac intensive care, to ensure very close monitoring and prevent adverse effects.
Background
Patients suffering from chronic congestive heart failure are often re-admitted to the Cardiac Intensive Care Unit (CICU) with worsening peripheral and acute pulmonary edema, and become resistant to intravenous loop diuretics [3]. Ultrafiltration has been used as an alternative therapy in order to improve clinical symptoms, relieve congestion and limit readmission [3]. Unlike diuretics, this therapy avoids electrolyte depletion [1-3]. Also, commercialization of CICU machines solely for the purpose of ultrafiltration has facilitated both training and implementation of this therapy in our unit [4].
Ultrafiltration Mechanism
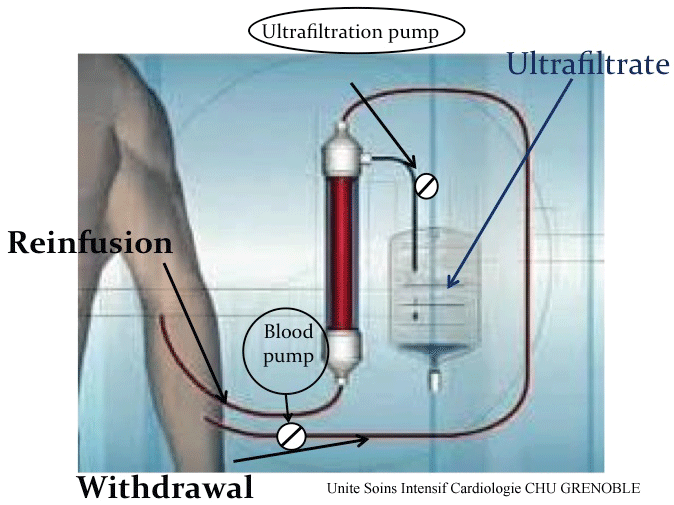
The ultrafiltration pump and kit are assembled by the nursing team, using a sterile field. Once the patient is connected to the machine, the initial ultrafiltration rate is set at 250 ml/hr according to our protocol. This is an advantage compared with conventional IV diuretics as the amount of ultrafiltrate can be closely monitored and recorded (Figure 1).
Once blood is drawn from the patient, plasma is filtered by a pump using negative hydrostatic pressure, to enable isotonic fluid removal. Ultrafiltration does not create electrolyte imbalances, an advantage compared with IV diuretics. Nonetheless, ultrafiltration is not a substitute for dialysis as it will not permit removal of toxins.
Patient Selection
Ultrafiltration is used in our institution for patients suffering from CHF, becoming resistant to diuretics and multiplying hospital admissions.
Patients with impaired renal function, with creatinine levels above 300 mmol/L, and/or contraindicated to anticoagulants are not eligible for treatment. Better results are obtained in patients who are NYHA 3 than in patients awaiting a heart transplant however our unit has provided ultrafiltration for 2 patients awaiting a heart transplant. These patients had become resistant to both diuretics and dobutamine, thus regular ultrafiltration sessions enabled congestion relief.
Anticoagulation
Venovenous ultrafiltration requires strict patient anticoagulation and monitoring as the principal machine mechanism resides in the filter composed of a semi-permeable membrane, which allows suction of excess fluid and salt in the patient's blood stream [4]. In order for successful treatment and to avoid premature filter thrombosis, activated partial thromboplastin time (APTT) for patients eligible for ultrafiltration has to be between 90 and 110 before treatment initiation and a heparin bolus is administrated before connecting the patient to the machine. Patients treated with Vitamin K antagonist therapy (VKA), should have an international normalized ratio (INR) between 2 and 3 and an APTT between 70 and 90. For all patients undergoing treatment, four to six hourly blood tests are performed to ensure adequate anticoagulation and prevent hemorrhagic risk.
Adverse Events
To avoid acute renal failure, diuretics and angiotensin converting enzyme (ACE) inhibitors are stopped during treatment and patients are closely monitored. Adverse events can include hypotension (due to excessive water depletion), therefore controlling and adapting the rate of ultrafiltration is extremely important as each patient mobilizes excess fluid from the interstitial spaces into the bloodstream differently (plasma refill rate). Usually between 10 and 40 ml/hr, the blood flow rate can be rapidly and easily changed, if modifications in blood pressure, creatinine or hematocrit levels are noted.
Three cases of acute renal failure have occurred on our unit since the implementation of ultrafiltration, due to excess fluid removal. Since, a fluid removal objective is systematically prescribed and once reached, ultrafiltration is stopped and the machine is removed. Monitoring the hematocrit level (with the use of the hematocrit monitor provided with the machine) is important as the level will increase when plasma volume is reduced. If hemoconcentration occurs, ultrafiltration can be suspended, allowing for interstitial edema to replenish the intravascular space before continuing ultrafiltration. No additional cases of renal failure have occurred since implementation of a precise fluid removal objective for each patient, in our unit.
Venous Access
Venous access is an important factor for the success of ultrafiltration. Use of four-lumen central venous lines is the standard in our center; the 14 gauge lumen is used for withdrawal and the 16 gauge lumen, for reinfusion. However, close monitoring of central venous lines is important to prevent complications related to infection. To prevent this, nursing staff use a sterile field when manipulating the central line and ensure twice a day temperature monitoring. Patients have daily blood tests to exclude infection.
Experience and Results
In our CICU, 12patients have been treated with ultrafiltration, from July 2012 until October 2014, with a total number of 21 sessions (3 patients received more than one session), with an average of 42 hours treatment and 7 liters of fluid loss per patient. 24% of sessions had to be suspended before the fluid removal objective was reached, due to filter thrombosis as anticoagulation was performed on a one-to-one basis. Since strict adherence to our anticoagulation protocol, no kit thrombosis has occurred, and ten sessions have been successful, since October 2013.
Patients experience clinical improvement during treatment, and can monitor the decrease in peripheral edema. They also report improvement in dyspnea and once discharged are able to regain independence at home. Although the majority of patients are satisfied, it is important to note that during the course of treatment (up to 72 hours), complete bed rest is recommended, which can be difficult for some patients. Limited mobility can also become an issue, especially for elderly patients. Despite medical and nursing staff training, implementing ultrafiltration in CICU, is challenging as time and staff has to be set aside in order to set up and monitor patients closely [5]. However, much like other ICU equipment, the ultrafiltration machine self-monitors, and alerts staff with an alarm. A list of potential problems and step-by-step solutions are then offered, to enable efficient troubleshooting and ensure completion of treatment.
Scope for the Future
Although this treatment has benefited patients in our center, controversies have been raised regarding ultrafiltration. The UNLOAD trial showed greater benefit in terms of fluid removal using ultrafiltration versus intravenous (IV) diuretics [3], however Bart et al. [1] reported more adverse events in the ultrafiltration group and no greater benefit versus IV diuretics. Implementing ultrafiltration in our day care ward, using a peripheral venous catheter, for patients presenting with CHF at an earlier stage could be of greater benefit and has yet to be implemented. Patients suffering from heart failure are closely followed by the RESIC 38 (Reseau des insuffisants cardiaques de lisere), which enables a link between primary care facilities and our university hospital to ensure patients can access timely treatment at an earlier stage and avoid long hospital stays. Further investigation into use of ultrafiltration as a day care therapy is needed, in order to establish if patients at an early stage of CHF could benefit from this therapy.
Conflict of Interest and Funding
No conflict of interests. No funding was needed.
References
-
Bart BA, Goldsmith SR, Lee KL, Givertz MM, O'Connor CM, et al. (2012) Ultrafiltration in decompensated heart failure with cardiorenal syndrome. N Engl J Med 367: 2296-2304.
-
Prieur C (2013) Ultrafiltration lente en unité de soins intensifs cardiologiques chez des patients en insuffisance cardiaque grave (expérience multicentrique)
-
Costanzo MR, Guglin ME, Saltzberg MT, Jessup ML, Bart BA, et al. (2007) Ultrafiltration versus intravenous diuretics for patients hospitalized for acute decompensated heart failure. J Am Coll Cardiol 49: 675-683.
-
Dahle TG, Sobotka PA, Boyle AJ (2008) A Practical Guide for Ultrafiltration in Acute Decompensated Heart Failure. Congest heart Fail 14: 83-88.
-
De Maria E, Pignatti P, Patrizi G, Benenati PM, Ricci S, et al. (2010) Ultrafiltration for the treatment of diuretic-resistant, recurrent, acute decompensated heart failure: experience in a single center. J Cardiovasc Med (Hagerstown) 11: 599-604.