International Journal of Clinical Cardiology
Effects of Erythropoietin Administration on Cardiomyocytes in Landrace/Large White Pigs after Ventricular Fibrillation
Armando Faa1*, Gavino Faa1, Eliana Di Felice1, Pietro Pampaloni1, Daniela Fanni1, Daniele Scano1, Clara Gerosa1, Maria Elena Pais1, Panagiotis Vasileiou2, Dimitrios Barouxis2, Pavlos Lelovas2, Theodoros Xanthos3, Charalampos Pantazopoulos2, Apostolos Papalois4 and Nicoletta Iacovidou5
1Department of Surgery, Section of Pathology, University of Cagliari, Cagliari, Italy
2National and Kapodistrian University of Athens, Medical School, Athens, Greece
3European University Cyprus, School of Medicine, Cyprus
4ELPEN Experimental Center, Athens, Greece
5Neonatal Department, National and Kapodistrian University of Athens, Medical School, Aretaieion Hospital, Athens, Greece
*Corresponding author: Armando Faa, MD, PhD, Department of Surgery, Section of Pathology, University of Cagliari, Via Ospedale 46, 09100, Cagliari, Italy, E-mail: armando.faa@hotmail.it
Int J Clin Cardiol, IJCC-3-071, (Volume 3, Issue 1), Research Article; ISSN: 2378-2951
Received: September 18, 2015 | Accepted: January 23, 2016 | Published: January 28, 2016
Citation: Faa A, Faa G, Felice ED, Pampaloni P, Fanni D, et al. (2016) Effects of Erythropoietin Administration on Cardiomyocytes in Landrace/Large White Pigs after Ventricular Fibrillation. Int J Clin Cardiol 3:071. 10.23937/2378-2951/1410071
Copyright: © 2016 Faa A, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Aims: Evaluating the effect of erythropoietin (EPO) administration on the heart in a swine model of ventricular fibrillation and resuscitation.
Methods: Ventricular fibrillation was induced via a pacing wire forwarded into the right ventricle in 20 female Landrace/Large-White pigs, allocated into 2 groups; the experimental group treated with a bolus dose of 5,000 U/Kg recombinant human erythropoietin and the control group which was administered saline. Cardiopulmonary resuscitation (CPR) was performed immediately after drug administration. After 2 min of CPR, defibrillation was attempted. Animals were monitored, mechanically ventilated, extubated, observed and euthanized. At necroscopy, heart samples from the left ventricle were formalin-fixed, paraffin-embedded and routinely processed. Sections were stained with hematoxylin and eosin, and immunostained for S100B protein.
Results: At histology, pathological changes in cardiomyocytes and in cardiac vessels were detected in all animals. The most frequent lesions were edema, wavy fibers, coagulative necrosis, apoptosis, and contraction band necrosis. The most frequent vascular lesions were endothelial loss and detachment, endothelial apoptosis, and thrombosis. Immunostaining for S100B was observed in all animals. When the frequency of changes was compared between animals in the control and the EPO-treated group, cardiomyocyte necrosis and thrombosis showed a significant higher frequency in experimental group, whereas the intensity of S100B expression was higher in the control group.
Conclusions: In animals exposed to ventricular fibrillation, erythropoietin administration was not able to halt cell death of cardiomyocytes, nor progression of endothelial damage to thrombosis. The inverse trend of S100B protein expression confirms that S100B at cardiac level should be considered as an early protective response to a cardiac insult.
Keywords
Erythropoietin, Heart, Myocardium, Ventricular fibrillation, Resuscitation, Pigs
Introduction
The hematopoietic cytokine erythropoietin (EPO) is produced by the kidney in response to hypoxia. It stimulates erythroid progenitor cells to increase the number of mature red blood cells, improving the O2 carrying capacity. EPO and its receptor (EPO-R) are widespread, being highly expressed in the heart [1-3], suggesting a potential role for EPO in cardiac pathophysiology. EPO elevates blood pressure and increases the incidence of thrombosis [4]. In contrast to these effects, evidence indicates that EPO may act as a cellular survival factor in neurons [1]; this suggests that it may play a role in the repair and regeneration of brain and spinal cord after an injury, including the recruitment of stem cells into the region of damage [5]. The cardio-protective role of EPO is supported by in vitro data in rat cardiomyocytes, as well as in vivo in a rat model of myocardial infarction with reperfusion. EPO markedly prevented the apoptosis of cultured adult rat myocardiocytes subjected to 28 hours of hypoxia. Moreover, in a rat experimental model of coronary ischemia-reperfusion, the administration of recombinant human EPO for 7 days reduced cardiomyocyte loss by 50% [5].
The effects of EPO on human heart are dual: it increases thrombotic events and mortality rate in patients with ischemic heart disease, by increasing haematocrit levels, and significantly improves cardiac function following myocardial infarction, protecting cardiomyocytes from apoptosis. Surprisingly, the cardioprotective effects of EPO were seen in the absence of any increase in haematocrit, suggesting that EPO may directly protect the ischemic and infarcted heart [6].
On the basis of these data, this study was aimed at evaluating the possible cardioprotective effect of erythropoietin at cardiac level, in an experimental model of ventricular fibrillation in piglets.
Materials and Methods
The Directorate of Veterinary Services of Prefecture of Athens, Attica, Greece approved the protocol, according to Greek legislation regarding ethical and experimental procedures. Twenty female Landrace/Large-White pigs aged 10-15 weeks with average weight 19 ± 2 kg, all from the same breeder (Validakis, Athens, Greece) were studied. All animals were prepared in a standardized fashion in the research facility (ELPEN Experimental-Research Center, Pikermi, Greece) as previously described [7]. Initial sedation was achieved by intramuscular injection of ketamine hydrochloride (10 mg/kg), midazolam (0.5 mg/kg) and atropine (0.05 mg/kg). Anaesthesia was induced with an intravenous (iv) bolus dose of propofol (2 mg/kg) via the marginal auricular vein. The pigs were then intubated with a 4.5 mm cuffed endotracheal tube. Animals were mechanically ventilated with a volume-controlled ventilator (tidal volume 15 ml/kg and FiO2 0.21). End-tidal CO2 (ETCO2) was monitored by waveform capnography (Tonocap TC-200-22-01, Engstrom Division Instrumentarium Corp., Helsinki, Finland), and the respiratory frequency was adjusted to maintain ETCO2 between 35 and 40 mm Hg. A bolus dose of cis-atracurium (0.15 mg/kg) was administered to ascertain synchrony with the ventilator. Continuous infusion of propofol 150 μg/kg/min was used to maintain adequate anaesthetic depth and fentanyl 4 μg/kg to ensure analgesia. Cardiac rhythm and heart rate were monitored by electrocardiography (ECG), using leads I, II, III, aVR, aVL and aVF. Pulse oximetry (SpO2) was monitored continuously. Right carotid artery and right internal jugular vein were surgically prepared and catheterized under aseptic conditions. Aortic pressures were measured using a fluid-filled catheter (model 6523, USCI CR, Bart, Papapostolou, Athens, Greece) advanced via the right carotid artery into the thoracic aorta. Mean arterial pressure (MAP) was determined by electronic integration of the aortic blood pressure waveform. A catheter was inserted into the right atrium via the right jugular vein for continuous measurement of right atrial pressures. All catheters were calibrated before use and their correct position was verified by the presence of the typical pressure waveform.
Experimental protocol
After surgery the animals were allowed a 30-minute stabilization period, before baseline data were collected. VF was induced with a 9V ordinary cadmium battery via a pacing wire forwarded into the right ventricle through the cannulated right jugular vein, as previously described [4,8]. VF was confirmed by ECG and by a sudden drop in MAP. Mechanical ventilation and administration of anaesthetics were discontinued simultaneously with the onset of VF and the animals were left untreated for 8 min. A bolus dose of adrenaline (0.02 mg/kg) was then administered, and at the same time, the animals (n = 10 per group) were randomly treated either with a bolus dose of 5000 U/kg recombinant human erythropoietin (rh-EPO) (Eprex, Epoetin, Recombinant Human Erythropoietin Alfa; Janssen-Cilag, Athens, Greece) (experimental group) or saline as placebo (control group). Randomization was performed with the use of a sealed envelope, randomizing the animals to control group and experimental group. All drugs were injected via the marginal auricular vein, followed by a 10 ml saline flush to assist faster circulation of medications. The researchers were blinded to the animal’s allocation, until the experiment was completed and all haemodynamic and survival data were collected. CPR was commenced immediately after drug administration. Mechanical ventilation was resumed with 21% oxygen and automatic continuous precordial compressions were initiated (LUCAS, Jolife, Lund, Sweden), at a rate of 100 per minute following the two-minute cycles as per the ERC ALS guidelines algorithm [5]. After 2 min of CPR, defibrillation was attempted with a 4 J/kg monophasic shock. CPR was resumed for another 2 minutes after each defibrillation attempt. Further bolus doses of adrenaline (0.02 mg/kg) were administered every 4 minutes during CPR. Each experiment continued until ROSC or if asystole/pulseless electrical activity (PEA) occurred > 10 min after CPR initiation. ROSC was defined as the presence of a perfusing cardiac rhythm with a MAP of at least 60 mmHg for a minimum of 5 minutes. After ROSC, the animals were monitored closely and were mechanically ventilated for 6 hours, under general anaesthesia, at the pre-arrest settings. No other interventions (drugs, cardioversion or defibrillation attempts) were made after ROSC. After 6 hours, all catheters were removed; the animals were allowed to recover from anaesthesia, were extubated and transferred to their observation cages. They remained under observation for 48 hours after ROSC before euthanasia with an iv bolus dose of propofol 40 mg, followed by 2 gr thiopental iv. Experimental endpoints were ROSC and 48 h-survival.
Measurements
There was no difference in the baseline haemodynamic variables prior to arrest in the groups. Histological analyses were carried out in all treated animals. Three cardiac tissue samples, obtained from the right ventricle, from the septum and from the left ventricle, were fixed in 10% formalin, routinely processed and paraffin-embedded. The initial block was cut into 6-7 blocks about 2-3 mm wide. Five micron sections were deparaffinizated and hydrated to water. They were then stained with hematoxylin for 15 minutes, washed in running tap water for 20 minutes and counterstained with eosin for 15 seconds to 2 minutes. Finally slides were dehydrated in 95% absolute alcohol and cleared in xylene. A pathologist, blinded to the animal outcome, assessed all hematoxylin-eosin (H&E)-stained slices. Five high power fields were observed for the count of cardiomyocyte elementary lesions in each sample. The mean value was obtained as the mean of 15 counts for each heart analyzed.
The variables are expressed as mean ± standard deviation (SD) for each elementary lesion found at histology. Repeated Student t-tests were used to evaluate any possible statistically significant difference between the control group and the experimental subgroups.
Results
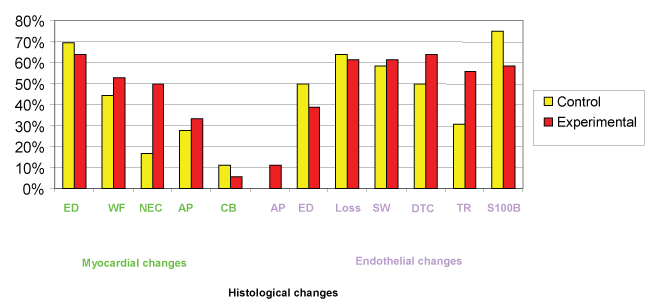
Pathological changes in the heart samples were detected in all pigs at histology. The most frequent lesions were observed in cardiomyocytes and in cardiac vessels (Figure 1).
.
Figure 1: Occurrence of myocardial and endothelial pathological changes in control (not receiving EPO) (yellow columns) and experimental animals treated with EPO (red columns). In axis x, the percentages of animals in which the single pathological heart changes were detected are reported. In axis y, in the left side the myocardial changes are reported in green, whereas in the right side, the endothelial changes are reported in violet. (ED: Edema, WF: Wavy Fibers, NEC: Necrosis, AP: Apoptosis, CB: Contraction Band, Loss: Endothelial Loss, SW: Swelling, DTC: Detachment, TR: Microthrombosis).
View Figure 1
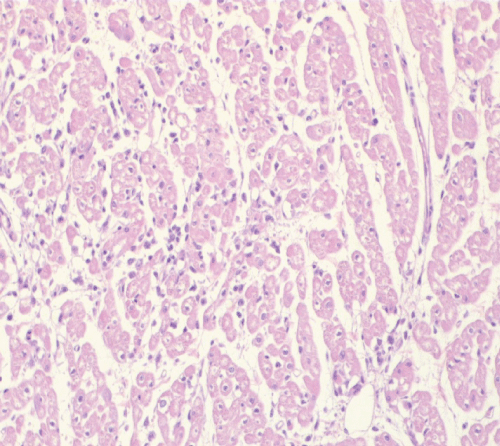
Oedema was the most frequent lesion observed, detected in 69% and 64% of control animals and EPO-treated animals respectively. The degree of cardiac oedema varied between animals, ranging from mild and focal in some cases, to intense and diffuse in other cases (Figure 2). No significant differences regarding the occurrence and the intensity of cardiac oedema was observed between the two groups of pigs.
.
Figure 2: EPO-treated pig. Histology of the heart shows a diffuse oedema, evidenced by a marked increase of the interstitial fluid, leading to dissociation of cardiomyocytes H&E.
View Figure 2
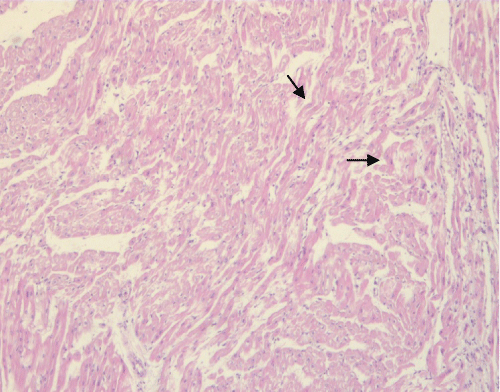
The abnormal architectural organization of cardiomyocytes in wavy fibres (Figure 3) represented also a frequent pathological lesion of cardiomyocytes in animals of both groups. This architectural change of cardiac cells was more frequently detected in EPO-treated animals, affecting 53% of them, than in the controls (44%), and the difference was statistically significant (p < 0.001).
.
Figure 3: Epo-treated pig: a panoramic view of an H&E-stained heart section shows a wavy arrangement of cardiac fibers (arrows).
View Figure 3
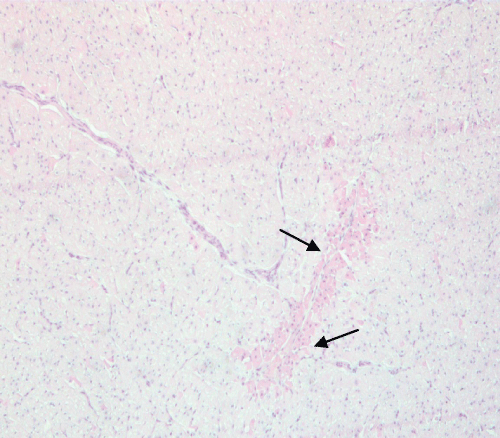
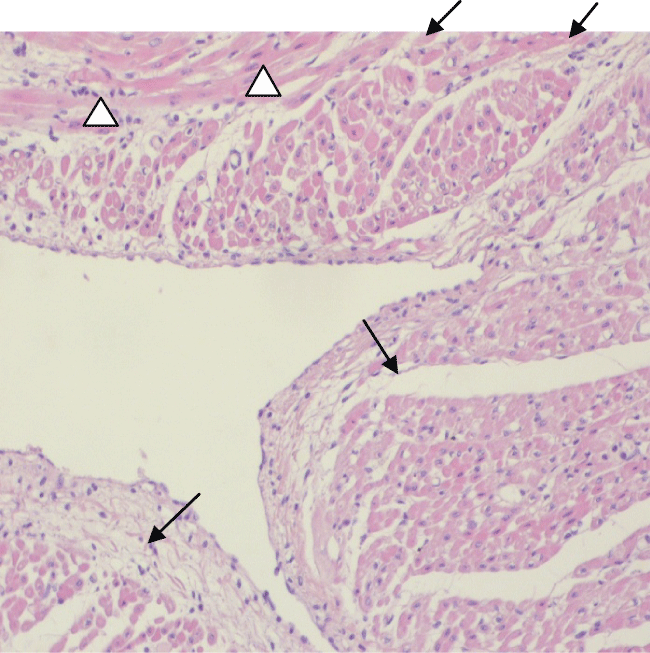
At low power, cardiomyocyte coagulative necrosis appeared as an eosinophilic zone, with irregular borders (Figure 4). At higher power, coagulative necrosis appeared as a group of roundish cardiomyocytes with strongly eosinophilic cytoplasm devoid of the nucleus, or with a dark picnotic nucleus (Figure 5). Areas of coagulative necrosis were detected more frequently in EPO-treated animals (50%) than in animals in the control group (17%).
.
Figure 4: EPO-treated pig: A low power view of cardiac tissue shows a hypereosinophilic irregular area of coagulative necrosis (arrows).
View Figure 4
.
Figure 5: EPO-treated pig heart. A large area of coagulative necrosis in the sub-endothelial region. At higher power, necrotic cardiomyocytes appear as roundish globules, with a hypereosinophilic cytoplasm and a shrunken dark nucleus (arrows). At the bottom, an area of surviving cardiomyocytes (arrowheads). L= ventricular lumen.
View Figure 5
Apoptosis of cardiomyocytes, appearing as isolated scattered cardiac cells detaching from adjacent cells and characterized by a hyperchromatic nucleus and a hypereosinophilic cytoplasm, was detected in 33% and in 28% of EPO-treated and animals of the control group, respectively.
Contraction band necrosis, appearing as fragmented cardiomyocytes with eosiniphilic cytoplasm, was only very rarely detected in the heart of the animals of both groups (11% in the control group and 6% in the EPO-treated animals).
Regarding endothelial lesions, endothelial loss was the lesion most frequently detected in both groups of animals. It was observed in 64% of EPO-treated animals and in 61% of animals of the control group. This pathological change was easily detected in the affected vessels, which showed absence of endothelial cells on the luminal surface. Endothelial loss was mainly detected in arterial vessels, and it was frequently associated with oedema of the perivascular areas of the myocardium.
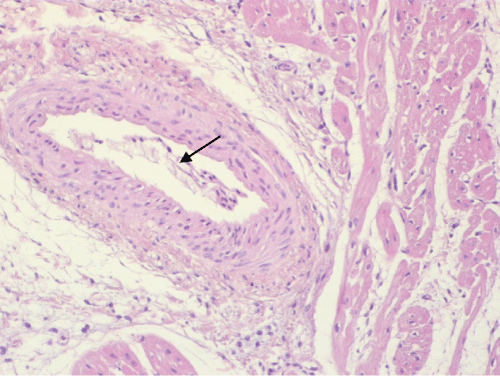
Endothelial detachment was observed in the majority of animals, appearing as strips of endothelial cells inside the vascular lumen (Figure 6) or as isolated endothelial cells detaching from the vascular wall. This lesion was more frequently detected in EPO-treated animals (64%) when compared with control animals (50%).
.
Figure 6: EPO-treated pig. A medium-sized cardiac vessel shows detachment of the endothelium from the arterial wall. Detached endothelial cells occupy the vessel lumen (arrow).
View Figure 6
The finding of endothelial cell apoptosis, appearing as shrunken endothelial cells with dark condensed chromatin, was restricted to a minority of EPO-treated animals (11%), whereas it was not observed in any of the control animals.
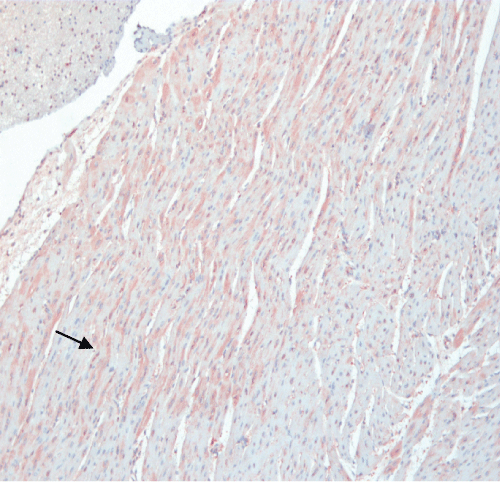
Immunoreactivity for S100B was detected in the vast majority of pigs of both groups. Immunostaining for S100B protein product was mainly localized in the cytoplasm of cardiomyocytes, appearing as immunoreactive granules of different size or as a homogenous immunostaining of the cardiomyocytic cytoplasm (Figure 7). Immunoreactivity for S100B was also detected in the cytoplasm of voluminous interstitial stromal cells surrounding the wall of intra-cardiac arteries. Significant differences were found in the incidence of immunostaining for S100B protein, detected in the heart of 58% of EPO–treated animals, versus 75% of animals of the control group. Immunostaining for S100 B in the majority of positive cases was mild and focal, and was detected in scattered cardiac cells.
.
Figure 7: EPO-treated pig heart. Immunhistochemistry for S100B protein shows a diffuse strong immunostaining in the majority of cardiomyocytes. Cardiac cells with a weavy arrangement (arrow) show a higher expression of S100B protein.
View Figure 7
Among all the cardiopathological changes described in this study, only cardiomyocyte necrosis and S100B immunoreactivity exhibited a significant statistical difference (p < 0.001) between animals that had received EPO and animals of the control group.
Discussion
Several experimental studies over the last years have focused on a better understanding of the role of EPO in human physiology. The initial role attributed to EPO and its receptor EPO-R is related to their indispensable function for survival, proliferation and differentiation of erythroid progenitor cells. EPO and EPO-R function are the primary mediators of a general protective response to tissue hypoxia. In addition, it maintains adequate tissue oxygenation through adjustments of circulating red cell mass via a hormonal feedback-control system involving kidneys and bone marrow cells. Subsequently, a role in upregulation of blood pressure and in predisposing to a higher incidence of thrombosis has been reported in subjects submitted to EPO treatment [4].
In contrast to these events associated with EPO treatment, accumulating evidence indicates that EPO is a cellular survival factor in neurons [1], supporting a role for EPO in repair and regeneration after brain and spinal cord injury [5].
Conflicting results have been reported regarding the effects of EPO on cardiomyocytes. In in vitro and experimental studies, there are data suggesting a protective role of EPO against apoptosis on cardiomyocytes subjected to hypoxia or to ischemia [5]. In humans, increased thrombotic events and mortality rates was reported with EPO in patients with ischemic heart disease, apparently by increasing the haematocrit [9]. On the other hand, EPO may significantly improve cardiac function following myocardial infarction, protecting cardiomyocytes against apoptosis, translating into more viable myocardium and less ventricular dysfunction [10-12]. These cardioprotective effects of EPO were reported without an increase in the peripheral red blood cell levels, suggesting that EPO may directly protect the ischemic and infarcted heart [6]. Several studies have recently shown that EPO also activates potent cell survival mechanisms during ischemia and reperfusion through genomic and non-genomic signaling pathways in a broad array of organs and tissues including the heart [8].
In a recent clinical study, administration of EPO in victims of out-of-hospital cardiac arrest was associated with higher rates of ROSC and 24-hour survival [10]. These clinical observations, based though on a small sample size, support the hypothesis that EPO preserves myocardial distensibility; this may lead to hemodynamically more effective chest compression resulting in higher resuscitation and survival rates [12]. Unfortunately previous experiments on the effects of EPO on myocardial protection during resuscitation [12] did not examine cardiac histology.
We have recently reported, in an experimental model of normocapnic hypoxia in newborn piglets, the association between the incidence of coagulative necrosis and the time of recovery following hypoxia. Coagulative necrosis was identified as the most important pathological change occurring in the heart of these piglets following hypoxia [13].
In the present study, we have shown that EPO administration in pigs exposed to VF has no positive effects on heart and in particular on cardiomyocytes and on endothelial cells. EPO administration did not protect the heart from oedema, as this pathological lesion was detected in similar rate in pigs of the control and experimental group. The increase of interstitial fluid in the heart tissue is generally considered to be the consequence of a vascular dysfunction or of the loss of the endothelial barrier, which may cause leakage of plasma into the heart leading to dissociation of cardiac cells. The wavy arrangement of cardiomyocytes, an index of hypoxic damage, was more frequent in EPO-treated animals, suggesting thus the absence of a protective role of EPO on cardiomyocytes in this experimental model. But the lesion that underlines the absence of any protective role for EPO is the coagulative necrosis of cardiomyocytes. This pathological change was significantly higher in EPO-treated animals, indicating a possible negative role for EPO.
The absence of a protective role of EPO on the heart is indicated in this study, by the occurrence of endothelial lesions in cardiac vessels of EPO-treated pigs in similar rate compared to control animals. Regarding apoptosis of endothelial cells, this lesion was restricted to the EPO-treated animals, suggesting an aggravating role for EPO on the endothelial barrier.
The presence of S100B immunoreactivity observed in heart cells in our experimental model deserves a particular comment. It is now generally accepted that S100B protein expression at cardiac level should be considered as a very fast protective response to hypoxia. In a previous study from our group, in an experimental animal model of hypoxia and oxygenation, the highest levels of cardiac S100B-positivity was detected in piglets recovering in less than 15 minutes [14].
The occurrence, in the present study, of S100B cardiac expression in a significant higher rate (75%) in the heart of control animals - characterized by lower levels of cardiomyocyte necrosis - confirms that the expression of this protein is a reliable indicator of early myocardial insult.
In conclusion, our study shows that EPO does not protect cardiomyocytes when exposed to ventricular fibrillation. The limitations of our study rely on the limited number of experimental animals. Further studies, carried out on a larger number of animals, are needed in order to confirm the possible aggravating role of EPO in causing cardiomyocyte necrosis in this clinical setting.
References
-
Xanthos T, Vasileiou PV, Kakavas S, Syggelou A, Iacovidou N (2011) The potential role of erythropoietin as a pleiotropic agent in post-cardiac arrest syndrome. Curr Pharm Des 17: 1517-1529.
-
Depping R, Kawakami K, Ocker H, Wagner JM, Heringlake M, et al. (2005) Expression of the erythropoietin receptor in human heart. J Thorac Cardiovasc Surg 130: 877-878.
-
Paffett-Lugassy N, Zon LI (2002) Epo-EpoR signaling not required for cardiovascular or neural development. Blood 100: 2277.
-
Rao SV, Stamler JS (2002) Erythropoietin, anemia, and orthostatic hypotension: the evidence mounts. Clin Auton Res 12: 141-143.
-
Calvillo L, Latini R, Kajstura J, Leri A, Anversa P, et al. (2003) Recombinant human erythropoietin protects the myocardium from ischemia-reperfusion injury and promotes beneficial remodeling. Proc Natl Acad Sci USA 100: 4802-4806.
-
Parsa CJ, Matsumoto A, Kim J, Riel RU, Pascal LS, et al. (2003) A novel protective effect of erythropoietin in the infarcted heart. J Clin Invest 112: 999-1007.
-
Xanthos T, Lelovas P, Vlachos I, Tsirikos-karapanos N, Kouskouni E, et al. (2007) Cardiopulmonary arrest and resuscitation in Landrace/Large White swine: a research model. Lab Anim 41: 353-362.
-
Parsa CJ, Kim J, Riel RU, Pascal LS, Thompson RB, et al. (2004) Cardioprotective effects of erythropoietin in the reperfused ischemic heart: a potential role for cardiac fibroblasts. J Biol Chem 279: 20655-20662.
-
Fishbane S, Besarab A (2007) Mechanism of increased mortality risk with erythropoietin treatment to higher hemoglobin targets. Clin J Am Soc Nephrol 2: 1274-1282.
-
Grmec S, Strnad M, Kupnik D, Sinkovic A, Gazmuri RJ (2009) Erythropoietin facilitates the return of spontaneous circulation and survival in victims of out-of-hospital cardiac arrest. Resuscitation 80: 631-637.
-
Gazmuri RJ, Ayoub IM, Radhakrishnan J (2010) NHE-1 inhibitors and erythropoietin for maintaining myocardial function during cardiopulmonary resuscitation. Signa Vitae 5: 6-13.
-
Singh D, Kolarova JD, Wang S, Ayoub IM, Gazmuri RJ (2007) Myocardial protection by erythropoietin during resuscitation from ventricular fibrillation. Am J Ther 14: 361-368.
-
Xanthos T, BassiaKou E, Koudouna E, Rokas G, Goulas S, et al. (2009) Combination Pharmacotherapy in the treatment of experimental cardiac arrest. Am J Emerg Med 27: 651-659.
-
Faa A, Xanthos T, Papalois A, Locci A, Pampaloni P, et al. (2013) S100B immunoreactivity: a new marker of hypoxia-related cardiac damage in newborn piglets. J Matern Fetal Neonatal Med 26 Suppl 2: 72-76.





