International Journal of Clinical Cardiology
Mycophenolate Mofetil-Induced Colitis in a Pediatric Heart Transplant Recipient
Lindy Moore1, Anita L Sengupta2 and Bibhuti Das3*
1Department of Pharmacy, Children's Medical Center, Dallas, Texas, USA
2Department of Pediatric Pathology, UTSW Medical Center, Dallas, Texas, USA
3Division of Pediatric Cardiology, UTSW Medical Center, Dallas, Texas, USA
*Corresponding author: Bibhuti Das, Associate Professor, Division of Cardiology, Department of Pediatrics, Children's Medical Center, UT Southwestern Medical center, Dallas, Texas, USA, Tel: 214-456-2333, Fax: 214-456-7758, E-mail: bdas99@hotmail.com
Int J Clin Cardiol, IJCC-2-055, (Volume 2, Issue 5), Case Report; ISSN: 2378-2951
Received: June 09, 2015 | Accepted: October 07, 2015 | Published: October 10, 2015
Citation: Moore L, Sengupta AL, Das B (2015) Mycophenolate Mofetil-Induced Colitis in a Pediatric Heart Transplant Recipient. Int J Clin Cardiol 2:055. 10.23937/2378-2951/1410055
Copyright: © 2015 Moore L, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Mycophenolate Mofetil (MMF) is a frequently used immunosuppressive medication in pediatric heart transplant (HT) patients. Although therapeutic monitoring of MMF has been an integral part of routine practice for many years, there is growing evidence that genetic variation in host can lead to adverse outcomes such as drug toxicities. Maintaining a reasonable balance between efficacy and toxicity in an individual patient remains one of the major challenges in pediatric HT recipients. Here we describe a pediatric HT recipient who has suffered MMF induced colitis despite acceptable therapeutic levels.
Introduction
Heart transplantation (HT) is a well-established therapy for end-stage otherwise untreatable heart disease. In recent years, outcome of pediatric HT have steadily improved with current 5-year overall survival rates estimated at 83% [1]. Despite these advances, children who receive HT experience a significant morbidity and mortality. There are many unknown clinical factors for considerable uncertainty in their outcome. One of the factors could be genetic variation in the host. There is growing evidence that genetic variation leads to differences in immune response, response to therapies and susceptibility to adverse outcomes such as drug toxicities [2].
Mycophenolate mofetil (MMF) is the most commonly prescribed adjunctive maintenance immunosuppressive medication in pediatric HT at the present time, with the use of azathioprine being on the decline. MMF is a prodrug that is rapidly hydrolyzed to the active form mycophenolic acid (MPA). MPA is a reversible inhibitor of inosine monophosphate dehydrogenase (IMPDH), which catalyzes purine synthesis. Because activated lymphocytes are dependent on the de novo synthesis of purine nucleotides, IMPDH inhibition causes decreased B-and T-cell proliferation and decreased antibody production [3]. MPA preferentially binds to IMPDH isoform type II, expressed in active lymphocytes.
The most common adverse effects of MMF in pediatric heart transplant recipients include watery diarrhea, nausea, abdominal cramping, and bone marrow suppression. MMF can lead to malignancy and infections (specifically CMV and herpes zoster infections), ulcerative esophagitis, reactive gastropathy, and graft-versus-host disease (GVHD)-like features in intestinal biopsies [4]. The MMF-related colitis is described in 2.7% adult renal transplant patients from a large series of 580 patients [5]. However, reports on the clinical course of colitis in pediatric heart transplant recipients are scarce. We describe a case of severe MMF induced colitis in a child 15 months after heart transplant.
Case
Our patient is a 3 year old Caucasian boy who underwent orthotopic HT at 14 months of age for hypoplastic left heart syndrome and Glenn failure. His induction immunosuppression consisted of basiliximab and methylprednisone, and for maintenance tacrolimus, MMF, and prednisone. Prednisone was discontinued at 12 months after HT. His tacrolimus dose was adjusted per level to maintain trough between 5-10 ng/mL. MMF dose was adjusted for MPA level with a therapeutic range between 1 and 3.5 mcg/ml. He was negative for cytomegalovirus (CMV) and Epstein-Barr virus (EBV) before HT, and received a CMV negative and EBV positive donor heart. He remained on antimicrobial prophylaxis with trimethoprim- sulfamethoxazole as per our institutional protocol. His serial polymerase chain reaction (PCR) for EBV and CMV were negative after HT.
His first 15 months post-HT was uneventful. In the 16th month he entered a 3 month period of intermittent non-bloody diarrhea alternating with constipation and severe weight loss (Figure 1). There was no fever, and his physical examination was only significant for nonspecific abdominal tenderness. The gastrointestinal panel was negative for aeromonas, campylobacter, clostridium difficile toxin A/B, plesiomonas, shigelloides, salmonella, yersinia enterocolitica, E. coli, cryptosporidium, cyclospora cayetanesis, entamoeba histolytica, giardia lamblia, norovirus and rotavirus. The only positive tests were for adenovirus from his stool, and transient disaccharides deficiency. Subsequent stool and blood PCR for adenovirus were negative. Elimination of dairy products reduced frequency of diarrhea from 12-15 times per day to 6-8 times. During this 3 month period, he was admitted to the hospital multiple times with recurrent vomiting, diarrhea, dehydration, and one episode of acute renal insufficiency. He underwent an esophagogastroduodenoscopy and a colonoscopy. His colon biopsy showed reactive changes with crypt dropout, regeneration, and focal active colitis extending from cecum to rectum and was attributed to MMF colitis (Figure 2A&B). The colonic tissue PCR for CMV, EBV and adenovirus were negative. There was no evidence of post-transplant lymphoproliferative disease. His MMF was stopped and azathioprine was started as adjunctive therapy for maintenance immunosuppression. Diarrhea improved within a week after MMF withdrawal and patient started gaining weight (Figure 1). He remained asymptomatic for the last 12 months as of this report.
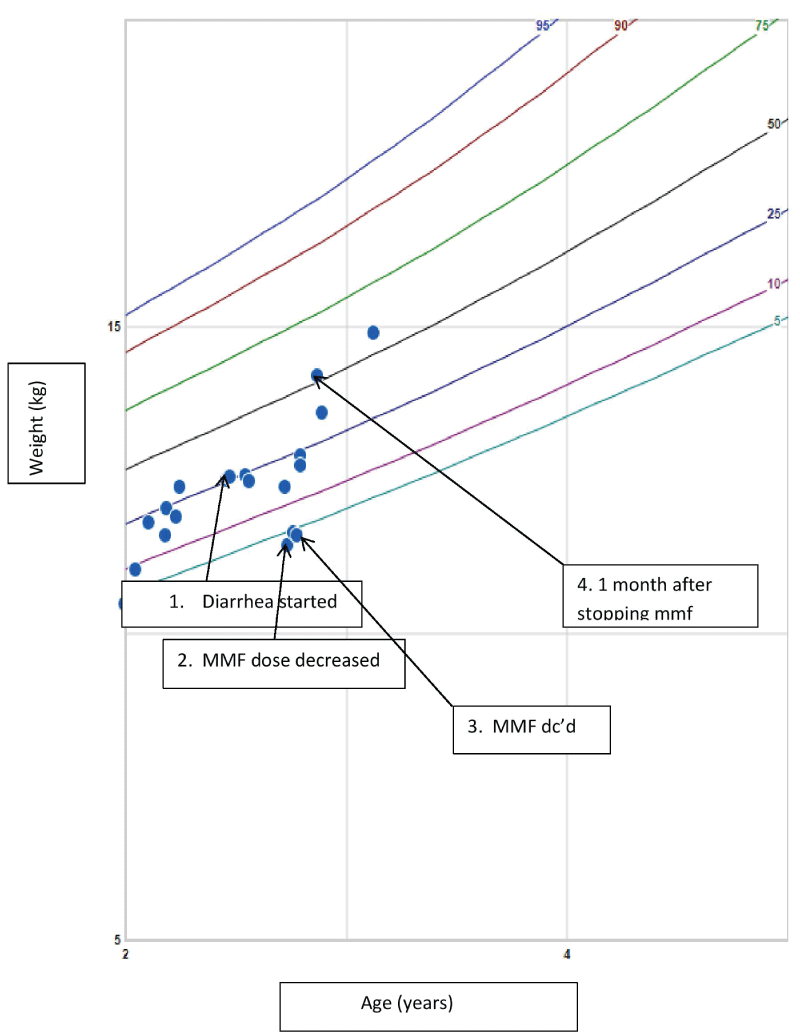
.
Figure 1: Growth chart demonstrating weight loss due to diarrhea and subsequent improvement after withdrawal of MMF.
View Figure 1
We monitored our patient every month with laboratory tests including cardiac biomarkers, end-organ functions and echocardiogram for allograft dysfunction. As our patient is clinically doing well, we did not repeat colonic biopsy nor re-challenged him with MMF. Our plan in future is if we see any evidence of either rejection or development of de novo donor specific antibodies, we will optimize his immunosuppression by reintroducing MMF instead of Azathioprine. We also plan to evaluate this patient for single nucleotide polymorphism (SNP) associated with genetic polymorphism and drug toxicities if we need to resume MMF in future.
Discussion
Recently, a case of late onset severe colitis attributed to MMF was described in an adult HT patient [6]. Pathak et al. [7] described a case of MMF induced colitis in a child after renal transplant which was confused with inflammatory bowel disease (IBD) [7]. Papadimitriou et al. [8] have described marked similarities in the histologic features of the MMF colitis and GVHD in children [8]. The common histopathological findings of MMF colitis GVHD and GVHD are prominent crypt enterocyte apoptosis accompanied by degenerative and regenerative changes (Figure 2A & B).
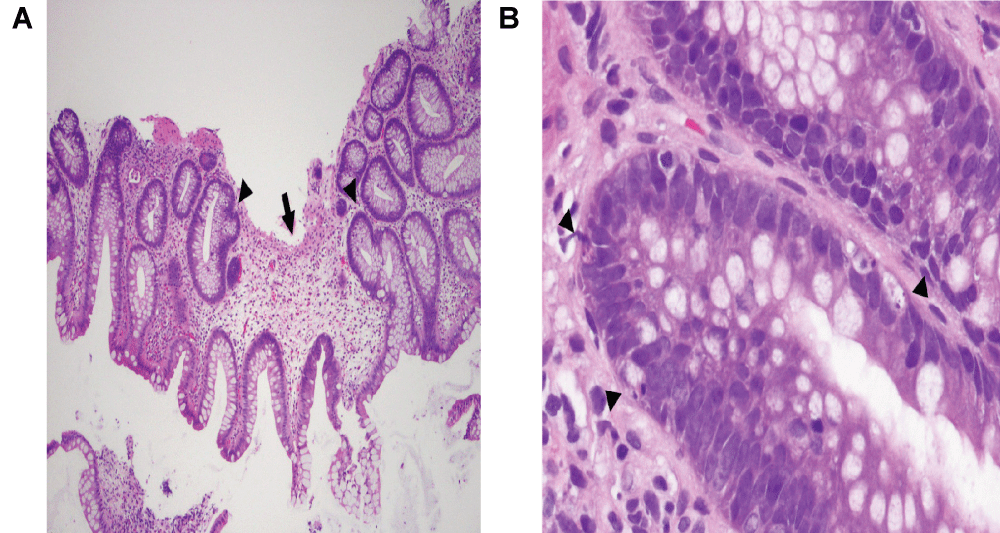
.
Figure 2A Colonic mucosa showing crypt dropout (arrow) and regenerative branching crypts (arrowheads)
Figure 2A Colonic mucosa (high power) showing apoptosis of crypt cells (arrowheads)
View Figure 2A & B
The pathogenetic mechanism through which MMF exerts its toxicity in the gastrointestinal tract is unknown. One of the proposed mechanisms by which MPA could lead to diarrhea is inhibition of the de novo purine synthesis pathway, thereby preventing gastrointestinal epithelial cell growth and replication [9]. It has been suggested that an MMF dose of > 600 mg/m2 every 12 hours is associated with higher plasma levels of MPA, and may be associated with a higher incidence of gastrointestinal side effects [10]. Our patient was on a dose of 740 mg/m2 every 12 hours and his MPA level was between 1-2 mcg/mL without any evidence of bone marrow suppression such as leucopenia or anemia, suggesting that the mechanism of MMF induced gastrointestinal toxicities could be multifactorial.
The pharmacogenomics study of MMF in 59 pediatric HT patients has shown that ABCC2 rs717620 GG phenotype was protective against gastrointestinal side effects due to MMF. The protective mechanism of this phenotype is attributed to decreased enterohepatic recirculation and lower intestinal MPA concentrations [11]. MPA is metabolized through phase 2 glucuronidation by UDP glucuronosyltransferases (UGTs). In pediatric renal transplant patients and adult HT patients, polymorphisms in UGT2B7 and UGT1A8 have influenced the metabolism, clearance and side effect profile of MMF [12-14].
MMF-induced colitis should be considered in the differential diagnosis of patients taking this drug. The discontinuation of MMF resulted in good clinical outcome with resolution of diarrhea and no evidence of allograft rejection in our patient. There was no correlation of MMF-induced colitis with MPA level in our patient and might suggest individual pharmacogenetic variations that may influence drug toxicities. In future, pharmacogenomics study of MMF as a routine clinical practice may provide opportunity to minimize rejection events while avoiding serious toxicities in pediatric heart transplant recipients.
References
-
Dipchand AI, Kirk R, Mahle WT, Tresler MA, Naftel DC, et al. (2013) Ten yr of pediatric heart transplantation: a report from the Pediatric Heart Transplant Study. Pediatr Transplant 17: 99-111.
-
Xie HG (2010) Personalized immunosuppressive therapy in pediatric heart transplantation: Progress, pitfalls and promises. Pharmacol Ther 126: 146-158.
-
de Jonge H, Naesens M, Kuypers DR (2009) New insights into the pharmacokinetics and pharmacodynamics of the calcineurin inhibitors and mycophenolic acid: possible consequences for therapeutic drug monitoring in solid organ transplantation. Ther Drug Monit 31: 416-435.
-
Liapis G, Boletis J, Skalioti C, Bamias G, Tsimaratou K, et al. (2013) Histological spectrum of mycophenolate mofetil-related colitis: association with apoptosis. Histopathology 63: 649-658.
-
de Andrade LG, Rodrigues MA, Romeiro FG, Garcia PD, Contti MM, et al. (2014) Clinicopathologic features and outcome of mycophenolate-induced colitis in renal transplant recipients. Clin Transplant 28: 1244-1248.
-
Curtin BF, Rachakonda VP, Von Rosenvinge EC (2014) Unusually late-onset mycophenolate mofetil-related colitis. Am J Health Syst Pharm 71: 1858-1861.
-
Phatak UP, Seo-Mayer P, Jain D, Selbst M, Husain S, et al. (2009) Mycophenolate mofetil-induced colitis in children. J Clin Gastroenterol 43: 967-969.
-
Papadimitriou JC, Cangro CB, Lustberg A, Khaled A, Nogueira J, et al. (2003) Histologic features of mycophenolate mofetil-related colitis: a graft-versus-host disease-like pattern. Int J Surg Pathol 11: 295-302.
-
Allison AC, Eugui EM (1993) The design and development of an immunosuppressive drug, mycophenolate mofetil. Springer Semin Immunopathol 14: 353-380.
-
Jacqz-Aigrain E, Khan Shaghaghi E, Baudouin V, Popon M, Zhang D, et al. (2000) Pharmacokinetics and tolerance of mycophenolate mofetil in renal transplant children. Pediatr Nephrol 14: 95-99.
-
Ohmann EL, Burckart GJ, Brooks MM, Chen Y, Pravica V, et al. (2010) Genetic polymorphism influence mycophenolate mofetil-related adverse events in pediatric heart transplant patients. J Heart Lung Transplant 29: 509-516.
-
Zhao W, Fakhoury M, Deschenes G, Roussey G, Brochard K, et al. (2010) Population pharmacokinetics and pharmacogenetics of mycophenolic acid following administration of mycophenolate mofetil in de novo pediatric renal-transplant patients. J Clin Pharmacol 50: 1280-1291.
-
Ting LS, Beenoit-Biancamano MO, Bernard O, Riggs KW, Guillemette C, et al. (2010) Pharmcogenetic impact of UDP-glucuronosyltransferase metabolic pathway and multidrug resistance-associated protein 2 transport pathway on mycophenolic acid in thoracic transplant recipients: an exploratory study. Pharmacotherapy 30: 1097-1108.
-
Woillard JB, Rerolle JP, Picard N, Rousseau A, Drouet M, et al. (2010) Risk of diarrhoea in a long-term cohort of renal transplant patients given mycophenolate mofetil: the significant role of the UGT1A8 2 variant allele. Br J Clin Pharmacol 69: 675-683.





